Effect of seminal plasma and sperm of boars valued by freezability on seminal cryopreservation
Efecto de plasma seminal y espermatozoides de verracos valorados por congelabilidad sobre la criopreservación seminal
DOI:
https://doi.org/10.15446/rfna.v69n2.59135Keywords:
Boar, Reproduction, Seminal preservation, Seminal quality (en)Cerdo, Reproducción, Preservación seminal, Calidad seminal (es)
DOI: https://doi.org/10.15446/rfna.v69n2.59135
Effect of seminal plasma and sperm of boars valued by freezability on seminal cryopreservation
Efecto de plasma seminal y espermatozoides de verracos valorados por congelabilidad sobre la criopreservación seminal
Francisco Javier Henao Uribe1, Julián Valencia Giraldo1, Henry Mesa1 and Germán Gómez Londoño1
1 Facultad de Ciencias Agropecuarias. Universidad de Caldas. A.A. 275, Manizales, Colombia. <fhenao@ucaldas.edu.co>
Received: March 4, 2016; Accepted: May 16, 2016
This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.

ABSTRACT
The aim of this study was to determine the effect of sperm and seminal plasma (SP) on the freezability of porcine semen. Semen of eight commercial males from two farms in the central-western region of Colombia (four boars in each farm) was frozen and tested to select two males with high freezability (MHF) and two with low freezability (MLF), according to the percentage of functionally competent sperm (FCS). Immediately after the collection was completed, the SP and sperm from the males selected were separated by centrifugation to combine the two types of plasma with the two types of sperm, incubate them for three hours and then freeze them. The variables evaluated were: sperm morphology, structural and functional integrity of plasmatic membrane, progressive and total motility, DNA fragmentation, acrosome integrity, capacitated sperm and FCS. The combination of sperm and plasma of MHF recorded the highest value (P<0.01) of acrosome integrity (24.3 ± 0.082 vs 6.076 ± 0.16) when compared to MLF plasma and cells. Membrane structural integrity was higher (P<0.01) with MHF (53.56 ± 0.0395) than with MLF plasma (47.49 ± 0.0419). The differences in porcine semen freezability depend on interactions between seminal plasma and sperm.
Key words: Boar, Reproduction, Seminal preservation, Seminal quality
RESUMEN
El objetivo del presente trabajo fue determinar el efecto de los espermatozoides y del plasma seminal (PS) sobre la congelabilidad del semen porcino. El semen de ocho machos comerciales, de dos granjas del centro-occidente de Colombia (cuatro machos en cada granja), fue congelado y evaluado para seleccionar dos machos con alta congelabilidad (MAC) y dos con baja (MBC), según el porcentaje de espermatozoides funcionalmente competentes (EFC). Inmediatamente finalizada la colección, fueron separados por centrifugación el PS y los espermatozoides de los machos seleccionados, para combinar los dos tipos de plasma con los dos tipos de espermatozoides, incubar tres horas y congelar. Las variables evaluadas fueron: morfología espermática, integridad estructural y funcional de la membrana plasmática, movilidad progresiva y total, fragmentación del ADN, integridad acrosómica, espermatozoides capacitados y EFC. La combinación de espermatozoides y plasma de MAC registró el valor más alto (P<0,01) de integridad acrosómica (24,3 ± 0,082 vs 6,076 ± 0,16) al compararlo con células y plasma de MBC. La integridad estructural de membrana fue mayor (P<0,01) con plasma de MAC (53,56 ± 0,0395) que con el de MBC (47,49 ± 0,0419). Las diferencias en la congelabilidad del semen de los porcinos dependen de las interacciones entre el plasma seminal y los espermatozoides.
Palabras claves: Cerdo, Reproducción, Preservación seminal, Calidad seminal
A dvancements such as deep intrauterine insemination and FlatPack™ containers have allowed the use of frozen-thawed porcine semen with acceptable results (Roca et al., 2006). However, they are of little consistence due mainly to the difference in freezability between males (Holt, 2000; Thurston et al., 2002) thus providing a variation of 70% in results (Roca et al., 2006) apparently due to genetic factors (Thurston et al., 2002) which, according to Holt (2000), make it unclear whether they affect the composition of SP and functionality of accessory sex glands, or the biochemistry and physiology of sperm.
Findings show that differences in freezability between males disappear when cryopreserving sperm obtained directly from the epididymis, which has had no contact with SP (Rath and Niemann, 1997), and reduce when freezing the sperm peak fraction (first 10 mL of the rich fraction) poor in SP and abundant in sperm (Saravia et al., 2007). In addition, it is known that SP has beneficial effects on post-thaw semen quality (Saravia et al., 2009) and that its components vary between males (Caballero, 2007) which suggests that SP is a factor closely related to freezability.
