Biological, botanical and chemical alternatives for the control of blackberry (Rubus glaucus Benth.) diseases
Alternativas biológicas, botánicas y quimicas para el control de enfermedades en el cultivo de la mora (Rubus glaucus Benth)
DOI:
https://doi.org/10.15446/rfna.v70n2.64521Keywords:
Fruit trees, Phytopathology, Antagonistic fungi, Biocontrollers, Integrated management, Organic management (en)Frutales, Fitopatología, Hongos antagónicos, Biocontroladores, Manejo integrado, Manejo orgánico (es)
Burkholderia cepacia (Botrycid), 8: extract of Swinglea glutinosa (Ecoswin), 9: traditional farming treatments (Mancozeb, Propamocarb), 10: chemical products applications (Mancozeb, Mandipropamida, Carbendazim, Propamocar and Metalaxil+Mancozeb) alternated according to the impact of the disease, 11: chemical products applications alternated with organic products according to the suppliers recommendations and presence of the diseases, 12: absolute control, no treatment was applied to the plants. The applications were carried out every 15 days, each plant was an experimental unit and each treatment was made of five experimental units. 12 treatments were made through a RCBD (randomized complete block design) with three repetitions for a total of 15 experimental units per treatment. The assessments were performed every 8 days, and the variables were: number of healthy and sick fruits/treatment, costs/treatment and gross profit. A variance analysis and a Tukey test 5% were made. The best treatments were T11 (rotation of chemicals with biological products), T10 (rotation of chemical products according to the impact of the disease) and T3 (T. koningiopsis); considering the obtained performance/treatment, treatment cost and profit.
Para el manejo de las principales enfermedades de la mora (Rubus glaucus Benth.), se realizó una investigación donde se aplicaron 12 tratamientos a plantas ecotipo San Antonio, procedentes de cultivo in vitro. Los tratamientos fueron 1: Trichoderma harzianum+Trichoderma koningii (Hongos del Trópico), 2: Trichoderma sp. (Bioprotección), 3: Trichoderma koningiopsis (Th003 Trichoderma), 4: Trichoderma asperellum (Th034 Trichoderma), 5: Trichoderma asperellum (T-30 Trichoderma), 6: Trichoderma asperellum (T-98 Trichoderma), 7: Burkholderia cepacia (Botrycid), 8: Extracto de Swinglea glutinosa (Ecoswin), 9: Tratamiento tradicional del agricultor (Mancozeb, Propamocarb), 10: aplicaciones de productos químicos (Mancozeb, Mandipropamida, Carbendazim, Propamocar y Metalaxil+Mancozeb), en rotación según incidencia de las enfermedades, 11: aplicación de productos químicos en rotación con biológicos, según recomendaciones de los proveedores y presencia de las enfermedades, 12: testigo absoluto, no se aplicó ningún tratamiento a las plantas. Las aplicaciones se realizaron cada 15 días, cada planta fue una unidad experimental y cada tratamiento estuvo compuesto por cinco unidades experimentales, se realizaron 12 tratamientos en un diseño de BCA con tres repeticiones, para un total de 15 unidades experimentales por tratamiento. Las evaluaciones se realizaron cada ocho días y las variables consideradas fueron: número de frutos sanos y enfermos/tratamiento, costos/tratamiento y ganancia bruta. Se realizó un análisis de varianza y una prueba de Tukey 5%. Los mejores tratamientos fueron el T11 (rotación de productos químicos con biológicos), el tratamiento T10 (rotación de productos químicos según la incidencia de las enfermedades) y el tratamiento T3 (T. koningiopsis); considerando los rendimientos obtenidos/tratamiento, el costo del tratamiento y la ganancia alcanzada.
