Antibiotic- and heavy-metal resistance in bacteria isolated from deep subsurface in El Callao region, Venezuela
Resistencia a antibioticos y metales pesados en bacterias aisladas de subsuelo en la región El Callao, Venezuela
DOI:
https://doi.org/10.15446/rev.colomb.biote.v16n2.41004Palabras clave:
subsurface bacteria, resistance, mercury, antibiotic, plasmid (en)bacterias del subsuelo, resistencia, mercurio, antibióticos, plásmidos (es)
https://doi.org/10.15446/rev.colomb.biote.v16n2.41004
ARTÍCULO DE INVESTIGACIÓN
Antibiotic- and heavy-metal resistance in bacteria isolated from deep subsurface in El Callao region, Venezuela
Resistencia a antibioticos y metales pesados en bacterias aisladas de subsuelo en la región El Callao, Venezuela
Maura Lina Rojas Pirela, Wilmar Alirio Botello Suárez, María Mercedes Ball Vargas
Laboratorio de Microbiología Molecular y Biotecnología, Facultad de Ciencias, Universidad de Los Andes. Mérida 5101, Venezuela.
maura.r@ula.ve, mball@ula.ve, wbotello@unisangil.co. Corresponding author: Laboratorio de Microbiología Molecular y Biotecnología, Facultad de Ciencias, Universidad de Los Andes. Mérida
5101, Venezuela. mball@ula.ve.
Recibido: febrero 10 de 2014 Aprobado: octubre 20 de 2014
Resumen
Se investigó el efecto de la contaminación con mercurio (Hg) en las comunidades bacterianas del subsuelo profundo en la región de El Callao (Estado Bolívar, Venezuela). Se estudiaron comunidades bacterianas de dos niveles de profundidad (-288 m y -388 m) en una mina de oro con el propósito de describir las características más relevantes de las bacterias indígenas cultivables que colonizaban esta mina. Se evaluaron los patrones de resistencia a antibióticos y metales pesados, presencia del gen merA y plásmidos en aislados resistentes. Se encontró una elevada frecuencia de bacterias indígenas resistentes al Hg y otros metales pesados. De 76 aislados Hg-resistentes probados 73.7 % fueron adicionalmente resistentes a ampicilina; 86.8 % a cloranfenicol; 67.1 % a tetraciclina; 56.6 % a estreptomicina y 51.3 % a kanamicina. Además, se encontró que 40.74 % (-328 m) y 26.53 % (-388 m) de las bacterias Hg-resistentes fueron simultáneamente resistentes tanto a cuatro como a cinco de estos antibióticos. Se detectó la presencia de plásmidos de alto y bajo peso molecular y, a pesar de que los aislados mostraban resistencia a compuestos mercuriales, la presencia del gen merA fue detectada solo en 71.05 % de los cepas. Estos resultados sugieren que la exposición a Hg podría ser una presión selectiva en la proliferación de bacterias resistentes a antibióticos y promover el mantenimiento y propagación de estos genes de resistencia. Sin embargo, la existencia de tales resistencias a estas profundidades podría también apoyar la idea de que la resistencia a antibióticos en estas bacterias es natural y tiene un origen más antiguo que su exposición al Hg.
Palabras clave: bacterias del subsuelo, resistencia, mercurio, antibióticos, plásmidos.
Abstract
The effect of contamination with mercury (Hg) in the deep subsurface bacterial communities in the region of El Callao (Bolívar State, Venezuela) was investigated. Bacterial communities from two deep levels (-288 m and -388 m) in a gold mine were studied with the aim of describe the most relevant features of their colonizing indigenous culturable bacteria. Antibiotic and heavy metals resistance patterns, presence of the merA gene and plasmids in resistant isolates were evaluated. A high frequency of resistant indigenous bacteria to Hg and other heavy metals was found. From 76 Hg-resistant isolates tested 73.7 % were, in addition, resistant to ampicillin, 86.8% to chloramphenicol, 67.1 % for tetracycline, 56.6 % streptomycin, and 51.3 % kanamycin. Furthermore, it was found that 40.74 % (-328 mm) and 26.53 % (-388 m) of Hg-resistant bacteria were simultaneously resistant to both four and five of these antibiotics. The presence of low and high molecular weight plasmids was detected and, despite that isolated showed resistance to mercurial compounds, the presence of the gene merA was detected only in 71.05 % of strains. These results suggest that exposure to Hg could be a selective pressure on the proliferation of antibiotic-resistant bacteria and promote the preservation and propagation of these resistance genes. However, the existence of such resistances to these depths could also support the idea that antibiotic resistance in these bacteria is natural and has a more ancient origin than their exposure to Hg.
Keywords: subsurface bacteria, resistance, mercury, antibiotic, plasmid.
Introduction
In the past decades it was thought that life of prokaryotic- type organisms was only possible in the shallow subsurface, these being not capable of living to great depths of the surface. However, now it is known that the deep subsurface represents one of the most promising reservoirs of living organisms, mainly prokaryotes, which may exhibit unexpected metabolic abilities (Moser et al., 2005; Parkes et al., 2010) Nowadays, the study of subsurface microorganisms has bright future, and is considered an emerging frontier in the fields of microbial biodiversity and life at extreme environments (Fredrickson and Balkwill, 2006, Wang et al., 2013).
On the other hand, the potential application of subsoil bacteria in bioremediation of contaminated environments is receiving increasing attention. In this regard, gold mines offer an excellent opportunity to study subterranean microbial communities because they provide an easy access (sometimes >8000 ft.) to greater depths of terrestrial subsurface, allowing thus cheaperand aseptic-horizontal drilling of deeper strata. Furthermore, by this route it is possible to have multiple access points at different depths to follow the evolutionary history of microbial populations, some of which may have stayed isolated from terrestrial surface by thousands of years. Moreover, the enormous amount of geologic and hydrologic information accumulated by mining companies can be easily integrated with biological results. To date, gold mines from South Africa (Takai et al., 2001, Chivian et al., 2008), Japan (Hirayama et al., 2005), USA (Rastogi et al., 2010, Rastogi et al., 2013) and Australia (Adams et al., 2013) have been screened for subsurface life.
Gold reservoirs in the El Callao area (Bolívar State, Venezuela) have been exploited since 1850, with over 200 metric tons produced (Hildebrand, 2005). Colombia Mine, a gold reservoir located near to El Callao town, is a >500m deep, active gold mine. A vast area surrounding Colombia Mine is highly contaminated with mercury (Hg). Indeed, this contaminant is discharged by artisanal miners in its metallic form into hundreds of artificial ponds ("tailing-ponds") spread over several square kilometers. Mercury emission in El Callao area has been estimated over 12tons/y (Veiga et al., 2005). As a consequence, bacteria thriving in natural water-bodies and/or tailing-ponds exhibit high frequencies of Hg-resistance, as it have been shown in our previous work (Ball et al., 2007, Gómez et al., 2013).