On the other hand, differences in the proportion of polyunsaturated fatty acids in the plasma membrane of the sperm of males with different freezability have been found (Waterhouse et al., 2006), and also the storage of semen at 17 °C, for a period prior to its cooling at 5 °C, modulates lipid changes of plasma membrane, increasing the resistance of the sperm to cold shock, possibly by an interaction between them and SP (Casas and Althouse, 2013). Based on this, it becomes relevant to try to establish the effect of plasma and sperm of boars valued by freezability on seminal cryopreservation.
MATERIALS AND METHODS
This study was conducted using an ejaculate from each of the eight boars (18 to 24 months) from two commercial farms (four from each farm) in the central-western region of Colombia, housed in individual pens, fed with 2 kg per animal per day of a commercial product for boars, with plenty of water available and ejaculated with a frequency ranging from four to eight days, between 6:00 and 9:00 am.
The freezing protocol used was Minitub®, comprising the following steps: rich fraction collection by the gloved hand technique and dilution 1:1.5 (1 semen and 1.5 Androhep® plus), at 36 °C; temperature reduction down to 17 °C within three hours and transport to the Biology of Reproduction Laboratory at Universidad de Caldas (Colombia) for freezing and seminal analysis; determination of sperm concentration by photometry and preparation of a 30x106 spermatozoa mL-1 aliquot for semen analysis; centrifugation of the remaining sperm at 800 g for 20 min, and pellet adjustment at a concentration of 2x109 spermatozoa mL-1 with cooling diluent (AndrostarCryo plus® + 20% egg yolk) at 17 °C, to subsequently take it to 5 °C at a rate of 1 °C every 7.5 min, adjusted to a concentration of 1x109 spermatozoa mL-1 with freezing diluent (diluent cooling + 6% glycerol) at 5 °C, and packaged into 0.5 mL French straws at 5 °C. Finally, the straws were exposed to nitrogen vapors (5 cm above the nitrogen level) for 20 min and stored in liquid nitrogen.
Three straws by boar were thawed in a water bath at 38 °C for 20 s, and the semen diluted in Androhep® plus at 26 °C at a rate of 7.5 mL per straw; each straw was evaluated independently.
The seminal evaluation considered the following variables:
- Membrane structural integrity (MSI), by staining with SYBR-14 and propidium iodide (PI) (LIVE/DEAD® Sperm Viability Kit L-7011, Molecular Probes Europe, Invitrogen); and visualization at 1000 X in a Nikon Eclipse 80i microscope with epifluorescence and B-2A filter (Garner and Johnson, 1995).
- Membrane functional integrity (MFI) by short Hypoosmotic Swelling Test (sHOST) (Pérez-Llano et al., 2001).
- Acrosomal integrity by Pursel and Johnson technique (1974).
- Sperm morphology and semen agglutination fixed with formol-saline, 1:1 dilution.
The visualization of the above three variables was performed on a Nikon Eclipse 50i microscope, phase contrast; sHOST and morphology at 400X and acrosomal integrity at 1000X.
- Semen motility in computer assisted semen analysis (CASA). The system used was: Spem-Class Analyzer, version 5.2.0.1 Update SCA®, 2010. Microptic, Barcelona, Spain.
- Combined sHOST test with viability (Pérez-Llano et al., 2009). Two vials were prepared: vial 1 with 1.5 mL of semen at 17 °C and vial 2 with 1 mL of hypoosmotic solution at 75 mOsm/kg, 5 µL SYBR-14 and 5 µL of PI, both taken to 37 °C in a water bath; 500 µL of semen were transferred from vial 1 to vial 2, incubated for 5 min at 37 °C, fixed with 10 µL of 2% glutaraldehyde in Beltsville thawing solution, to evaluate acrosome domain (Pursel and Johnson, 1974). In this test, a Nikon Eclipse 80i microscope with interference contrast and epifluorescence filter B-2A at 1000X was used. The result of the test expressed two populations: functionally competent sperm percentage (FCS), that is, total living cells, morphologically normal, sHOST+ with intact acrosome, and the percentage of sHOST+ sperm, alive with true acrosome reaction or capacitated sperm (CS). Both populations were adjusted by previous agglutination values and round shapes tail.
- Sperm DNA integrity by chromatin dispersion test (Enciso et al., 2006) with the commercial Kit Halotech® (ChromaCell SL, Madrid, Spain). The result was expressed as DNA fragmentation index.
Freezability was determined by FCS testing in frozen-thawed semen. One male with high freezability (MHF) and one with low freezability (MLF) were selected in each farm.
In a second phase, in order to apply a model for evaluating the effect of SP and sperm on freezability, it was necessary to perform centrifugation of the rich fraction of each selected male immediately after collection, this in order to avoid interaction between these two components. This centrifugation was performed at 800 g for 20 min; besides, a 10 mL aliquot was not centrifuged to evaluate the preliminary effect of centrifugation. Then, based on the seminal material obtained (SP and sperm), 4 combinations were carried out as follows: MLF sperm + MHF SP (LFS+HFSP), MHF sperm + MLF SP (HFS+LFSP), MLF sperm + its own SP (LFS+LFSP) and MHF sperm + their own SP (HFS+HFSP).