Recibido: 31 de mayo de 2016; Aceptado: 21 de diciembre de 2016
RESUMEN
Para el manejo de las principales enfermedades de la mora (Rubus glaucus Benth.), se realizó una investigación donde se aplicaron 12 tratamientos a plantas ecotipo San Antonio, procedentes de cultivo in vitro. Los tratamientos fueron 1: Trichoderma harzianum+Trichoderma koningii (Hongos del Trópico), 2: Trichoderma sp. (Bioprotección), 3: Trichoderma koningiopsis (Th003 Trichoderma), 4: Trichoderma asperellum (Th034 Trichoderma), 5: Trichoderma asperellum (T-30 Trichoderma), 6: Trichoderma asperellum (T-98 Trichoderma), 7: Burkholderia cepacia (Botrycid), 8: Extracto de Swinglea glutinosa (Ecoswin), 9: Tratamiento tradicional del agricultor (Mancozeb, Propamocarb), 10: aplicaciones de productos químicos (Mancozeb, Mandipropamida, Carbendazim, Propamocar y Metalaxil+Mancozeb), en rotación según incidencia de las enfermedades, 11: Aplicación de productos químicos en rotación con biológicos, según recomendaciones de los proveedores y presencia de las enfermedades, 12: Testigo absoluto, no se aplicó ningún tratamiento a las plantas. Las aplicaciones se realizaron cada 15 días, cada planta fue una unidad experimental y cada tratamiento estuvo compuesto por cinco unidades experimentales, se realizaron 12 tratamientos en un diseño de BCA con tres repeticiones, para un total de 15 unidades experimentales por tratamiento. Las evaluaciones se realizaron cada ocho días y las variables consideradas fueron: número de frutos sanos y enfermos/tratamiento, costos/tratamiento y ganancia bruta. Se realizó un análisis de varianza y una prueba de Tukey 5%. Los mejores tratamientos fueron el T11 (rotación de productos químicos con biológicos), el tratamiento T10 (rotación de productos químicos según la incidencia de las enfermedades) y el tratamiento T3 (T. koningiopsis); considerando los rendimientos obtenidos/tratamiento, el costo del tratamiento y la ganancia alcanzada.
Palabras clave:
Frutales, Fitopatología, Hongos antagónicos, Biocontroladores, Manejo integrado, Manejo orgánico.ABSTRACT
In order to control the main diseases that affect blackberries (Rubus glaucus Benth.), a research in which 12 treatments to San Antonio ecotype plants originated in vitro was conducted. These treatments were: 1: Trichoderma harzianum +Trichoderma koningii (Tropical Fungus), 2: Trichoderma sp. (Bioprotection), 3: Trichoderma koningiopsis (Th003 Trichoderma), 4: Trichoderma asperellum (Th034 Trichoderma), 5: Trichoderma asperellum (T-30 Trichoderma), 6: Trichoderma asperellum (T-98 Trichoderma), 7) Burkholderia cepacia (Botrycid), 8: Extract of Swinglea glutinosa (Ecoswin), 9: Traditional farming treatments (Mancozeb, Propamocarb), 10: Chemical products applications (Mancozeb, Mandipropamida, Carbendazim, Propamocar and Metalaxil+Mancozeb) alternated according to the impact of the disease, 11: Chemical products applications alternated with organic products according to the suppliers recommendations and presence of the diseases, 12: Absolute control, no treatment was applied to the plants. The applications were carried out every 15 days, each plant was an experimental unit and each treatment was made of five experimental units. 12 treatments were made through a RCBD (randomized complete block design) with three repetitions for a total of 15 experimental units per treatment. The assessments were performed every 8 days, and the variables were: number of healthy and sick fruits/treatment, costs/treatment and gross profit. A variance analysis and a Tukey test 5% were made. The best treatments were T11 (rotation of chemicals with biological products), T10 (rotation of chemical products according to the impact of the disease) and T3 (T. koningiopsis); considering the obtained performance/treatment, treatment cost and profit.
Keywords:
Fruit trees, Phytopathology, Antagonistic fungi, Biocontrollers, Integrated management, Organic management.Mora de Castilla (Rubus glaucus Benth.) is affected by diverse diseases mostly caused by fungi, amongst these diseases there is the Anthracnose, which is caused by Glomerella cingulate (Stoneman) Spauld. and Schrenk. (anamorph Colletotrichum gloeosporioides (Penz.) Penz. and Sacc.), Colletotrichum acutatum Simmonds., Colletotrichum boninense Moriw., Sato and Tsukib.; these pathogens affect stems, petioles and reproductive structures causing lesions that can end in the death of the plant's branches affecting the crop production (Saldarriaga et al., 2008; Saldarriaga, 2006; Afanador et al., 2009; Tamayo, 2009; Rueda, 2010). The Fungus that causes the Anthracnose can also damage flower buds, tender shoots, stalks and fruits (Franco and Giraldo, 2000). When the fungus infects the plant's side branches, this creates a deformation on the group of flowers, the tender shoots blacken, and the branches die in a descendent and progressive way (Castro and Díaz, 2001). In the inflorescences, this infection appears as a necrosis of the tissues in which the fungus sporulates (Rueda, 2010). In ripen fruits, the disease appears sporadically. In this case, the affected fruits show dehydration, necrosis and wet rot in the presence of some fungal structure (Saldarriaga and Bernal, 2000; Botero et al., 2002). This disease deteriorates between 50 and 70% of the cultivated blackberry plants' stems and causes a loss of a 5% of the pathogens-affected fruits (Tamayo, 2009). The incidence of this disease has been registered in a 52% under the conditions given by the blackberry crops in the Colombian coffee belt region (Botero et al., 2002).