Material and methods
Site and sampling methods
Colombia Mine is located near to El Callao town (Bolivar State, Venezuela; latitude: 7 21' 00''; longitude: -61 49' 00''). Most water samples were collected from existing exploratory boreholes and roof fractures located at -288m (level IV) and -388m (level VI) below the mine entrance (22m above the sea level and -78m below the sea level, respectively). The flowing boreholes, with different configurations and ages ranging from 10 to 50 years at the sampling time, exhibited high flow rates (i.e. tens of liters.h-1). Water samples were collected directly from the flowing boreholes into sterile glass tubes.
Chemical analysis
Chemical analysis of major cations and trace metals,i.e. Al, As, Ba, Ca, Cd, Co, Cu, Cr, Fe, Mn, Se, Ni, Pb and Zn, in the water samples was conducted at the Laboratorio Regional de Servicios Analíticos (LARSA, Universidad de Los Andes, Mérida, Venezuela). All water samples were prefiltered with a 0.45 μm filter and analyzed by Inductively Coupled Plasma Atomic Emission Spectroscopy on a Varian Liberty AX instrument. Mercury was detected on aliquots of each sample, previously acidified by adding a few drops of pure HCl, by Atomic Absorption Spectroscopy (AAS), using a recently developed method (Duran et al., 2005).
Isolation of pure cultures
This study focused specifically on aerobic chemoheterotrophic bacteria. Therefore, bacterial strains were isolated from water samples at each sampling site for further study by serially diluting and spreading on 1/3-strength Luria-Bertani agar (LB(1:3); tryptone 3.3g/L, yeast extract 1.6g/L, NaCl 3.3g/L, agar 15.0g/L). The plates were incubated at 30°C and colony forming units (CFU) were enumerated after 48h of growth. Morphologically distinct colony types were selected for further studies. The isolates were subsequently streaked and re-streaked on fresh plates in order to obtain axenic cultures. Then, the isolated strains were grown in liquid LB (1:3) and frozen in 20% glycerol at -80 °C.
Determination of heavy metal and antibiotic-resistance
Mercury-resistance for each isolate was determined by streaking the cells onto 1/3-strength LB agar plates amended with a) mercuric chloride (HgCl2) (from 10 to 80mg/L) and b) methyl mercury (MeHg) (from 3 to 5mg/L). Stock solutions of HgCl2 and Me-Hg were sterilized by ultra-filtration, kept cold in the dark, and added to the medium after autoclaving and cooling. The minimum HgCl2 concentration which allowed distinguishing resistant from sensitive isolates was determined by performing preliminary assays using reference strains. These consisted of strains carrying characterized Hg-resistance determinants (Pseudomonas stutzeri OX and Bacillus cereus 5) (Izaki, 1981; Reniero, 1998) and either their sensitive mutants or phylogenetically related strains (Pseudomonas stutzeri OX 1 and Bacillus cereus ATCC 14579) (Benyehuda et al., 2003).
Therefore, strains resistant to at least 10mg/L of HgCl2 were considered as HgR and selected for further studies. On the other hand, heterotrophic isolates were grown in the presence of either 2 or 5mM of the following metals: Cu2+ (used as CuSO4), Pb2+ (used as Pb(NO2)3), Ni2+ (used as NiSO4) and Zn2+ (used as ZnCl2). The plates were incubated at 30°C during 48h. Strains DH5α and JM101 of E. coli K-12 were used as negative controls (= sensitive strains).
All subsurface isolates were screened also for resistance to a wide range of classes of antibiotics: tetracycline (Tet) (30μg/ml), chloramphenicol (Cam) (30μg/ml), kanamycin (Kan) (30μg/ml), streptomycin (Strp) (30 μg/ml), and ampicillin (Amp) (40μg/ml). Selective media were prepared by incorporating the antimicrobial agent into the 1/3-strength LB agar. Concentrations used were above the highest inhibitory concentration listed in the Performance Standards for Antimicrobial Susceptibility Testing (2005), except for kanamycin. The strains were reactivated from frozen stocks, plated on selective media (triplicate) and checked for growth after 24, 48, and 72h. Results were recorded as sensitive or resistant; intermediate growth was not considered.
Minimum Inhibitory Concentration Assays
In order to establish the Minimal Inhibitory Concentration (MIC) for Hg, the HgR strains were assayed with an Hg2+ concentration series in 1/3-strength LB broth using a 96-well microtiter plate. The well contained 200μl of broth and Hg at concentrations ranging from 10, 20, 40 and 80μM (2.72 to 21.8 mg/L HgCl2). Each well was inoculated with a 10μl aliquot of the correspondent HgR strain in the late exponential phase, at an initial optical density at 600nm (OD600) of 0.03, and the plates were incubated at 30°C for 24h. Each experiment was performed in triplicate. The lowest concentration of the metallic compound that caused no visible growth was considered as the MIC. The isolates were considered resistant if the MIC value exceeded that of the E. coli K-12 strain, which was used as the control (Akinbowale et al., 2007).
Detection of merA orthologs
The merA gene, which belongs to the mer operon and encodes a cytoplasmic mercuric reductase, was amplified using degenerate primers A1s-n.F (5'TCCGCAAGTNGCVACBGTNGG3') and A5-n.R (5'ACCATCGTCAGRTARGGRAAVA3') and cycling conditions previously reported (Ni Chadhain et al., 2006). PCR reactions were prepared in 10 μl volumes containing 1 × PCR buffer, 0.8 mM each forward and reverse primers, 1.5 mM MgCl2, 0.2 mM dNTPs, 0.025 U/ml Taq Polymerase (Promega). Whole cell suspensions in sterile distilled water were used as source of genomic DNA (1 μl per reaction). Amplifications were performed as follows: an initial denaturalization step of 94°C for 5 min, followed by 45 cycles of 94 °C for 10 s, 54 °C for 60 s, and 72 °C for 60 s, and by a final extension of 72 °C for 7 min.
In preliminary experiments, fragments of the expected size (~285 bp) were purified from 1% agarose gels (NucleoSpin Extract, Macherey-Nagel) and sequenced (Macrogen Inc., Seoul, South Korea). The results obtained validated the PCR protocol described above. Therefore, PCR products of the expected size were considered to belong to putative merA orthologs.
Plasmid isolation
Some selected HgR isolates were screened for the presence of plasmids as previously described by Ball et al. (2007).
Results
Chemical analysis
This indicated that, even though several dozens processing centers with their respective, heavily-contaminated tailing ponds are located in the area surrounding Colombia Mine, neither Hg nor any other heavy metal (except manganese) was present at detectable concentrations on groundwater collected at both subsurface levels (table 1). Calcium was found in both levels.