The effect of SP and sperm on freezability was analyzed by a 2x2 factorial design, two SPs (MHF SP and MLF SP) and two types of sperm (MHF sperm and MLF sperm) in a randomized complete block design (blocked by farm); the dependent variables were evaluated three times. A preliminary comparison employing an aliquot without spinning was made to trace a possible effect on semen quality. The different effects were analyzed using a log-linear model, consisting of a Poisson regression with a logarithmic function with the PROC GENMOD-SAS program (SAS Inst. Cary, NC). The results were expressed as minimum mean-square error.
RESULTS AND DISCUSSION
The seminal evaluations of the eight boars are recorded in Table 1. The males with high freezability (MHF) were number 1 in each farm and the two males with low freezability (MLF) were number four.
Effect of sperm and seminal plasma on semen quality
There was no significant effect (P>0.05) of factors in this study: sperm x SP, SP and sperm interaction on values: DNA fragmentation, cytoplasmic droplets and agglutinated cells. What was found for DNA fragmentation is consistent with the findings of Chanapiwat et al. (2010) who showed little DNA damage in the frozen-thawed semen, and differs from the findings of Urrego et al. (2008) who showed DNA fragmentation caused by centrifugation and by generation of reactive oxygen species. In connection with cytoplasmic droplets, the results of this study differ from the findings of a previous experience (Henao et al., 2011) in which it was possible to reduce this malformation by incubation with SP in boars with high levels of cytoplasmic droplets. In the case of agglutination, no reports on its effect on semen freezability were found.
Sperm x SP interaction did not affect (P>0.05) DNA integrity, cytoplasmic droplets, agglutinated cells, MSI, MFI, FCS, normal sperm, or coiled tails. However, it affected in great extent (P<0.01) acrosome integrity, and significantly (P<0.05) total and progressive motility, CS and intensely coiled tails. SP factor only affected (P<0.05) MSI; and sperm factor affected (P<0.01) MFI, normal sperm, coiled tails and FCS.
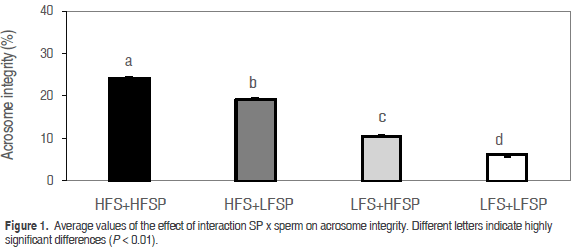
The multiple comparisons test for acrosome integrity allowed establishing differences (P<0.01) between and among treatments, with the highest value for HFS+HFSP (24.3 ± 0.082) followed by HFS+LFSP (19.18 ± 0.092), LFS+HFSP (10.55 ± 0.12) and LFS+LFSP (6.076 ± 0.16) (Figure 1).
Differences in the acrosome integrity between high and low freezability males (Flores et al., 2009; Casas et al., 2009) were found in previous reports, without considering specifically the effects of seminal plasma and origin of sperm. There are reports on the presence of spermadhesines PSP-I/PSP-II (seminal porcine plasma heterodimers I and II) in SP (Caballero, 2007) and proteins of sperm surface called DQH (glutamine-histidine-aspartic acid), preserving the integrity of the acrosome (Maňásková et al., 2007), which may be related to the fact that the mixture of MHF SP with MLF sperm have registered acrosome integrity higher than the mixture of MLF SP with MLF sperm (P<0.01) in this study (Figure 1). Better values for acrosome integrity (P<0.01) in both mixtures with MHF sperm were recorded, which may be associated with the amount of fatty acids in the sperm membrane associated with acrosome reaction (Tortolero et al., 2005), and the proportion of phosphorylcholine and cholesterol in the sperm membrane, since these two molecules are recipients of most acrosome protective proteins (Jonáková et al., 2000).
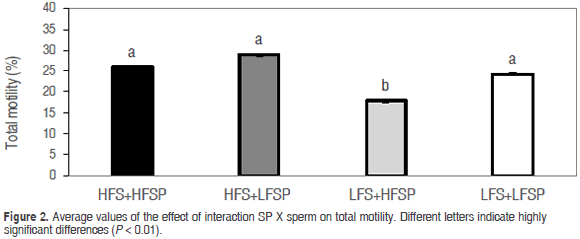
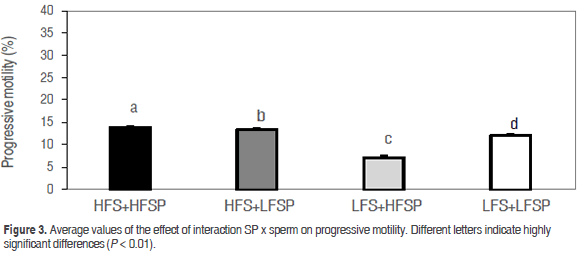
Total motility values were statistically equal in treatments HFS+HFSP (25.9 ± 0.07), HFS+LFSP (28.9 ± 0.07) and LFS+LFSP (24.34 ± 0.06), while treatment LFS+HFSP (17.86 ± 0.07) presented the lowest value (P<0.01) (Figure 2); in the case of progressive motility, it was found that HFS+HFSP, HFS+LFSP and LFS+LFSP showed equal values (P<0.05) (13.85 ± 0.1, 13.48 ± 0.11 and 12.01 ± 0.12, respectively) and a significantly lower value (P<0.01) for LFS+HFSP (7.13 ± 0.15) (Figure 3).
