Abstract
Introduction: Buerger’s disease (BD) generally affects men, young people, and smokers, but it can also affect women. Its incidence is rare in Latin America.
Case report: A 40-year-old Colombian woman, active smoker and user of psychoactive substances, attended the emergency department of a tertiary care center due to symptoms of 3 days consisting of retraction of the corner of the mouth, drooling, and involuntary tongue movements. The patient, who had a history of uncontrolled diabetes and recent acute ischemia of the right upper limb due to acute thrombosis, required surgical management and subsequent use of oral anticoagulation. She later developed necrotic changes in the distal phalanges of the right hand that required ablative therapy. Since age, sex and limb involvement were not typical for BD, collagenosis, vasculitis or thrombophilia were ruled out, but after excluding these disorders, BD with atypical features was considered. The patient was discharged with oral anticoagulation, aspirin, combined analgesia, physiotherapy, and recommendation for smoking cessation.
Conclusions: Age, sex, smoking and comorbidities such as diabetes are risk factors for BD. Imaging and histopathology are the gold standard for the definitive diagnosis of this entity. Multidisciplinary management, lifestyle changes, smoking cessation, pain control, good wound healing and social support are key aspects for better clinical outcomes in patients with BD.
Resumen
Introducción. La enfermedad de Buerger (EB) afecta generalmente a hombres, jóvenes y fumadores, y aunque también puede afectar a mujeres, su incidencia es rara en Latinoamérica.
Presentación del caso. Mujer colombiana de 40 años, fumadora activa y consumidora de sustancias psicoactivas, quien acudió al servicio de urgencias de una institución de tercer nivel por síntomas de 3 días de evolución consistentes en retracción de la comisura bucal, babeo y movimientos involuntarios de la lengua. La paciente, que tenía antecedente de diabetes no controlada y reciente isquemia aguda de miembro superior derecho por trombosis aguda, requirió manejo intervencionista y subsecuente uso de anticoagulación oral. Posteriormente, desarrolló cambios necróticos en falanges distales de mano derecha y requirió terapia ablativa. Dado que la edad, el género y la afectación de las extremidades no eran típicos para EB, se procedió a descartar colagenosis, vasculitis o trombofilia, pero tras excluir estas patologías se consideró EB con características atípicas. La paciente fue dada de alta con anticoagulación oral, aspirina, analgesia combinada, fisioterapia y recomendación de suspender el consumo de tabaco.
Conclusiones. La edad, el género, el tabaquismo y las comorbilidades como diabetes son factores de riesgo para EB. La imagenología e histopatología son estándar de oro en el diagnóstico definitivo de esta entidad. El manejo multidisciplinario, los cambios en el estilo de vida, la cesación del tabaquismo, el control del dolor, la buena cicatrización de heridas y el apoyo social son aspectos importantes para obtener mejores resultados clínicos en pacientes con EB.
Introduction
Buerger’s disease (BD), also known as thromboangiitis obliterans (TAO), is a non-atherosclerotic inflammatory disorder of unknown etiology that affects small and medium-sized vessels of the extremities. It usually develops in men around 40 years of age and is strongly associated with smoking (1). During its acute phase, highly inflammatory thrombi are observed, as well as inflammation of the blood vessels that is not as pronounced as in other types of vasculitis, and the internal elastic lamina is preserved. However, given the mild inflammatory changes associated with blood vessels, BD is indeed considered a vasculitis (2). The prevalence of this disease varies depending on the geographic location, being very rare in Latin America (3), and even though it is uncommon, recent data reports an increased prevalence among women (4-6).
Clinical diagnostic criteria include current or recent history of tobacco use and presence of distal-extremity ischemia (indicated by claudication, pain at rest, ischemic ulcers or gangrene) documented by non-invasive vascular testing (7,8). It is also necessary to rule out autoimmune diseases, thrombophilia and diabetes mellitus, as well as a proximal source of emboli using echocardiography or arteriography, which, in turn, will allow confirming consistent findings in the clinically affected limbs and ruling out atherosclerosis (6,8).