Resistance to mercury and others metals
The total number of heterotrophic, cultivable bacteria in each one of the two levels monitored varied between 1.9-103CFU/ml (level IV) and 2.5-103CFU/ml (level VI). Resistance to HgCl2 (10mg/L), tested among pure isolates, varied between 83 to 90%, depending on the level being monitored (Table 1). Under these conditions, control strains B. cereus ATCC 14579 and P. stutzeri OX1 were unable to grow at concentration above 5mg/L of HgCl2, while strains B. cereus 5 and P.stutzeri OX grew up to 80mg/L of HgCl2. On the other hand, resistance to MeHg (3mg/L) varied between 13.33% and 18.64% (table 1). Strikingly, nearly 6% of isolates were resistant up to 5mg/L of MeHg in the deeper stratus. A total of 27 strains (90%) from level IV and 49 strains (83.05%) from level VI, resistant to at least 10mg/L of HgCl2, were isolated and purified from the primary cultures to be further tested.
It is well known that resistance values obtained by growing strains in Hg-supplemented agar media may cause an overestimation of this parameter. Thereby, the values for MIC of Hg2+ for each strain were estimated in liquid medium. The 76 HgR strains were incubated in 1/3-strength LB broth containing HgCl2 concentrations ranging from 2.72mg/L (10μM Hg2+) to 21.8mg/L (80μM Hg2+) of HgCl2. 59.25% of the isolates tested from water samples from level IV were resistant to Hg2+ 10μM, while in level VI this value attained 100% (table 2).
When the combined results of both levels are considered it is clear that the great majority of the strains tested (85.52 %) were able to grow in the presence of Hg2+ 10μM; on the other hand, only 3 out of 76 strains tested (3.94%) were resistant to Hg2+ 80μM (table 3). It was also demonstrated that these strains showed resistance to other heavy metals, including Cu2+, Pb2+, Ni2+ and Zn2+ (table 4). Strikingly, a great majority of the isolates (between 76.31 and 100%) were able to grow in the presence of 2 mM of each one of the metals tested, a concentration which inhibited growth of the control strain of E. coli (table 4).
As expected, the frequencies of resistant isolates decreased when increasing the concentration of the toxic element. A general tendency can be also appreciated, indicating that most isolates were more susceptible, under similar concentrations, to Pb2+ than Cu2+ and Ni2+, whereas Zn2+ turned out the least toxic among the tested metals.
Antibiotic resistance
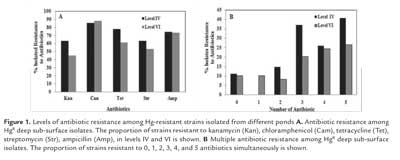
All of the 76 HgR subsurface isolates were screened for resistance to 5 antibiotics through plating on selective media. The highest frequency of resistance was to chloramphenicol (>85 % of the strains in both levels, figure. 1A) and Amp (> 70 % of the strains in both levels). The antibiotic with the highest efficacy was kanamycin, with 62.96% and 44.89 % and streptomycin with 62.96 % and 53.06% of strains showing resistance in levels IV and VI, respectively. The two levels (IV and VI) studied showed a high frequency of isolates resistant to more than one antibiotic (figure 1B). In level VI, 26.53% of the strains were resistant to, at least, five antibiotics, while in level IV this value attained 40.74%.
Subsurface strains from each one of the two levels exhibited distinct antibiotic resistance profiles. Nine distinct profiles were detected among 27 HgR strains in level IV while in level VI; thirteen distinct profiles were detected among 49 HgR isolates (figure 2).
Combined results of the two strains groups shows that 76 HgR isolates possess 15 distinct resistance profiles, 12 of which were found in more than one strain (figure 2). Interestingly, 24 isolates (31.58%) showed the same profile of multi-resistance (KCTSA).
Amplification of merA gene
In order to detect the presence of the merA gene encoding the enzyme responsible for Hg2+ reduction, PCR amplification experiments using degenerate primers were performed. A band corresponding to an internal fragment of putative merA orthologs was amplified from the genome for 54 of the 76 isolates (71.05%, figure 3). In level IV this value reach 66.66% while in level VI 73.47% of the isolates exhibited putative merA orthologs.
Plasmid isolation
With of the purpose to establishing a possible transfer of heavy metal- and antibiotic-resistance genes by conjugation, 20 HgR strains were screened for the presence of large plasmids. Results obtained confirm that 11 isolates (55 %) contained at least one plasmid; 62.9% of them showed plasmid bands with apparent sizes >30kbp (figure 3). In addition, previous studies in vitro conjugation clearly show the potential for conjugal gene transfer in this indigenous bacteria and this transfer can occur between phylogenetically distant bacteria (unpublished data).
Discussion
Little is known about the impact for Hg contamination of the microbial communities colonizing the deep subsurface in the El Callao region. Results in this study shown that resistance to Hg and others heavy metals is very frequent among chemoheterotrophic bacteria that colonize deep subsurface in this region. This fact may be considered as indicative of a possible exposure of the subsurface microbial communities to Hg, even though chemical analysis of the water do not detected the presence of these heavy metals including mercury. Probably this metal exposure occur through a process of cross-contamination, via filtration of water from rivers, streams and tailings ponds highly contaminated during local rainy season (Ball et al., 2007, Gómez et al., 2013) (from September to February), which is accompanied by major flooding and overflowing of bodies of surface waters polluted (García-Sánchez et al., 2008). Moreover, the absence of detectable concentrations of mercury can be due to the mineralogical composition of the mine (rich in carbonates, oxides and silicate) (Paredes et al., 2007) and pH values (neutral to basic) that may alter the solubility of the metals (Uchimiya et al., 2010; Woodruff et al., 2010) and prevent detection by the used technique. It can be argued, however, constant flow unpolluted water would act as natural attenuation events. This could be reflected in the level of tolerance to mercury of the bacterial communities of groundwater. The mercury tolerance level of bacterial communities from polluted surface water is greater than in subsurface communities. This may be due to constant exposure of these bacteria of the surface to high mercury concentrations (Ball et al., 2007, Gómez et al., 2013), while the sporadic exposure to mercury of subsurface bacteria may lead to lower tolerance to this metal. This indicates that subsurface communities pre exposed to mercury could maintain their mercury tolerance even when the current exposure is low. Also, this could indicate a high capacity for acclimation the subsurface communities to mercury.