Although the highest value was obtained in the combination of sperm and SP of MHF, upon adding this plasma to MLF sperm, a negative effect occurred on total and progressive motility. This differs from the positive effect of SP on motility found by Saravia et al. (2009), described by these authors with different concentrations of protein and bicarbonate, and improved motility in frozen-thawed semen by addition of SP of MHF to semen of MLF (Hernandez et al., 2007), associated with the protective effect of spermadhesines PSP I and II (Caballero, 2007). However, these studies did not include a mixture of sperm and SP in the period between collection and cooling to 17 °C.
The combinations with the lowest CS values in the test of multiple comparisons were LFS+HFSP (7.2 ± 0.14) and LFS+LFSP (8.1 ± 0.13), statistically different (P<0.05), and higher (P<0.01), simultaneously different between them (P<0.01), corresponded to combinations HFS+HFSP (17.41 ± 0.09) and HFS+LFSP (11.69 ± 0.11) (Figure 4). However, when considering the total percentage of living sHOST+ sperm (with intact and capacitated acrosome), the combination LFS+LFSP registered the maximum reduction of FCS due to 61.6% sperm capacitation. In addition to assess the effect of the sperm factor on FCS, it was found that MHF sperm preserved more functional capacity than MLF sperm (13.88 ± 0.08 vs 5.43 ± 0.124 of FCS, P<0.01). All combinations showed reduced FCS by capacitation in frozen-thawed sperm. This phenomenon is known as cryo-capacitation, and it represents one of the most important effects of freezing by reducing the number of sperm with fertilizing potential, that is, the relevant functionally (Bravo et al., 2005). In this context, cryo-capacitation prevention is necessary, and trials have been developed on SP addition for this purpose, because it contains capacitated proteins (Saravia et al., 2009) which possibly had a lower concentration in the MLF seminal plasma.
MSI value was higher (P<0.01) (53.56 ± 0.0395) with MHF SP compared to that of MLF (47.49 ± 0.0419). In turn, MFI value was higher (P<0.01) (62.38 ± 0.04 for MHF sperm vs 47.97 ± 0.04 for MLF sperm). It has previously been reported that the addition of MHF SP improves MSI and motility of semen in low freezability males (Hernandez et al., 2007). In addition, the cooling of semen at 17 °C for 3 to 24 hours prior to cold shock has protective effect on the sperm, as it modulates lipid changes of the plasma membrane and maintains its integrity, this possibly due to SP proteins (Casas and Althouse, 2013) such as spermadhesines PSP I and II, AQN (alanine-glutamine-asparagine) and AWN (alanine-tryptophan-asparagine), and protein DQH which bind to the sperm membrane for protection and stabilization (Jonáková et al., 2000). On the other hand, there are reports of differences in MFI between males of different fertility (Pérez-Llano et al., 2001) and SP effect of the poor-sperm fraction on this variable (Henao et al., 2011). However, it is unknown whether the plasma of the rich fraction has an effect on MFI in the period between collection and cooling to 17 °C.
Sperm with normal morphology and coiled tails were affected (P<0.01) by the factor sperm, in benefit of the MHF ones. Most of the changes related to these two variables occur during spermatogenesis and maturation (Briz and Fabrega, 2013) and in these processes the sperm has not yet had contact with secretions from the accessory sex glands, which could explain the absence of SP effect of high or low freezability males.
Intensely coiled tails were affected (P<0.05) by the interaction sperm x SP, and statistically equal values were found for treatments HFS+HFSP, HFS+LFSP and LFS+HFSP (0.157 ± 1.01, 0.314 ± 0.72 and 0.47 ± 0.595 respectively) and coiled tails were found for treatment LFS+LFSP with highly significant difference (P<0.01). It is noteworthy that, although there was a difference in the value of this variable in one of the treatments, all values were well below those found for this type of abnormality (1 to 5%) (Briz and Fabrega, 2013) to consider any condition in semen quality.