BD generally begins with distal vascular ischemia and usual clinical signs and symptoms include migratory thrombophlebitis (43%), Raynaud’s phenomenon (40%), claudication (70%), pain at rest (91%), and sensory symptoms in the limbs (77%). Moreover, two or more extremities are commonly involved, and its progression is cephalad (8).
Smoking cessation is the cornerstone of treatment, and it has been shown to prevent amputations. Other strategies include daily hand and foot hygiene, injury prevention, physical activity, pain management, and pharmacological vasospasm prevention (9).
This article presents the case of 40-year-old Colombian woman, active smoker and user of psychoactive substances, who attended the emergency department of a tertiary care center due to symptoms of 3 days consisting of retraction of the corner of the mouth, drooling, and involuntary tongue movements. The patient, who had a history of uncontrolled diabetes and recent acute ischemia of the right upper limb due to acute thrombosis, required surgical management and subsequent use of oral anticoagulation. She later developed necrotic changes in the distal phalanges of the right hand that required ablative therapy.
Case presentation
This is the case of a 40-year-old Colombian woman, housewife, from a low-income household, with a personal history of active heavy smoking (50 packs years - PYI), use of psychoactive substances, and diabetes, as well as a recent history (September 2020) of acute right upper extremity ischemia caused by thrombosis on the brachial artery, extending to the radial and ulnar arteries, which required thrombectomy and subsequent use of oral anticoagulants.
She was admitted to the emergency department of a tertiary care center on October 3, 2020, due to a 3-day history of self-limiting episodes lasting 5-20 minutes, retraction of the corner of the mouth, drooling, and abnormal involuntary tongue movements, without loss of consciousness, sphincter relaxation, gaze deviation or tongue biting. She also experienced other clinical signs including stabbing pain in the right upper limb associated with progressive worsening with acrocyanosis, slow scarring, and necrosis in the second, fourth and fifth fingers of the right hand (Figure 1). The patient reported a positive Allen test.
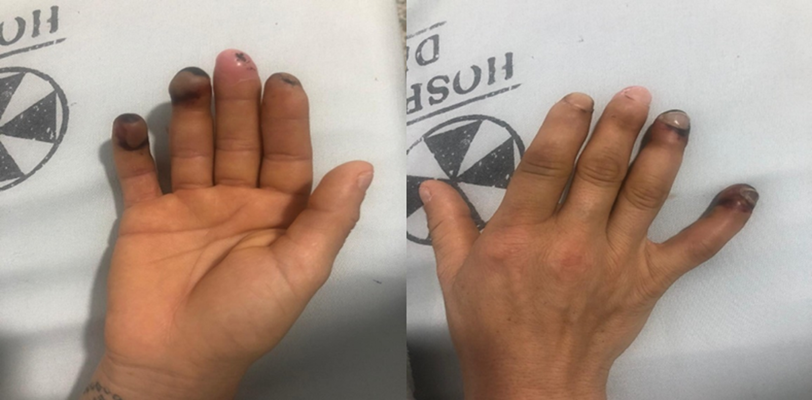
Figure 1. Dry gangrene in the second, fourth, and fifth fingers of the right hand.
Source: Document obtained during the study.
Consequently, she was immediately admitted to the hospital. During the first 24 hours, initial analytical studies were performed (October 3, 2020), and the results were normal. Differential diagnoses were considered, including systemic embolization or thrombophilia, BD, vasculitis, atherosclerosis, and vasculitis related to systemic lupus erythematosus. Her infectious profile was negative, and autoimmunity and thrombophilia studies were inconclusive (Table 1). A possible source of systemic embolism was ruled out using echocardiography and carotid-vertebral Doppler; both tests were performed on October 6, 2020.