Additionally, it was found that antibiotic resistance is widespread among subsurface bacteria, a high proportion of isolates exhibited multiple antibiotic resistance (MAR; resistance to two or more antibiotics) in accordance with a previous report (Brown et al., 2009; Diptendu and Gouta, 2013; Stepanauskas et al., 2006). Besides Hg-resistance, the subsurface isolated did also showed resistance to other heavy-metals (Cu, Pb, Ni, Zn) and, several antibiotics. These results are very similar to the results obtained in previous studies in surface waters in the region of El Callao (Ball et al., 2007, Gómez et al., 2013). These results could support the idea that microbial exposure to a toxic substance could result in indirect selection for bacteria with resistance to multiple toxic substances, such as antibiotics (Baker-Austin et al., 2006, Seiler and Berendonk, 2012; Telmer et al., 2009). Thus, in other words, exposure to toxic metals may select for bacterial strains resistant to antibiotics and vice versa. Indeed, in a work, Stepanauskas et al. (2006) demonstrated that exposure of freshwater environments to either metals or antibiotics selects for multiresistant microorganisms. It is known that genes conferring resistance to heavy metal, anti biotic and other toxic compounds are often genetically linked and located on mobile elements (i.e., conjugative plasmids, transposons, and integrons), some of which are easily exchanged among phylogenetically distant bacteria (Summers, 2002; Davies and Davies, 2010; van Hoek et al., 2011). Therefore, microbial exposure to mercury (even for occasional contamination) can co-select antibiotic resistance and/or induce the maintenance of resistance genes in the absence of the antibiotic selective pressure. These co-selection mechanisms include coresistance, cross-resistance and indirect but shared regulatory responses to metal and antibiotic exposure, such as biofilm induction, representing potential co-selection mechanisms used by prokaryotes (Baker-Austin et al., 2006, Seiler and Berendonk, 2012). Resistance to multiple antibiotics in bacterial communities studied in this work confirms that this ability is common and widespread among subsurface bacteria, with a high proportion of isolates exhibiting MAR. The presence of antibiotic-resistance genes in bacterial communities from natural environments, without apparent selection pressure, has been observed. Thus, Bhullar et al. (2012) discovered an antibiotic-resistant bacteria community in an isolated cave 400 meters below the Earth's surface. They report that these bacteria were highly resistant to antibiotics and, some strains were resistant up to 14 different antibiotics. Similar results were obtained with bacteria isolated on natural environments in differents glacier environments (Segawa et al., 2013) as Siberian permafrost (Mindlin etal., 2008) and the Arctic (Sudha et al., 2013).
Previous results can support the notion that organisms on natural environments are a reservoir of resistance genes and that antibiotic resistance is a natural and dominant component in ancient microbial pangenomes with a long evolutionary past (Bhullar et al. 2012). The antibiotic resistance and its occurrence in these subsurface bacterial communities could be due to others reasons. Antibiotic-resistance genes could play different roles in natural environments just like the one studied. i.e., these could have a protective role either in the producer organisms or in some of their coexisting organisms (Laskaris et al., 2010) while some of them could play an important role in the biosynthesis of antibiotics (Allen et al., 2010) and other could be involved in metabolic and signaling of bacterial processes occurring in natural ecosystems (Laskaris et al., 2010) and some others could cooperate in the degradation and use of antibiotics as food (Davies and Davies, 2010). Owing the environmental conditions of the mine (i.e., low nutrient availability) it is likely that competition for resources can play an important role in the persistence of these bacteria. This competition can be developed through various adaptations such as production of antibiotic to compete with their nutritional antagonists, acquisition and development of defense mechanisms to enable them growth in the presence of noxious compounds, changes in cell and growth physiology (Bhullar et al., 2012). Hence, it might be thought that antibiotic resistance in these isolates of groundwater is anterior to the emergence of antibiotic. Information on current mechanisms for horizontal gene transfer (HGT) in depth subsurface environments is scarce. Considering the characteristics of this environment (e.g. low density of microbes) is difficult to imagine how it can occur there. The high frequency of plasmids in HgR strains could indicate that elevated levels of metal- and antibiotic-resistance are related to carrying of resistance genes by these plasmids. In studies have observed the plasmid presence and the occurrence of HGT in communities from subsurface environment (although this may occur at a low frequency) (Coombs 2009; Hemme et al. 2010). HGT in these communities would play an important role in their gene evolution, which have permitted them survival in hostile environments.
The presence of putative merA orthologs in a high proportion (71.5%) of the bacterial isolates analyzed in the present work seem to indicate that bacterial detoxification of mercury proceed via the classical pathway involving ionic-mercury reduction by a mercuric reductase. For the remaining 28.5% of the strains, the resistance seems to be not due to enzymatic volatilization of mercury, another possible mechanism of resistance that could be involved. Some bacteria possess proteins in their plasmatic membranes which alter the mercury permeability inhibit mercury uptake (or binding) by the cells (Pan-Hou et al., 1981). Plasmid-encoded efflux systems have also been suggested as alternative systems to mercury resistance (Reyes et al., 1999).
Conclusions
Bacterial communities found in groundwater from the subsurface mine "Colombia", located on the auriferous reservoirs of El Callao, Venezuela, presented a significant frequency of mercury-resistant strains, to both inorganic (HgCl) and organic (MeHg) markers, at the two levels of depth explored (-288 m and -388m below the mine entrance and -22m and -78m below the sea level, respectively).
Likewise, it can be ascertained that strains carrying HgR genes also exhibited resistance to antibiotics and other heavy metals, which may be an indication that heavy metals resistance could assist the co selection of antibiotics-resistance genes. However, it is important to consider that observation of antibiotic-resistant microorganisms in natural environments free of pollutants has become ever more common, suggesting a origin natural and more ancient of these genes. The presence of low and high molecular weight plasmids of conjugative type in these bacteria, as envisioned in the preliminary studies carried out, could suggest the occurrence of HGT processes in these communities, allowing a more widespread dissemination of these genes.
The presence of putative orthologs merA in a high proportion of bacterial isolates analyzed indicate that detoxification of bacterial mercury proceed via the classical pathway involving ionic mercury-reduction by a mercuric reductase. However, in other isolates the resistance appears to be due to another mechanism of resistance that may be involved.
Acknowledgements
We are indebted to Dr. Tamar Barkay (Department of Biochemistry and Microbiology at Rutgers University, New Brunswick, NJ) for kindly providing the HgR and HgS reference strains used in this study. We are also grateful to Dr. Pablo Carrero (Facultad de Ciencias, Universidad de Los Andes. Venezuela) for performing chemical analysis of water samples (Hg2+ and Me-Hg determination) and Dr. Wilfredo Quiñones (Facultad de Ciencias, Universidad de Los Andes, Venezuela) and Dr Cristóbal Lárez Velásquez for his critical revision of this work. We thank CVG-Minerven C.A. for logistic support in El Callao. This work was financed partially by CDCHTULA (project number C-1428-06- 03-B).
References
1. Adams, A.; Raman, A.; Hodgkins, D.; Nicol, H. 2013. Accumulation of heavy metals by naturally colonizing Typha domingensis (Poales Typhaceae) in waste-rock dump leachate storage ponds in a gold-copper mine in the central tablelands of New South Wales, Australia. International Journal of Mining, Reclamationand Environment. 27: (4). 294- 307.