Finally, in the preliminary comparison between spin and no spin semen, a possible affectation was found on: acrosome integrity, progressive and total motility, FCS and CS. It is accepted that the damage caused by centrifugation may be due to direct mechanical damage (Carvajal et al. 2004) or excessive generation of reactive oxygen species by lipid peroxidation of the membrane, leading to DNA fragmentation (Urrego et al., 2008), alteration in the membrane structural integrity and reduced motility (Carvajal et al., 2004). It is important to note that, according to Pérez-Llano et al. (2009), sperm with real acrosome reaction correspond to those which have had a capacitation process, different from acrosome damage by membrane degeneration in dead sperm. On the other hand, it is known that reactive oxygen species, increased by effect of centrifugation (Urrego et al., 2008) are involved in the process of sperm capacitation and acrosome reaction as they affect membrane fluidity by degradation of polyunsaturated fatty acids (Tortolero et al., 2005).The above preliminary findings show that centrifugation could lead to alterations in semen quality.
CONCLUSIONS
Evidence was found in this study that differences in boar semen freezability depend on both the seminal plasma and sperm; therefore, the two types of substitution (plasma or cells) could improve post-freeze semen quality.
The effect of the interaction between sperm and seminal plasma affected (P<0.01) acrosome integrity. The combination of cells and plasma of high freezability males recorded the highest values of acrosome integrity, and those of low freezability cells and plasma, lower values. Apparently, the substitution of low freezability cells with high ones is more effective than the substitution of low freezability plasma with high one; in the first case, a much larger increase is evident in acrosome integrity.
High freezability male sperm combinations recorded higher percentage of functionally competent sperm than low freezability males. The combination of cells and plasma of low freezability males recorded the maximum reduction of functionally competent sperm due to sperm capacitation, and adding high-freezability plasma to low freezability sperm reduced the capacitation percentage.
The plasma of high freezability males was associated with higher values for structural integrity of the membrane; similarly, the sperm of high freezability males had better functional integrity of the membrane.
REFERENCES
Bravo MM, Aparicio IM, Garcia-Herreros M, Gil MC, Pena FJ and Garcia-Marin LJ. 2005. Changes in tyrosine phosphorylation associated with true capacitation and capacitation-like state in boar sperm. Molecular reproduction and development 71(1): 88-96. doi: 10.1002/mrd.20286
Briz MD and Fábrega A. 2013. Chapter 1: Boar Sperm Within the Male Genital Tract, The boar spermatozoon. pp. 3-47. In: Bonet S, Casas I, Holt WV and Yeste M. (eds.). Boar Reproduction. First edition, Springer Berlin Heidelberg, Berlin. 632 p.
Caballero I. 2007. Estudio del plasma seminal y la espermadhesina PSP-I/PSP-II sobre la funcionalidad de los espermatozoides de verraco. Doctoral thesis. Veterinary School. University of Murcia. 263 p.
Carvajal G, Cuello C, Ruiz M, Vazquez JM, Martinez EA and Roca J. 2004. Effects of centrifugation before freezing on boar sperm cryosurvival. Journal of Andrology 25(3): 389-396. doi: 10.1002/j.1939-4640.2004.tb02805.x
Casas I and Althouse GC. 2013. The protective effect of a 17 degrees C holding time on boar sperm plasma membrane fluidity after exposure to 5 degrees C. Cryobiology 66(1): 69-75. doi: 10.1016/j.cryobiol.2012.11.006
Casas I, Sancho S, Briz M., Pinart E, Bussalleu E, Yeste M and Bonet S. 2009. Freezability prediction of boar ejaculates assessed by functional sperm parameters and sperm proteins. Theriogenology 72(7): 930-948. doi: 10.1016/j.theriogenology.2009.07.001
Chanapiwat P, Kaeoket K, Tummaruk P. 2010. The Sperm DNA Damage after Cryopreservation of Boar Semen in Relation to Post-thawed Semen Qualities, Antioxidant Supplementation and Boars Effects. The Thai Journal of Veterinary Medicine 40(2):187-193.
Enciso M, Lopez-Fernandez C, Fernandez JL, Garcia P, Gosalbez A and Gosalvez J. 2006. A new method to analyze boar sperm DNA fragmentation under bright-field or fluorescence microscopy. Theriogenology 65(2): 308-316. doi: 10.1016/j.therioge nology.2005.05.044
Flores E, Fernandez-Novell JM, Peña A and Rodriguez-Gil JE. 2009. The degree of resistance to freezing-thawing is related to specific changes in the structures of motile sperm subpopulations and mitochondrial activity in boar sperm. Theriogenology 72(6): 784-797. doi: 10.1016/j.theriogenology.2009.05.013
Garner DL and Johnson LA. 1995. Viability assessment of mammalian sperm using SYBR-14 and propidium iodide. Biology of reproduction 53(2): 276-284. doi: 10.1095/biolreprod53.2.276
Henao FJ, Valencia JA, Díaz O, Rangel MY. 2011. Efecto de la adición de plasma seminal sobre la eliminación de gotas citoplásmicas en semen de sus scrofa linnaeus. Boletín científico centro de museos museo de historia natural 15(2): 94-104.