Table 1. Serological studies performed.
|
Study |
Results |
|
Hemoglobin |
10 g/dL |
|
Leukocytes |
6 600/mm³ |
|
Platelets |
234 000/mcL |
|
Differential count |
|
|
Neutrophils |
3 860 (58.4%) |
|
Lymphocytes |
2 370 (35.9%) |
|
Monocytes |
380 (5.7%) |
|
Eosinophils |
40 (0.6%) |
|
Basophils |
10 (0.15%) |
|
Acute phase reactants |
|
|
Erythrocyte sedimentation rate |
14 mm/hour |
|
C-reactive protein |
26.5 mg/L |
|
Coagulation |
|
|
Prothrombin time |
11.9 s |
|
International Normalized Ratio |
1.06 |
|
Partial thromboplastin time |
36.6 s |
|
Renal function |
|
|
Creatinine |
0.76 mg/dL |
|
Blood urea nitrogen |
6.6 mg/dL |
|
Liver function |
|
|
Aspartate aminotransferase |
25 U/L |
|
Alanine transaminase |
34 U/L |
|
Total bilirubin |
0.58 mg/dL |
|
Direct bilirubin |
0.07 mg/dL |
|
Indirect bilirubin |
0.51 mg/dL |
|
Lactate dehydrogenase |
142 U/L |
|
Electrolytes |
|
|
Sodium |
136 mEq/L |
|
Potassium |
3.7 mEq/L |
|
Magnesium |
2.1 mg/dL |
|
Chlorine |
102 mEq/L |
|
Infectious |
|
|
Venereal disease research laboratory test |
Non-reactive |
|
Hepatitis B |
0.68 |
|
Hepatitis C |
0.08 |
|
Human immunodeficiency virus |
Negative |
|
Metabolic |
|
|
Beta-human chorionic gonadotropin |
0.41mUI/mL |
|
Random blood glucose |
190 mg/dL |
|
Glycosylated hemoglobin |
14.26% |
|
Thyroid stimulating hormone |
3.8 mUI/L |
|
Total cholesterol |
142 mg/dL |
|
Triglycerides |
300 mg/dL |
|
Low-density lipoprotein |
46 mg/dL |
|
High-density lipoprotein |
36 mg/dL |
|
B12 vitamin |
203 pg/mL |
|
Autoimmunity |
|
|
Complement C3 |
107.2 mg/dL |
|
Complement C4 |
29.2 mg/dL |
|
Rheumatoid factor |
Negative |
|
Antinuclear antibodies |
Negative |
|
Cytoplasmic-antineutrophil cytoplasmic autoantibody |
Negative |
|
Perinuclear anti-neutrophil cytoplasmic antibodies |
Negative |
|
Cryoglobulins |
Negative |
|
Anti-myeloperoxidase antibodies |
Negative |
|
Anti-proteinase 3 antibodies |
Negative |
|
Thrombophilias |
|
|
Lupus anticoagulant |
24.3 seg |
|
Anti-cardiolipin IgM antibodies |
16.07 U/mL |
|
Anti-cardiolipin IgG antibodies |
2.01 U/mL |
|
IgM antiphospholipid antibodies |
6.95 U/mL |
|
IgG antiphospholipid antibodies |
7.08 U/mL |
|
Protein S coagulation activity |
87% |
|
Protein C coagulation activity |
191% |
|
Beta-2-Glycoproteins IgM |
24.4 U/mL |
|
Beta-2-Glycoproteins IgG |
2.68 U/mL |
|
Homocysteine |
6.8 mcmol/L |
IgM: Immunoglobulin M; IgG Immunoglobulin G.
Source: Own elaboration.
Magnetic resonance imaging was performed on the fifth day of hospital stay (October 8, 2020) due to suspected focal seizure syndrome, which showed an area of ischemia in the right frontal region with petechial hemorrhagic transformation. This was considered an event related to a right upper limb arterial thrombosis that could have migrated and cause brain injury. However, there was no contraindication for anticoagulation. Subsequently, magnetic resonance angiogram showed no evidence of vascular injury, and video telemetry revealed no potentially epileptiform activity.