The gray mold caused by Botrytis cinerea Pers, Ex. Fr., is another important disease that appears during the stage of production and postharvest of the blackberry. This fungus affects the flower buds since their opening and appears in the fruition and ripening, causing necrosis and mummification of the fruits (Forero de La-Rotta, 2007) (Quinatoa, 2015). In the presence of humidity, the affected fruits show a growth in the fungus with a velvety aspect of colors gray, brown or olive green. The fruits dry and mummify stuck to the cluster (Saldarriaga and Bernal, 2000; Tamayo, 2009). The fungus can also affect leaves, flowers and stalks (Franco and Giraldo, 2000). This disease has created losses between 50 to 76% of the harvested fruit affected by pathogens (Tamayo and Peláez, 2000).
The downy mildew caused by Peronospora Corda affects the stems, stalks, flower buds, flowers and fruits. The sick stems and stalks show purple discolorations with whitish lesions on which a downiness of light gray color grows (Saldarriaga and Bernal, 2000; Tamayo, 2003). The flowers show yellowing then their petals get dry to fall later on. The sepals show a light brown to black color lesion which advances until the flower buds mummification (Tamayo, 2009). In fruits, the fungus causes an irregular development, uneven ripening, and loss of fruit brightness, which depreciates the fruit in its commercialization. The green fruits stop growing (Saldarriaga and Bernal, 2000; Franco and Giraldo, 2000). This disease can cause losses of a 20 to 30% of the harvested fruits (Tamayo and Peláez, 2000).
Another disease that can affect the blackberry is the powdery mildew. This is caused by the fungus Oidium Link. This infection appears mainly in young leaves through deformation (leaf curling), it is associated to the presence of chlorotic, irregular and diffused areas that can be seen on the surface of the foliar laminae. Occasionally, the leaves are covered on a fine white dust that corresponds to the growth and sporulation of the fungus. The yellow rust is also an important disease; this one is caused by the fungus Gerwasia lagerheimii (Magnus) Buritica, it appears in the form of pustules of orange color in the stems, stalks, the reverse of leaves and in the fruits (Franco and Giraldo, 2000; Tamayo, 2009).
There are reports of foliar conditions caused by the fungi Septoria Sacc, Phyllosticta Pers, Alternaria Nees, which importance is still secondary due to the low incidence of the diseases caused by them (Tamayo, 2009). The stems and roots of the blackberry also present some diseases caused by fungi such as: Coniothyrium fuckelii Sacc, Verticillium albo-atrum Reinke and Berth, Rosellinia De Not, Fusarium roseum Link: Fr. and Fusarium oxysporum Schlechtend. Fr, which importance has not been established yet (Tamayo, 2009).
Traditionally the treatment of the diseases of the Mora de Castilla has been based on the development of some cultural practices and the periodic application of different fungicides -there is no official record of most of them being used for the blackberry-. The current tendencies in food production are oriented towards getting products with less pesticide charges, besides the demands on environmental conservation and food harmlessness. Nowadays, in the field of blackberry harvesting, the use of fungicides is starting to be restricted or forbidden, such as the products based on Clorotalonil, Mancozeb, Propineb, Metalaxil + Mancozeb, Propineb + Cymoxanil (Arroyave and Salazar, sf., quoted by Díaz, 2009). Research institutions and some companies in the commercial sector are developing products of botanical origins such as vegetables extracts, and biological origins such as antagonistic microorganisms with the potential use of the control of the diseases caused by fungi of the genus Colletotrichum, Botrytis, Peronospora, Oidium (Restrepo et al., 2001; Agrobiológicos Safer and Natural Control Sf, Vélez and Estrada sf, Bioprotección sf, Ecoflora, 2007; Velosa et al., 2002; 2003, Castro and Díaz, 2001).
This situation has created the need for more research on new products and management strategies that allow the offering to the blackberry producers of more efficient and sustainable alternatives to the integral management of the main diseases that affect this crop, contributing to the obtaining of a product harmless to the consumers. With the results from this research, some key knowledge is provided to the creation of blackberry disease control proposals, considering among others, the efficiency of the products and the economic sustainability to satisfy the demands of the food and agriculture chain of the blackberry.