Hernandez M, Roca J, Calvete JJ, Sanz L, Muino-Blanco T, Cebrian-Perez JA, Vazquez JM and Martinez EA. 2007. Cryosurvival and in vitro fertilizing capacity postthaw is improved when boar sperm are frozen in the presence of seminal plasma from good freezer boars. Journal of andrology 28(5): 689-697. doi: 10.2164/jandrol.107.002725
Holt WV. 2000. Fundamental aspects of sperm cryobiology: the importance of species and individual differences. Theriogenology 53(1): 47-58. doi: 10.1016/S0093-691X(99)00239-3.
Jonáková V, Manaskova P, Kraus M, Liberda J and Ticha M. 2000. Sperm surface proteins in mammalian fertilization. Molecular reproduction and development 56(2 Suppl): 275-277. doi: 10.1002/(SICI)1098-2795(200006)56:2+<275::AID-MRD13>3.0.CO;2-G
Maňásková P, Peknicova J, Elzeinova F, Ticha M and Jonáková V. 2007. Origin, localization and binding abilities of boar DQH sperm surface protein tested by specific monoclonal antibodies. Journal of Reproductive Immunology 74(1-2): 103-113. doi: 10.1016/j.jri.2006.11.003
Pérez-Llano B, Lorenzo JL, Yenes P, Trejo A and Garcia-Casado P. 2001. A short hypoosmotic swelling test for the prediction of boar sperm fertility. Theriogenology 56 (3): 387-398. doi: 10.1016/S0093-691X(01)00571-4.
Pérez-Llano B, Sala R, Reguera G and Garcia-Casado P. 2009. Changes in subpopulations of boar sperm defined according to viability and plasma and acrosome membrane status observed during storage at 15 degrees C. Theriogenology 71 (2): 311-317. doi: 10.1016/j.theriogenology.2008.07.023
Pursel VG. and Johnson LA. 1974. Glutaraldehyde fixation of boar sperm for acrosome evaluation. Theriogenology 1(2): 63-68. doi: 10.1016/0093-691X(74)90008-9
Rath D and Niemann H. 1997. In vitro fertilization of porcine oocytes with fresh and frozen-thawed ejaculated or frozen-thawed epididymal semen obtained from identical boars. Theriogenology 47(4): 785-793. doi: 10.1016/S0093-691X(97)00034-4
Roca J, Hernández M, Carvajal G, Vázquez JM and Martínez EA. 2006. Factors influencing boar sperm cryosurvival. Journal of Animal Science 84: 2692-2699. doi: 10.2527/jas.2006-094
Saravia F, Hernandez M, Wallgren M, Johannisson A and Rodriguez-Martinez H. 2007. Controlled cooling during semen cryopreservation does not induce capacitation of sperm from two portions of the boar ejaculate. International journal of andrology 30 (6): 485-499. doi: 10.1111/j.1365-2605.2006.00741.x
Saravia F, Wallgren M, Johannisson A, Calvete JJ, Sanz L, Peña FJ, Roca J and Rodriguez-Martinez H. 2009. Exposure to the seminal plasma of different portions of the boar ejaculate modulates the survival of sperm cryopreserved in MiniFlatPacks. Theriogenology 71 (4): 662-675. doi: 10.1016/j.theriogenology.2008.09.037
Thurston LM, Siggins K, Mileham AJ, Watson PF and Holt WV. 2002. Identification of amplified restriction fragment length polymorphism markers linked to genes controlling boar sperm viability following cryopreservation. Biology of reproduction 66 (3): 545-554. doi: 10.1095/biolreprod66.3.545.
Tortolero I, Arata-Bellabarba G, Osuna JA, Gómez R, Regadera J. 2005. Estrés oxidativo y función espermática. 2005. Revista Venezolana de Endocrinología y Metabolismo 3 (3): 12-19.
Urrego R, Ríos A, Olivera Ángel M, Camargo O. 2008. Efecto de la centrifugación sobre la membrana plasmática y el ADN de espermatozoides bovinos. Revista Colombiana de Ciencias Pecuarias 21 (1):19-26.
Waterhouse KE, Hofmo PO, Tverdal A and Miller Jr. RR. 2006. Within and between breed differences in freezing tolerance and plasma membrane fatty acid composition of boar sperm. Reproduction 131 (5): 887-894. doi: 10.1530/rep.1.01049
References
Bravo MM, Aparicio IM, Garcia-Herreros M, Gil MC, Pena FJ and Garcia-Marin LJ. 2005. Changes in tyrosine phosphorylation associated with true capacitation and capacitation-like state in boar sperm. Molecular reproduction and development 71(1): 88-96. doi: 10.1002/mrd.20286
Briz MD and Fábrega A. 2013. Chapter 1: Boar Sperm Within the Male Genital Tract, The boar spermatozoon. pp. 3-47. In: Bonet S, Casas I, Holt WV and Yeste M. (eds.). Boar Reproduction. First edition, Springer Berlin Heidelberg, Berlin. 632 p.