2. Allen, H.; Donato, J.; Wang, H.H.; Cloud-Hansen, K.A.; Davies, J.; Handelsman, J. 2010. Call of the wild: antibiotic resistance genes in natural environments. Nature Reviews Microbiology. 8 (4):251-59.
3. Akinbowale, O.; Peng, H.; Grant, P.; Barton, M. 2007. Antibiotic and heavy metal resistance in motile Aeromonads and Pseudomonas spp. from rainbow trout (Oncorhynchusmykiss) farms in Australia. International Journal of Antimicrobial Agents. 30(2):177-82.
4. Ball, M.; Carrero, P.; Castro, D.; Yarzábal, A. 2007. Mercury resistance in bacterial strains isolated from tailing ponds in a gold mining area near El Callao (Bolívar State, Venezuela). CurrentMicrobiology. 54(2):149–154.
5. Baker-Austin, C.; Wright, M.; Stepanauskas, R.; McArthur J. 2006. Co-selection of antibiotics and metals resistance. Trends Microbiology. 14(4): 176–182.
6. Benyehuda, G.; Coombs, J.; Ward, P.; Balkwill, D.; Barkay, T. 2003. Metal resistance among aerobic chemoorganotrophic bacterial isolates from the deep terrestrial subsurface. Canadian Journal of Microbiology. 49(2):151-156.
7. Bhullar, K.; Waglechner, N.; Pawlowski, P.; Koteva, K.; Banks, E.; Johnston, M.; et al., 2012. Antibiotic resistance is prevalent in an isolated cave microbiome. PLoSONE. 7(4):e34953.
8. Brown, M.; Balkwill, D. 2009. Antibiotic resistance in bacteria isolated from the deep terrestrial subsurface. Microbial Ecology. 57(3):484-493.
9. Coombs, J. 2009. Potential for horizontal gene transfer in subsurface microbial communities. Totowa, New Jersey. In Gogarten M, Gogarten J, Olendzenski L (Eds).Humana Press. Horizontal Gene Transfer: Genomes in Flux. p 413-433.
10. Clinical and Laboratory Standards Institute. 2005. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing. Wayne, PA Fifteenth informational supplement, CLSI document M100- S15.
11. Chivian, D.; Brodie, E.; Alm, E.; Culley, D.; Dehal, P.; DeSantis, T.; & Onstott, T. C. 2008. Environmental genomics reveals a single species ecosystem deep within Earth. Science. 322(5899):275- 278.
12. Davies, J.; Davies, D. 2010. Origins and Evolution of Antibiotic Resistance.Microbiology and Molecular Biology Reviews. 74 (3): 417-433.
13. Diptendu, S.; Goutam, P. 2013. Molecular Characterization of Metal and Antibiotic Resistance Activities in a Bacterial Population Isolated from Waste Water Sample. Intenational Journal of Biotechnology and Bioengineering Research. 4(1):21-30.
14. Duran, F.; Carrero, P.; Rondón, C. 2005. A new method to determine mercury species in water samples through cold vapor generation and atomic absorption spectroscopy detection. Proceedings of the VII Venezuelan Chemistry Congress, Faculty of Sciences. Universidad de Los Andes (Mérida, Venezuela). p 35–37.
15. Fredrickson, J.; Balkwill, D. 2006. Geomicrobial processes and biodiversity in the deep terrestrial subsurface. Geomicrobiology Journal. 23(6): 345-356.
16. García-Sánchez, A.; Contreras, F.; Adams, M.; Santos, F. 2008. Mercury contamination or surface water and fish in a gold mining región (Cuyuni River Basin,Venezuela). International Journal Environment Pollution. 33(2/3) 260-274.
17. Gómez, W.; Ball, M.; Botello, W.; Yarzábal, A. 2013. Horizontal transfer of heavy metal and antibiotic-resistance markers between indigenous bacteria, colonizing mercury contaminated tailing ponds in southern Venezuela, and human pathogens. Revista de la Sociedad Venezolana de Microbiología. 33: 110- 115.
18. Hemme, C.; Deng, Y.; Gentry, T.; Fields, M.; Wu, L.; Barua, S.; & Zhou, J.. 2010. Metagenomic insights into evolution of a heavy metal - contamined groundwater microbial community. Multidisciplinary Journal of Microbial Ecology. 4: 660-672.
19. Hildebrand, R. 2005. Autochthonous and allochthonous strata of the El Callao greenstone belt: Implications for the nature of the Paleoproterozoic Trans- Amazonian orogeny and the origin of gold-bearing shear zones in the El Callao mining district, Guayana shield, Venezuela. Precambrian Research. 143(1): 75–86.
20. Hirayama, H.; Takai, K.; Inagaki, F.; Yamato, Y.; Suzuki, M.; Nealson, K.; Horikoshi, K. 2005. Bacterial community shift along a subsurface geothermal water stream in a Japanese gold mine. Extremophiles. 9 (2):169-184.
21. Izaki, K. 1981. Enzymatic reduction of mercurius and mercuric ions in Bacillus cereus. Canadian Journal of Microbiology. 27:192- 197.
22. Laskaris, P.; Tolbas, S.; Calvo-Bado, L.; Wellington E. 2010. Coevolution of antibiotic production and counter-resistance in soil bacteria. Enviromental Microbiology. 12(3):783-796.
23. Moser, D.; Gihring, T.; Brockman, F.; Fredrickson, J.; Balkwill, D.; Dollhopf, M.; & Onstott, T. C. 2005. Desulfotomaculum spp. and Methanobacterium spp. Dominate a 4 to 5 km Deep Fault. Applied and Environmental Microbiology. 71(12):8773-8783.
24. Mindlin, S.Z.; Soina, V.S.; Petrova, M.A.; Gorlenko, Zh.M. 2008. Isolation of antibiotic resistance bacterial strains from eastern Siberia permafrost sediments. Russian Journal of Genetics. 44(1): 36-44.
25. Ni Chadhain, S.; Schaefer, J.; Crane, S.; Zylstra, G.; Barkay, T. 2006. Analysis of mercuric reductase (merA) gene diversity in ananaerobic mercury-contaminated sediment enrichment. Environmental Microbiology. 8(10): 1746-1752.
26. Pan-Hou, H.; Nishimoto, M.; Imura, N.1981 Possible role of membrane protein in mercury resistance of Enterobacter aerogenes. Archives of Microbiology. 130(2): 93-95.
27. Paredes, S.; Hidalgo-Prada, B.; Ávila, R.; Albornoz, A. 2007. Análisis de mineral aurífero de la mina Colombia, El Callao, Venezuela, mediante MEB/EDX Y DRX. Acta Microscópica. 16: s301-302.