During the same week, considering the patient’s history of extensive and repetitive thrombosis, a skin biopsy of the affected fingers was performed, which revealed late findings of necrosis. Arterial and venous duplex studies of the upper and lower limbs were performed and showed diffuse atheromatosis.
On October 18, 2020, two weeks after being admitted to the hospital, the patient presented with gangrene in the fingers, requiring ablative therapy with exploration of vessels in the right forearm plus sympathectomy, as well as amputation of the distal phalanges of the little and ring fingers. She was given oral anticoagulation with warfarin and a calcium channel antagonist (nifedipine 30 mg BID) to prevent vasospasm crisis and favor clinical wound healing. She was also advised to quit smoking in order to assess her progress and avoid further amputations.
On October 23, 2020, she complained of a throbbing headache in the temple, so a Doppler of the temporal arteries was performed on the same day, with normal results, as well as angiography of the thorax vessels that ruled out large vessel involvement.
In order to support the diagnosis of BD and to assess the critical findings described in non-symptomatic places, considering the risk of severe vasospasm evidenced in the arteriography of the four extremities, a CT angiography of the extremities was performed on October 25, 2020, in which absence of opacification of the left radial artery in the distal half of the forearm (Figure 2) was observed, while other segments evaluated revealed no other relevant findings.
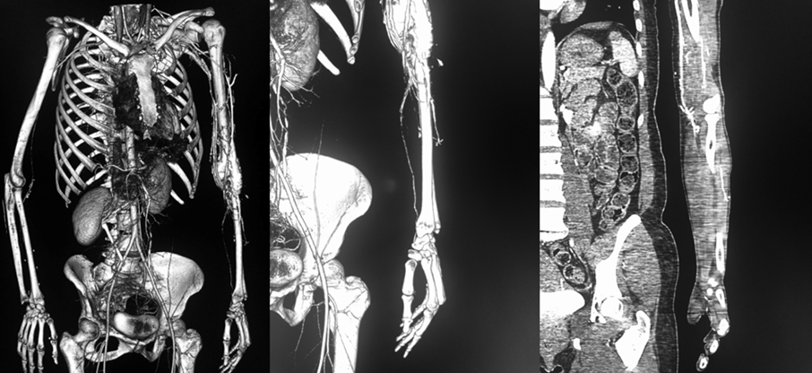
Figure 2. Angiotomography of the extremities
Left upper limb: absence of opacification of the left radial artery in the distal half of the forearm
Source: Document obtained during the study.
Finally, in November 2020, clinic wound healing was completed, discharge was ordered, and a combination therapy with oral anticoagulation with warfarin 5mg once daily, aspirin 100mg once daily, nifedipine 30 mg twice a day, titration of analgesia with pregabalin 150mg twice a day, and acetaminophen 1g three times a day was prescribed, in addition to physical therapy with connective tissue manipulation (CTM) and interferential therapy (IT) three times per week for three months, and smoking cessation.
Discussion
In the present case, some characteristics of the medical condition stand out and deserve to be examined in light of current knowledge. Initially, collagen disease, vasculitis or thrombophilia were suspected, since the age, sex, and limb involvement of the patient are not typical of BD. Although these findings could have ruled out a BD diagnosis at first, most clinical and imaging features associated with this disorder were demonstrated and, after contemplating other causes, they were finally considered atypical characteristics of BD.
Regarding the atypical findings, BD has been increasingly described in women, 11-30% in a case series of 26 patients (10), and this particular increase in prevalence in this group has been attributed to smoking (11,12). It has also been reported that the maximum incidence of this disease occurs in the fourth decade of life, followed by the fifth and second decades of life (13), and most of the cases present with detectable lower extremity involvement. In the study by Sasaki et al. (14), in which the medical records of 825 patients with thromboangiitis obliterans were analyzed, isolated lower extremity involvement was found in 74% of cases, while involvement of both lower and upper extremities was observed in 20.2%, and signs and symptoms in the upper extremities only in 5.1%.