Caballero I. 2007. Estudio del plasma seminal y la espermadhesina PSP-I/PSP-II sobre la funcionalidad de los espermatozoides de verraco. Doctoral thesis. Veterinary School. University of Murcia. 263 p.
Carvajal G, Cuello C, Ruiz M, Vazquez JM, Martinez EA and Roca J. 2004. Effects of centrifugation before freezing on boar sperm cryosurvival. Journal of Andrology 25(3): 389-396. doi: 10.1002/j.1939-4640.2004.tb02805.x
Casas I and Althouse GC. 2013. The protective effect of a 17 degrees C holding time on boar sperm plasma membrane fluidity after exposure to 5 degrees C. Cryobiology 66(1): 69-75. doi: 10.1016/j.cryobiol.2012.11.006
Casas I, Sancho S, Briz M., Pinart E, Bussalleu E, Yeste M and Bonet S. 2009. Freezability prediction of boar ejaculates assessed by functional sperm parameters and sperm proteins. Theriogenology 72(7): 930-948. doi: 10.1016/j.theriogenology.2009.07.001
Chanapiwat P, Kaeoket K, Tummaruk P. 2010. The Sperm DNA Damage after Cryopreservation of Boar Semen in Relation to Post-thawed Semen Qualities, Antioxidant Supplementation and Boars Effects. The Thai Journal of Veterinary Medicine 40(2):187-193.
Enciso M, Lopez-Fernandez C, Fernandez JL, Garcia P, Gosalbez A and Gosalvez J. 2006. A new method to analyze boar sperm DNA fragmentation under bright-field or fluorescence microscopy. Theriogenology 65(2): 308-316. doi: 10.1016/j.therioge nology.2005.05.044
Flores E, Fernandez-Novell JM, Peña A and Rodriguez-Gil JE. 2009. The degree of resistance to freezing-thawing is related to specific changes in the structures of motile sperm subpopulations and mitochondrial activity in boar sperm. Theriogenology 72(6): 784-797. doi: 10.1016/j.theriogenology.2009.05.013
Garner DL and Johnson LA. 1995. Viability assessment of mammalian sperm using SYBR-14 and propidium iodide. Biology of reproduction 53(2): 276-284. doi: 10.1095/biolreprod53.2.276
Henao FJ, Valencia JA, Díaz O, Rangel MY. 2011. Efecto de la adición de plasma seminal sobre la eliminación de gotas citoplásmicas en semen de sus scrofa linnaeus. Boletín científico centro de museos museo de historia natural 15(2): 94-104.
Hernandez M, Roca J, Calvete JJ, Sanz L, Muino-Blanco T, Cebrian-Perez JA, Vazquez JM and Martinez EA. 2007. Cryosurvival and in vitro fertilizing capacity postthaw is improved when boar sperm are frozen in the presence of seminal plasma from good freezer boars. Journal of andrology 28(5): 689-697. doi: 10.2164/jandrol.107.002725
Holt WV. 2000. Fundamental aspects of sperm cryobiology: the importance of species and individual differences. Theriogenology 53(1): 47-58. doi: 10.1016/S0093-691X(99)00239-3.
Jonáková V, Manaskova P, Kraus M, Liberda J and Ticha M. 2000. Sperm surface proteins in mammalian fertilization. Molecular reproduction and development 56(2 Suppl): 275-277. doi: 10.1002/(SICI)1098-2795(200006)56:2+<275::AID-MRD13>3.0.CO;2-G
Maňásková P, Peknicova J, Elzeinova F, Ticha M and Jonáková V. 2007. Origin, localization and binding abilities of boar DQH sperm surface protein tested by specific monoclonal antibodies. Journal of Reproductive Immunology 74(1-2): 103-113. doi: 10.1016/j.jri.2006.11.003
Pérez-Llano B, Lorenzo JL, Yenes P, Trejo A and Garcia-Casado P. 2001. A short hypoosmotic swelling test for the prediction of boar sperm fertility. Theriogenology 56 (3): 387-398. doi: 10.1016/S0093-691X(01)00571-4.