28. Parkes, R.; Linnane, C.; Webster, G.; Sass, H.; Weightman, A.; Hornibrook, R.; & Horsfield, B. 2011. Prokaryotes stimulate mineral H2 formation for the deep biosphere and subsequent thermogenic activity. Geology. 39(3): 219-222.
29. Rastogi, G.; Osman, S.; Kukkadapu, R.; Engelhard, M.; Vaishampayan, P.; Andersen, G.; Sani, R. 2010. Microbial and minerological characterizations of soils collected from the deep biosphere of the former Homestake gold mine, South Dakota. Microbial Ecology. 60 (3): 539-550.
30. Rastogi, G.; Gurram, R.; Bhalla, A.; Gonzalez, R.; Bischoff, K.; Hughes, S.; Kumar, S.; Sani, R. 2013. Presence of glucose, Xylose, and glycerol fermenting bacteria in the deep biosphere of the Homestake gold mine, South Dakota. Frontiers in Microbiology. 9: 18.
31. Reniero, D.; Mozzon, E.; Galli, E.; Barbieri, P. 1998. Two aberrant mercury resistance transposons in the Pseudomonas stutzeri plasmid pPB. Gene. 208(1): 37-42.
32. Reyes, N.; Marc, E.; Frischer, M.; Sobecky, P. 1999. Characterization of mercury resistance mechanisms in marine sediment microbial communities. FEMS Microbiology Ecology. 30 (3): 273-84.
33. Segawa, T.; Takeuchi, N.; Rivera, A.; Yamada, A.; Yoshimura, Y.; Barcaza, G., & Ushida, K. 2013. Distribution of antibiotic resistance genes in glacier environments. Environmental Ecology of Pathogens and Resistances. 5(1): 127-134.
34. Seiler, C.; Berendonk, T. 2012. Heavy metal driven co-selection of antibiotic resistance in soil and water bodies impacted by agriculture and aquaculture. Frontiers in Microbiology. 3:399.
35. Sudha, A.; Augustine, N.; Thomas, S. 2013. Emergence of multi-drug resistant bacteria in the Arctic, 790N. Journal of Cell and Life Sciences. 1(1): 1-5.
36. Summers, A. 2002. Generally Overlooked Fundamentals of Bacterial Genetics and Ecology. Clinical Infectious Diseases. 34: s84-s92.
37. Stepanauskas, R.; Glenn, T.; Jagoe, C.; Tuckfield, R.; Lindell, A.; King, C.; & McArthur, J. V.. 2006. Coselection for microbial resistance to metals and antibiotics in freshwater microcosms. Environmental Microbiology. 8(9): 1510–1514.
38. Takai, K.; Moser, D.; Deflaum, M.; Onstott, T.; Fredrickson, J. 2001. Archaeal Diversity in Waters from Deep South African Gold Mines. Applied and Environmental Microbiology. 67(12): 5750- 5760.
39. Telmer, K.; Veiga, MM. 2009. World emissions of mercury from artisanal and small scale gold mining. Pirrone N, Mason R (Eds). Springer Science + Business Media, p 131-172.
40. Uchimiya, M.; Lima, L.; Thomas, K.; Chang, S.; Wartelle, L.; Rodgers, J. 2010. Immobilization of heavy metal ions (CuII,CdII, NiII, and PbII) by broiler litter-derived biochars in water and soil. Journal of Agricultural Food Chemistry. 58(9): 5538-5344.
41. Veiga, M.; Bermudez, D.; Pacheco-Ferreira, H.; Martins, P.; Gunson, A.; Berrios, G.; Vos, L.; Hiudobro, P.; Roeser, M. 2005. Mercury from artisanal gold mining in Block B, El Callao, Bolivar State, Venezuela. In Pirrone N and Mahaffey K (Eds). Norwell, MA, USA. Dynamics of Mercury Pollution on Regional and Global Scales: Atmospheric Process and Human Exposure Around the World. Springer Publisher, p. 421-450.
42. Wang, F.; Lu, S.; Orcutt, B.; Xie, W.; Chen, P.; Xiao, X.; Edwards, J. 2013. Discovering the roles of subsurface microorganisms: Progress and future of deep biosphere investigation. ChineseScience Bulletin. 58 (4-5): 456-467.
43. Woodruff, L.; Cannon, W. 2010. Immediate and long-term fire effects on total mercury in forests soils of Northeastern Minnesota. Environmental Science & Technology. 44(14): 5371-5376.
Referencias
Adams, A.; Raman, A.; Hodgkins, D.; Nicol, H. 2013. Accumulation of heavy metals by naturally colonizing Typha domingensis (Poales Typhaceae) in waste-rock dump leachate storage ponds in a gold-copper mine in the central tablelands of New South Wales, Australia. International Journal of Mining, Reclamationand Environment. 27: (4). 294- 307.
Allen, H.; Donato, J.; Wang, H.H.; Cloud-Hansen, K.A.; Davies, J.; Handelsman, J. 2010. Call of the wild: antibiotic resistance genes in natural environments. Nature Reviews Microbiology. 8 (4):251-59.
Akinbowale, O.; Peng, H.; Grant, P.; Barton, M. 2007. Antibiotic and heavy metal resistance in motile Aeromonads and Pseudomonas spp. from rainbow trout (Oncorhynchusmykiss) farms in Australia. International Journal of Antimicrobial Agents. 30(2):177-82.
Ball, M.; Carrero, P.; Castro, D.; Yarzábal, A. 2007. Mercury resistance in bacterial strains isolated from tailing ponds in a gold mining area near El Callao (Bolívar State, Venezuela). CurrentMicrobiology. 54(2):149–154.
Baker-Austin, C.; Wright, M.; Stepanauskas, R.; McArthur J. 2006. Co-selection of antibiotics and metals resistance. Trends Microbiology. 14(4): 176–182.
Benyehuda, G.; Coombs, J.; Ward, P.; Balkwill, D.; Barkay, T. 2003. Metal resistance among aerobic chemoorganotrophic bacterial isolates from the deep terrestrial subsurface. Canadian Journal of Microbiology. 49(2):151-156.
Bhullar, K.; Waglechner, N.; Pawlowski, P.; Koteva, K.; Banks, E.; Johnston, M.; et al., 2012. Antibiotic resistance is prevalent in an isolated cave microbiome. PLoSONE. 7(4):e34953.
Brown, M.; Balkwill, D. 2009. Antibiotic resistance in bacteria isolated from the deep terrestrial subsurface. Microbial Ecology. 57(3):484-493.
Coombs, J. 2009. Potential for horizontal gene transfer in subsurface microbial communities. Totowa, New Jersey. In Gogarten M, Gogarten J, Olendzenski L (Eds).Humana Press. Horizontal Gene Transfer: Genomes in Flux. p 413-433.