Moreover, the involvement of cerebral vessels in BD is rare and its confirmation has varied. Retrospective case evaluations have found an association with atherosclerosis or vasculitis, while the characteristic findings of granular atrophy of the cortex, whitish arteries of the cortical surface, prominent meningeal vascular anastomoses, and narrowing of the arterial lumen by connective tissue are uncommon. The cerebral form of the disease is closely related to tobacco use (15) and approximately 15% of BD patients experience cerebrovascular events before exhibiting an apparent peripheral vascular disease; the duration of these cerebral or peripheral symptoms can last up to 20 years (16). In the case reported here, this event could have represented an epiphenomenon or a specific syndrome of premature vascular disease.
On the other hand, marijuana use has been reported as a cause of BD in several cases, but its role as a causative agent has often been mistaken for concomitant smoking (17). When associated with marijuana, BD seems to affect upper extremities half as often, although involvement of the lower and upper limbs has also been described (18). Cases of arteritis associated with marijuana use have been reported, and most patients have concomitantly been identified as smokers (18-20). Similar to BD associated with tobacco use, the majority of cases of arteritis associated with marijuana use have been described in younger patients, more often males, who have more frequent involvement of the lower limbs rather than the upper ones (19).
However, unlike BD, marijuana-associated arteritis has been described to develop earlier and with lower amounts than with tobacco. Most of the cases that reported arteritis associated with marijuana consumption involved larger arteries than in classic BD associated with tobacco (21). In this case, the disease was limited to small and medium-sized arteries, which may suggest that marijuana use may have led to a pathological syndrome that is identical to or mimics BD.
Clinical diagnosis should ideally be supported when all clinical criteria are met following the Shionoya criteria (7) or the Olin criteria (8), even if they are not universally accepted. The most commonly used are the Olin criteria (8), which include onset before the age of 45; current (or recent) tobacco use; distal extremity ischemia (infrapopliteal and/or intrabrachial), such as claudication, pain at rest, ischemic ulcers, and gangrene documented with noninvasive testing; laboratory tests to exclude autoimmune or connective tissue diseases and diabetes mellitus; ruling out a proximal source of emboli with echocardiography and arteriography; and demonstrating consistent arteriographic findings in the involved and clinically noninvolved limbs (8). Biopsy is suggested for definitive diagnosis in the presence of unusual characteristics such as age greater than 45 years, large artery involvement, diabetes, positive antinuclear antibodies, or anticardiolipin antibodies (5,6).
In the present case, not all clinical criteria were met. A biopsy was performed, but the patient’s medical condition was consistent with imaging findings and complementary serological studies that ruled out other causes, including atherosclerotic-occlusive disease, collagen disease, and thrombophilia.
If possible, histological evaluation of the affected vessels should be conducted in search of pathognomonic histological findings (5,6), which typically include inflammatory thrombi affecting both arteries and veins, although histology varies depending on the phase of the disease. In the acute phase, findings are more likely to be diagnostic and comprise occlusive inflammatory thrombus developing within the vessel. Polymorphonuclear leukocytes, microabscesses, and multinucleated giant cells may also be present. Furthermore, intermediate phase findings are suggestive of the diagnosis, and they are characterized by progressive organization of the thrombus. In the chronic phase, only organized thrombus and fibrosis remain, and the inflammation is no longer present (8).
Nevertheless, it is rare that vascular specimens are available for histological analysis in patients with suspected BD (5). In this case, a biopsy was performed but it failed to characterize the involved vascular lumen and showed late changes of necrosis.
Imaging studies may also be useful for confirmation of occlusive disease and ruling out other causes, among them, potential emboli. Gadolinium-enhanced magnetic resonance (MR) angiography or multidetector computed tomographic (CT) angiography can be used to observe the vasculature (6). The clinical and angiographic criteria considered for diagnosis include involvement of small and medium sized vessels in upper or lower extremities; absence of angiographic evidence of atherosclerotic disease or thromboembolism; segmental occlusion with diseased segments interspersed between normal-appearing segments; and “tree root” or “spider’s leg” shape of collaterals from the point of abrupt arterial occlusion (22).