Pérez-Llano B, Sala R, Reguera G and Garcia-Casado P. 2009. Changes in subpopulations of boar sperm defined according to viability and plasma and acrosome membrane status observed during storage at 15 degrees C. Theriogenology 71 (2): 311-317. doi: 10.1016/j.theriogenology.2008.07.023
Pursel VG. and Johnson LA. 1974. Glutaraldehyde fixation of boar sperm for acrosome evaluation. Theriogenology 1(2): 63-68. doi: 10.1016/0093-691X(74)90008-9
Rath D and Niemann H. 1997. In vitro fertilization of porcine oocytes with fresh and frozen-thawed ejaculated or frozen-thawed epididymal semen obtained from identical boars. Theriogenology 47(4): 785-793. doi: 10.1016/S0093-691X(97)00034-4
Roca J, Hernández M, Carvajal G, Vázquez JM and Martínez EA. 2006. Factors influencing boar sperm cryosurvival. Journal of Animal Science 84: 2692-2699. doi: 10.2527/jas.2006-094
Saravia F, Hernandez M, Wallgren M, Johannisson A and Rodriguez-Martinez H. 2007. Controlled cooling during semen cryopreservation does not induce capacitation of sperm from two portions of the boar ejaculate. International journal of andrology 30 (6): 485-499. doi: 10.1111/j.1365-2605.2006.00741.x
Saravia F, Wallgren M, Johannisson A, Calvete JJ, Sanz L, Peña FJ, Roca J and Rodriguez-Martinez H. 2009. Exposure to the seminal plasma of different portions of the boar ejaculate modulates the survival of sperm cryopreserved in MiniFlatPacks. Theriogenology 71 (4): 662-675. doi: 10.1016/j.theriogenology.2008.09.037
Thurston LM, Siggins K, Mileham AJ, Watson PF and Holt WV. 2002. Identification of amplified restriction fragment length polymorphism markers linked to genes controlling boar sperm viability following cryopreservation. Biology of reproduction 66 (3): 545-554. doi: 10.1095/biolreprod66.3.545.
Tortolero I, Arata-Bellabarba G, Osuna JA, Gómez R, Regadera J. 2005. Estrés oxidativo y función espermática. 2005. Revista Venezolana de Endocrinología y Metabolismo 3 (3): 12-19.
Urrego R, Ríos A, Olivera Ángel M, Camargo O. 2008. Efecto de la centrifugación sobre la membrana plasmática y el ADN de espermatozoides bovinos. Revista Colombiana de Ciencias Pecuarias 21 (1):19-26.
Waterhouse KE, Hofmo PO, Tverdal A and Miller Jr. RR. 2006. Within and between breed differences in freezing tolerance and plasma membrane fatty acid composition of boar sperm. Reproduction 131 (5): 887-894. doi: 10.1530/rep.1.01049
How to Cite
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Download Citation
CrossRef Cited-by
1. Leyland Fraser, Karolina Wasilewska-Sakowska, Łukasz Zasiadczyk, Elżbieta Piątkowska, Krzysztof Karpiesiuk. (2021). Fractionated Seminal Plasma of Boar Ejaculates Analyzed by LC–MS/MS: Its Effects on Post-Thaw Semen Quality. Genes, 12(10), p.1574. https://doi.org/10.3390/genes12101574.
2. Francisco Javier Henao. (2019). Animal Genetics - Approaches and Limitations. https://doi.org/10.5772/intechopen.75906.
3. Julian Valencia, Marc Yeste, Armando Quintero-Moreno, Carolina del Pilar Niño-Cardenas, Francisco Javier Henao. (2020). Relative content of Niemann-Pick C2 protein (NPC2) in seminal plasma, but not that of spermadhesin AQN-1, is related to boar sperm cryotolerance. Theriogenology, 145, p.181. https://doi.org/10.1016/j.theriogenology.2019.10.023.
4. Samara Sandy Jerônimo Moreira, Arthur Emannuel de Araújo Lago, Arlindo Alencar Araripe Moura, Alexandre Rodrigues Silva. (2022). Impact of Seminal Plasma Composition on Sperm Freezability in Wild Mammals: A Review. Biopreservation and Biobanking, 20(1), p.90. https://doi.org/10.1089/bio.2021.0026.
Dimensions
PlumX
Article abstract page views
Downloads
License
Copyright (c) 2016 Revista Facultad Nacional de Agronomía Medellín

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
The journal allows the author(s) to maintain the exploitation rights (copyright) of their articles without restrictions. The author(s) accept the distribution of their articles on the web and in paper support (25 copies per issue) under open access at local, regional, and international levels. The full paper will be included and disseminated through the Portal of Journals and Institutional Repository of the Universidad Nacional de Colombia, and in all the specialized databases that the journal considers pertinent for its indexation, to provide visibility and positioning to the article. All articles must comply with Colombian and international legislation, related to copyright.
Author Commitments
The author(s) undertake to assign the rights of printing and reprinting of the material published to the journal Revista Facultad Nacional de Agronomía Medellín. Any quotation of the articles published in the journal should be made given the respective credits to the journal and its content. In case content duplication of the journal or its partial or total publication in another language, there must be written permission of the Director.
Content Responsibility
The Faculty of Agricultural Sciences and the journal are not necessarily responsible or in solidarity with the concepts issued in the published articles, whose responsibility will be entirely the author or the authors.