Clinical and Laboratory Standards Institute. 2005. Clinical and Laboratory Standards Institute. Performance standards for antimicrobial susceptibility testing. Wayne, PA Fifteenth informational supplement, CLSI document M100- S15.
Chivian, D.; Brodie, E.; Alm, E.; Culley, D.; Dehal, P.; DeSantis, T.; & Onstott, T. C. 2008. Environmental genomics reveals a single species ecosystem deep within Earth. Science. 322(5899):275- 278.
Davies, J.; Davies, D. 2010. Origins and Evolution of Antibiotic Resistance.Microbiology and Molecular Biology Reviews. 74 (3): 417-433.
Diptendu, S.; Goutam, P. 2013. Molecular Characterization of Metal and Antibiotic Resistance Activities in a Bacterial Population Isolated from Waste Water Sample. Intenational Journal of Biotechnology and Bioengineering Research. 4(1):21-30.
Duran, F.; Carrero, P.; Rondón, C. 2005. A new method to determine mercury species in water samples through cold vapor generation and atomic absorption spectroscopy detection. Proceedings of the VII Venezuelan Chemistry Congress, Faculty of Sciences. Universidad de Los Andes (Mérida, Venezuela). p 35–37.
Fredrickson, J.; Balkwill, D. 2006. Geomicrobial processes and biodiversity in the deep terrestrial subsurface. Geomicrobiology Journal. 23(6): 345-356.
García-Sánchez, A.; Contreras, F.; Adams, M.; Santos, F. 2008. Mercury contamination or surface water and fish in a gold mining región (Cuyuni River Basin,Venezuela). International Journal Environment Pollution. 33(2/3) 260-274.
Gómez, W.; Ball, M.; Botello, W.; Yarzábal, A. 2013. Horizontal transfer of heavy metal and antibiotic-resistance markers between indigenous bacteria, colonizing mercury contaminated tailing ponds in southern Venezuela, and human pathogens. Revista de la Sociedad Venezolana de Microbiología. 33: 110- 115.
Hemme, C.; Deng, Y.; Gentry, T.; Fields, M.; Wu, L.; Barua, S.; & Zhou, J.. 2010. Metagenomic insights into evolution of a heavy metal - contamined groundwater microbial community. Multidisciplinary Journal of Microbial Ecology. 4: 660-672.
Hildebrand, R. 2005. Autochthonous and allochthonous strata of the El Callao greenstone belt: Implications for the nature of the Paleoproterozoic Trans- Amazonian orogeny and the origin of gold-bearing shear zones in the El Callao mining district, Guayana shield, Venezuela. Precambrian Research. 143(1): 75–86.
Hirayama, H.; Takai, K.; Inagaki, F.; Yamato, Y.; Suzuki, M.; Nealson, K.; Horikoshi, K. 2005. Bacterial community shift along a subsurface geothermal water stream in a Japanese gold mine. Extremophiles. 9 (2):169-184.
Izaki, K. 1981. Enzymatic reduction of mercurius and mercuric ions in Bacillus cereus. Canadian Journal of Microbiology. 27:192- 197.
Laskaris, P.; Tolbas, S.; Calvo-Bado, L.; Wellington E. 2010. Coevolution of antibiotic production and counter-resistance in soil bacteria. Enviromental Microbiology. 12(3):783-796.
Moser, D.; Gihring, T.; Brockman, F.; Fredrickson, J.; Balkwill, D.; Dollhopf, M.; & Onstott, T. C. 2005. Desulfotomaculum spp. and Methanobacterium spp. Dominate a 4 to 5 km Deep Fault. Applied and Environmental Microbiology. 71(12):8773-8783.
Mindlin, S.Z.; Soina, V.S.; Petrova, M.A.; Gorlenko, Zh.M. 2008. Isolation of antibiotic resistance bacterial strains from eastern Siberia permafrost sediments. Russian Journal of Genetics. 44(1): 36-44.
Ni Chadhain, S.; Schaefer, J.; Crane, S.; Zylstra, G.; Barkay, T. 2006. Analysis of mercuric reductase (merA) gene diversity in ananaerobic mercury-contaminated sediment enrichment. Environmental Microbiology. 8(10): 1746-1752.
Pan-Hou, H.; Nishimoto, M.; Imura, N.1981 Possible role of membrane protein in mercury resistance of Enterobacter aerogenes. Archives of Microbiology. 130(2): 93-95.
Paredes, S.; Hidalgo-Prada, B.; Ávila, R.; Albornoz, A. 2007. Análisis de mineral aurífero de la mina Colombia, El Callao, Venezuela, mediante MEB/EDX Y DRX. Acta Microscópica. 16: s301-302.
Parkes, R.; Linnane, C.; Webster, G.; Sass, H.; Weightman, A.; Hornibrook, R.; & Horsfield, B. 2011. Prokaryotes stimulate mineral H2 formation for the deep biosphere and subsequent thermogenic activity. Geology. 39(3): 219-222.
Rastogi, G.; Osman, S.; Kukkadapu, R.; Engelhard, M.; Vaishampayan, P.; Andersen, G.; Sani, R. 2010. Microbial and minerological characterizations of soils collected from the deep biosphere of the former Homestake gold mine, South Dakota. Microbial Ecology. 60 (3): 539-550.
Rastogi, G.; Gurram, R.; Bhalla, A.; Gonzalez, R.; Bischoff, K.; Hughes, S.; Kumar, S.; Sani, R. 2013. Presence of glucose, Xylose, and glycerol fermenting bacteria in the deep biosphere of the Homestake gold mine, South Dakota. Frontiers in Microbiology. 9: 18.
Reniero, D.; Mozzon, E.; Galli, E.; Barbieri, P. 1998. Two aberrant mercury resistance transposons in the Pseudomonas stutzeri plasmid pPB. Gene. 208(1): 37-42.
Reyes, N.; Marc, E.; Frischer, M.; Sobecky, P. 1999. Characterization of mercury resistance mechanisms in marine sediment microbial communities. FEMS Microbiology Ecology. 30 (3): 273-84.
Segawa, T.; Takeuchi, N.; Rivera, A.; Yamada, A.; Yoshimura, Y.; Barcaza, G., & Ushida, K. 2013. Distribution of antibiotic resistance genes in glacier environments. Environmental Ecology of Pathogens and Resistances. 5(1): 127-134.
Seiler, C.; Berendonk, T. 2012. Heavy metal driven co-selection of antibiotic resistance in soil and water bodies impacted by agriculture and aquaculture. Frontiers in Microbiology. 3:399.
Sudha, A.; Augustine, N.; Thomas, S. 2013. Emergence of multi-drug resistant bacteria in the Arctic, 790N. Journal of Cell and Life Sciences. 1(1): 1-5.