MATERIALS AND METHODS
The in-field observations were carried out in a period of 20 months in Corpoica R.C. La Selva, Rionegro, Antioquia, under conditions of a Lower Montane moist forest (LM-mf). 12 treatments were applied to blackberry plants of the San Antonio ecotype, originated in vitro. The evaluated treatments were the following: T1: Trichoderma harzianum+Trichoderma koningii (commercial product of Hongos del Trópico), T2: Trichoderma sp. (comercial product of Bioprotección), T3: Trichoderma koningiopsis (Th003 Trichoderma, product of CORPOICA), T4: Trichoderma asperellum (Th034 Trichoderma, product of CORPOICA ), T5:Trichoderma asperellum (T-30 Trichoderma, product of the Corporación para Investigaciones Biológicas - CIB, in scaling process), T6: Trichoderma asperellum (T-98 Trichoderma, product of the CIB, in scaling process), T7: Burkholderia cepacia (Botrycid, commercial product of Safer), T8: extract of Swinglea glutinosa (EcoSwing® commercial product of Ecoflora), T9: corresponds to the traditional treatments of the farmer based on the applications of chemical products with active ingredients such as (Mancozeb) and (Propamocarb), T10: Application of chemical products (the products with the following active ingredients were applied: (Mancozeb), (Mandipropamida), (Carbendazim), (Propamocarb), (Metalaxil+Mancozeb) alternating according the effect and incidence of the blackberry diseases, T11: Application of chemical products alternating with biological products (bio-control microorganisms and vegetal extracts) according to the recommendations of the suppliers and the phenological state of the plants; the following products were applied: extract of Swinglea glutinosa, Burkholderia cepacia, Trichoderma koningiopsis (Th003), Trichoderma asperellum (T-98), Trichoderma sp., Propamocarb, Metalaxil +Mancozeb, Mandipropamida, taking into account the presence and incidence of the diseases, T12: Absolute control, no treatment was applied to the plants. The 12 treatments were distributed in three completely randomized blocks, each plant was an experimental unit and each treatment was made of five experimental units.
The plants that underwent the treatment were surrounded by blackberry plant sources of inoculum, for a total of 270 plants sown with a distance of 2 m x 2 m between plants and 3 m x 3 m between furrows, in an area of 1530 m2 (Díaz, 2009; Hincapié, 2010). Previous to the crops' establishment, a chemical analysis of the soil was made which was used as a base to the fertilization process. The cultural labors proper to the crops were made such as the plateo (keeping the young plants clear of weeds), weed control, pruning, wiring on a modified double T trellis system, maintenance pruning (leaving 5 stems per plant), sanitary pruning, and destruction of the pruning remainders. The plot was monitored on a weekly basis and the application of the treatments started once a 50% of the plants were blossomed. These applications were carried out every 15 days, using the doses recommended by the products manufacturers and traders. The evaluations were carried out every eight days, by collecting and separating the healthy fruits from the sick on each type of treatment in order to be quantified in the laboratory later on through the processes of counting, weighing, classification and registry of the fruits affected by the diseases. The following were taken into consideration: the amount of healthy fruits and their weight, the number of fruits affected by the gray mold (Botrytis cinerea), downy mildew or peronospora (Peronospora sp.), and the anthracnose (Colletotrichum sp.). For each of the treatments the performance per treatment (kg ha-1) was estimated. For the mentioned variables, an analysis of variance and the medians separation according to the Tukey test on a 5% level were conducted. Besides, the cost of the different treatments was calculated and the gross profit was estimated per hectare sown (1666 plants). The experiment was suspended once the plants reached the 11 month of the productive stage (the peak or production under the agro-ecological conditions where the plot was established is reached 18 months after the beginning of the productive stage) (Díaz, 2009; Hincapié, 2010).
RESULTS AND DISCUSSION
The results obtained through this research are the following: on the Absolute Control treatment (T12), the treatments that showed a higher percentage of healthy fruits, with no significant statistical differences, by the gray mold, downy mildew and Anthracnose diseases (Figure 1) were: T11, T10, T3 and T9 (Table 1). For this group of treatments, the percentage of sick fruits was between 35 and 39%; whereas the Absolute Control group presented an 81% of sick fruits. In this same group of treatments, the best results for the gray mold management were achieved with the use of T3 and T11 (with percentages between the 2.2 and 2.5%) in contrast to the Control group that presented a 15%. For the downy mildew the percentage of sick fruits was between the 32.3 and 36%, being the best treatments the T10, T11 and T9 in contrast to the Control group which presented a 64.2% (Table 1). Regarding the Anthracnose, the percentage of affected fruits was less than 2.1%. Treatment T8 did not show any difference from the Absolute Control T12, but both presented differences from the rest of treatments (Table 1).
Figure 1: A. Blackberry fruits affected by gray mold (Botrytis cinerea). B. Blackberry fruits affected by downy mildew (Peronospora sp). C. Blackberry fruits affected by Anthracnose (Colletotrichum sp).