BD affects small arteries in men more than women, and particularly heavy smokers. CT angiography can detect arterial wall thinning and thickening in addition to stenosis and occlusion, and findings usually reveal normal proximal arteries and absence of distal opacification due to inflammation (23). The patient in this case complied with the findings of ischemia of the upper limbs, absence of evidence of proximal atherosclerotic disease, and segmental involvement of small and medium arteries with occlusion.
Amputation is a common long-term complication of BD, and complete smoking cessation is the most effective treatment to improve symptoms and reduce the risk of amputation (24). In this case, even with less than a year after the onset of clinical symptoms, the disease progressed very quickly and exacerbation occurred early in the clinical course of the disease, requiring early surgical management, while placing emphasis on smoking cessation to avoid disease progression and new amputations.
Regarding the role of diabetes mellitus in BD, it has been described that it can accelerate the pathological process, without being associated as a cause of obliterative atherosclerosis (25), and in the absence of obliterative atherosclerosis, diabetes mellitus cannot cause small artery disease (26). In the case described above, the finding of poorly controlled diabetes was coincidental, and imaging findings consistent with vascular complications of diabetes were excluded.
Conclusions
In BD, age, sex, history of smoking, and complications such as diabetes are relevant, albeit not definitive factors, and imaging and histopathologic evaluation are significant as decisive diagnostic criteria. The association of marijuana use with the development of BD has become more widely known. Emphasis should be placed on obtaining a detailed history of the patient with evidence of digital ischemia, whether they are active smokers or not.
Multidisciplinary management, emphasis on lifestyle changes, smoking cessation, pain control, wound healing, and support of the social network are necessary to achieve better clinical outcomes.
Ethical considerations
The informed consent of the patient was obtained. This case report was approved by the research committee of the Hospital Universitario de La Samaritana E.S.E. Bogotá D.C., Colombia.
Conflict of interests
None stated by the authors.
Financing
None stated by the authors.
Acknowledgements
To the Hospital Universitario de La Samaritana E.S.E.
References
1.Hartmann P, Mohokum M, Schlattmann P. The association of Raynaud syndrome with thromboangiitis obliterans--a meta-analysis. Angiology. 2012;63(4):315-9. https://doi.org/bx2tvw.
2.Piazza G, Creager MA. Thromboangiitis obliterans. Circulation. 2010;121(16):1858-61. https://doi.org/cf3gmz.
3.Jimenez-Paredes CA, Cañas-Davil CA, Sanchez A, Restrepo JF, Peña M, Iglesias-Gamarra A. Buerger’s disease at the ‘San Juan De Dios’ Hospital, Santa Fe De Bogota, Colombia. Int J Cardiol. 1998;66(Suppl 1):S267-72. https://doi.org/b545r9.
4.Hida N, Ohta T. Current status of patients with buerger disease in Japan. Ann Vasc Dis. 2013;6(3):617-23. https://doi.org/jnhv.
5.Rivera-Chavarría IJ, Brenes-Gutiérrez JD. Thromboangiitis obliterans (Buerger’s disease). Ann Med Surg. 2016;29(7):79-82. https://doi.org/jnhw.
6.Vijayakumar A, Tiwari R, Kumar-Prabhuswamy V. Thromboangiitis Obliterans (Buerger’s Disease)-Current Practices. Int J Inflam. 2013;2013:156905. https://doi.org/gb64tb.
7.Shionoya S. Diagnostic criteria of Buerger’s disease. Int J Cardiol. 1998;66(Suppl 1):S243-5. https://doi.org/cq5b67.
8.Olin JW. Thromboangiitis obliterans (Buerger’s disease). N Engl J Med. 2000;343(12):864-9. https://doi.org/fcfdnk.
9.Akar AR, İnan MB, Baran Ç. Thromboangiitis Obliterans. Curr Treat Options in Rheum. 2016;2:178-95. https://doi.org/jnhx.