Summers, A. 2002. Generally Overlooked Fundamentals of Bacterial Genetics and Ecology. Clinical Infectious Diseases. 34: s84-s92.
Stepanauskas, R.; Glenn, T.; Jagoe, C.; Tuckfield, R.; Lindell, A.; King, C.; & McArthur, J. V.. 2006. Coselection for microbial resistance to metals and antibiotics in freshwater microcosms. Environmental Microbiology. 8(9): 1510–1514.
Takai, K.; Moser, D.; Deflaum, M.; Onstott, T.; Fredrickson, J. 2001. Archaeal Diversity in Waters from Deep South African Gold Mines. Applied and Environmental Microbiology. 67(12): 5750- 5760.
Telmer, K.; Veiga, MM. 2009. World emissions of mercury from artisanal and small scale gold mining. Pirrone N, Mason R (Eds). Springer Science + Business Media, p 131-172.
Uchimiya, M.; Lima, L.; Thomas, K.; Chang, S.; Wartelle, L.; Rodgers, J. 2010. Immobilization of heavy metal ions (CuII,CdII, NiII, and PbII) by broiler litter-derived biochars in water and soil. Journal of Agricultural Food Chemistry. 58(9): 5538-5344.
Veiga, M.; Bermudez, D.; Pacheco-Ferreira, H.; Martins, P.; Gunson, A.; Berrios, G.; Vos, L.; Hiudobro, P.; Roeser, M. 2005. Mercury from artisanal gold mining in Block B, El Callao, Bolivar State, Venezuela. In Pirrone N and Mahaffey K (Eds). Norwell, MA, USA. Dynamics of Mercury Pollution on Regional and Global Scales: Atmospheric Process and Human Exposure Around the World. Springer Publisher, p. 421-450.
Wang, F.; Lu, S.; Orcutt, B.; Xie, W.; Chen, P.; Xiao, X.; Edwards, J. 2013. Discovering the roles of subsurface microorganisms: Progress and future of deep biosphere investigation. ChineseScience Bulletin. 58 (4-5): 456-467.
Woodruff, L.; Cannon, W. 2010. Immediate and long-term fire effects on total mercury in forests soils of Northeastern Minnesota. Environmental Science & Technology. 44(14): 5371-5376.
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. Preeti Ranawat, Seema Rawat. (2018). Metal-tolerant thermophiles: metals as electron donors and acceptors, toxicity, tolerance and industrial applications. Environmental Science and Pollution Research, 25(5), p.4105. https://doi.org/10.1007/s11356-017-0869-2.
2. Paula Fernanda Alves Ferreira, Fernando Igne Rocha, Adina Howe, Daniele Rodrigues Barbosa, Ederson da Conceição Jesus, Nelson Moura Brasil do Amaral Sobrinho, Irene da Silva Coelho. (2024). Chemical attributes, bacterial community, and antibiotic resistance genes are affected by intensive use of soil in agro-ecosystems of the Atlantic Forest, Southeastern Brazil. Environmental Geochemistry and Health, 46(4) https://doi.org/10.1007/s10653-024-01894-8.
3. Delfina C. Domínguez, Luz María Chacón, D’Janique Wallace. (2021). Anthropogenic Activities and the Problem of Antibiotic Resistance in Latin America: A Water Issue. Water, 13(19), p.2693. https://doi.org/10.3390/w13192693.
4. Di Chen, Qiyan Feng. (2023). Microbial Community Structure, Metabolic Function, and Phenotypic Characteristics of Sediment in Deep Coal Mine Underground Environment, China. Water, 15(13), p.2371. https://doi.org/10.3390/w15132371.
5. S. Elizabeth George, Yongshan Wan. (2020). Advances in characterizing microbial community change and resistance upon exposure to lead contamination: Implications for ecological risk assessment. Critical Reviews in Environmental Science and Technology, 50(21), p.2223. https://doi.org/10.1080/10643389.2019.1698260.
6. Zaima Azira Zainal Abidin, Puteri Nur Ezzati Badaruddin, Ahmed Jalal Khan Chowdhury. (2020). Isolation of heavy metal resistance bacteria from lake sediment of IIUM, Kuantan. Desalination and Water Treatment, 188, p.431. https://doi.org/10.5004/dwt.2020.25298.
7. Haliru Musa, Farizul Hafiz Kasim, Ahmad Anas Nagoor Gunny, Subash C.B. Gopinath, Suresh V. Chinni, Mohd. Azmier Ahmad. (2019). Whole genome sequence of moderate halophilic marine bacterium Marinobacter litoralis SW-45: Abundance of non-coding RNAs. International Journal of Biological Macromolecules, 133, p.1288. https://doi.org/10.1016/j.ijbiomac.2019.05.003.
8. Luis F. Espinosa-Camacho, Gabriela Delgado, Alejandro Cravioto, Rosario Morales-Espinosa. (2022). Diversity in the composition of the accessory genome of Mexican Pseudomonas aeruginosa strains. Genes & Genomics, 44(1), p.53. https://doi.org/10.1007/s13258-021-01155-3.
9. Mustafa Ture, Muhammed Baran Kilic, Ilhan Altinok. (2021). Relationship Between Heavy Metal Accumulation in Fish Muscle and Heavy Metal Resistance Genes in Bacteria Isolated from Fish. Biological Trace Element Research, 199(4), p.1595. https://doi.org/10.1007/s12011-020-02246-0.
10. Ivana Timková, Miroslava Lachká, Jana Kisková, Lenka Maliničová, Lea Nosáľová, Peter Pristaš, Jana Sedláková-Kaduková. (2020). High frequency of antibiotic tolerance in deep subsurface heterotrophic cultivable bacteria from the Rozália Gold Mine, Slovakia. Environmental Science and Pollution Research, 27(35), p.44036. https://doi.org/10.1007/s11356-020-10347-5.
11. Cristóbal Lárez Velásquez, Maura Rojas, Milibeth Peña, María José Peña-Vera, Miguel Sulbarán, Elizabeth Pérez. (2019). Characterization and determination of antimicrobial and metal resistant profiles of Xanthomonas strains isolated from natural environments. Journal of Analytical & Pharmaceutical Research, 8(2), p.55. https://doi.org/10.15406/japlr.2019.08.00312.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2014 Revista Colombiana de Biotecnología

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
Esta es una revista de acceso abierto distribuida bajo los términos de la Licencia Creative Commons Atribución 4.0 Internacional (CC BY). Se permite el uso, distribución o reproducción en otros medios, siempre que se citen el autor(es) original y la revista, de conformidad con la práctica académica aceptada. El uso, distribución o reproducción está permitido desde que cumpla con estos términos.
Todo artículo sometido a la Revista debe estar acompañado de la carta de originalidad. DESCARGAR AQUI (español) (inglés).