10.Mills JL, Taylor LM Jr, Porter JM. Buerger’s disease in the modern era. Am J Surg. 1987;154(1):123-9. https://doi.org/cw3b2s.
11.Cooper LT, Tse TS, Mikhail MA, McBane RD, Stanson AW, Ballman KV. Long-term survival and amputation risk in thromboangiitis obliterans (Buerger’s disease). J Am Coll Cardiol. 2004;44(12):2410-1. https://doi.org/c3367m.
12.Puéchal X, Fiessinger JN, Kahan A, Menkés CJ. Rheumatic manifestations in patients with thromboangiitis obliterans (Buerger’s disease). J Rheumatol. 1999;26(8):1764-8.
13.Watanabe Y, Miyata T, Shigematsu K, Tanemoto K, Nakaoka Y, Harigai M, et al. Current Trends in Epidemiology and Clinical Features of Thromboangiitis Obliterans in Japan - A Nationwide Survey Using the Medical Support System Database. Circ J. 2020;84(10):1786-96. https://doi.org/jnhz.
14.Sasaki S, Sakuma M, Kunihara T, Yasuda K. Distribution of arterial involvement in thromboangiitis obliterans (Buerger’s disease): results of a study conducted by the Intractable Vasculitis Syndromes Research Group in Japan. Surg Today. 2000;30(7):600-5. https://doi.org/d43p9w.
15.Hurelbrink CB, Barnett Y, Buckland ME, Wilkinson M, Leicester J, Anderson C, et al. Revisiting cerebral thromboangiitis obliterans. J Neurol Sci. 2012;317(1-2):141-5. https://doi.org/f2g926.
16.Drake ME. Winiwarter-Buerger Disease (‘Thromboangiitis Obliterans’) With Cerebral Involvement. JAMA. 1982;248(15):1870-2. https://doi.org/c6wt5s.
17.Alexander JQ, Ciagne MX, Kong JH. Thromboangitis obliterans associated with cannabis use: A case report and literature review. Vasc Dis Manag. 2011;8(7):E124-6.
18.Martin-Blondel G, Koskas F, Cacoub P, Sène D. Is thromboangiitis obliterans presentation influenced by cannabis addiction? Ann Vasc Surg. 2011;25(4):469-473. https://doi.org/btpfwd.
19.Grotenhermen F. Cannabis-associated arteritis. Vasa. 2010;39(1):43-53. https://doi.org/fss998.
20.Desbois AC, Cacoub P. Cannabis-associated arterial disease. Ann Vasc Surg. 2013;27(7):996-1005. https://doi.org/f49k8s.
21.Ducasse E, Chevalier J, Dasnoy D, Speziale F, Fiorani P, Puppinck P. Popliteal artery entrapment associated with cannabis arteritis. Eur J Vasc Endovasc Surg. 2004;27(3):327-32. https://doi.org/bg59c5.
22.Dimmick SJ, Goh AC, Cauzza E, Steinbach LS, Baumgartner I, Stauffer E, et al. Imaging appearances of Buerger‘s disease complications in the upper and lower limbs. Clin Radiol. 2012;67(12):1207-11. https://doi.org/jnh3.
23.Cook TS. Computed Tomography Angiography of the Lower Extremities. Radiol Clin North Am. 2016;54(1):115-30. https://doi.org/gg8p4r.
24.Dargon PT, Landry GJ. Buerger‘s disease. Ann Vasc Surg. 2012;26(6):871-80. https://doi.org/fx97dz.
25.Poznyak A, Grechko AV, Poggio P, Myasoedova VA, Alfieri V, Orekhov AN. The Diabetes Mellitus-Atherosclerosis Connection: The Role of Lipid and Glucose Metabolism and Chronic Inflammation. Int J Mol Sci. 2020;21(5):1835. https://doi.org/gmv76s.
26.Thiruvoipati T, Kielhorn CE, Armstrong EJ. Peripheral artery disease in patients with diabetes: Epidemiology, mechanisms, and outcomes. World J Diabetes. 2015;6(7):961-9. https://doi.org/gh5jrv.