Table 1: Behavior of the different treatments according to the percentage of healthy and sick fruits.

In addition to this, after analyzing all treatments, and independently the behavior of the diseases, the following was observed: for the gray mold the best treatments were the T1, T3 and T11 which presented the lowest percentage of fruits affected by the disease (2.2% to 2.5%) (Table 1); to this group the treatments that followed are T7, T5, T10, T4 and T8 with percentages between the 3 and 4% of the fruits affected by the disease with no significant differences between them. Treatments T6, T9 and T12 were the less efficient. Treatment T12 showed significant differences from all treatments and reached the highest percentage of fruits affected by Botrytis cinerea, corresponding to a 15%. Figure 1A illustrated the symptoms of the fruit affected by gray mold, these are consistent to a soft rot in which appears a growth of grayish color that corresponds to the sporulation of the fungus.
Regarding the downy mildew, this was the disease that showed the high percentages of affected fruits (32% to 64%). With treatments T10, T11, T9, the lowest percentages of sick fruits were obtained, around 32% (Table 1). For the other treatments the percentage of sick fruits was between the 36 and 52.7%. Some significant differences were established with the Absolute Control group, which reach a 64% of fruits with symptoms of the disease. This disease manifests with bad filled drupes, uneven ripening, irregular development (Figure 1B), and the fruits harden when touched.
Regarding the Anthracnose, this disease showed the lower percentage of affected fruits (less than 2.1%) Other studies have reported losses by Anthracnose estimated in approximately a 5% of the fruits affected by different diseases (Tamayo, 2009). In the fruits the disease appears sporadically and the symptoms correspond to drupes dehydration, necrosis (Figure 1C), and wet rot with presence of fungal structures (Saldarriaga and Bernal, 2000).
During this study, besides the behavior of the diseases in front of the different treatments, the production (kg of healthy fruits per treatment ha-1), the production costs (assuming $2000 COP as the sale value of a kilo of blackberry), and the cost of the treatment were considered. Table 2 registers the respective values and costs for each of the treatments.
Table 2: Costs and gross profit for every treatment.
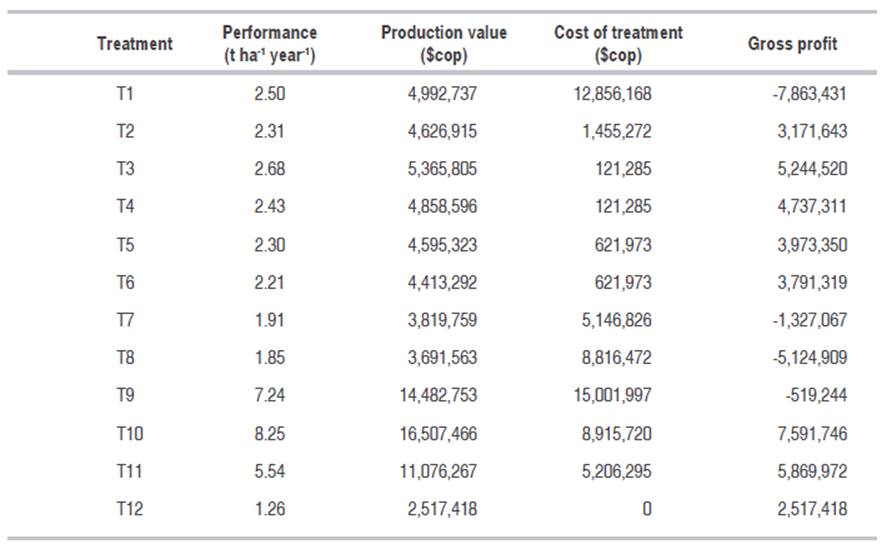
The highest production (t ha-1 per year) was reached on treatments T10, T9 and T11; however, when the costs analysis was made, it showed that the value of consumables for some treatments was considerable and affected negatively the profit; leading in some moments to produce losses, like can be seen on treatment T9.
T11 (chemical products alternating with biological products and vegetal extract) was considered as a promising treatment due to the gross profit and the possibility of reducing the charge of chemical products and the addition of biological products, which can contribute to the environmental conservation and the obtaining of a harmless product. In order to apply this treatment it's important to monitor the diseases regarding their happening and severity to define the type of product that will be used, besides the weather conditions at the moment of application have to be considered. Treatment T10 (chemical products rotation according to the happening of diseases) allowed to obtain the highest production and gross profit (Table 2), and a most rational management of the diseases but it needs the implementation of a monitoring component of the diseases as a selection criteria of the products to apply. Treatment T3 (Th003 - T. koningiopsis) can be an alternative to contribute to the integrated management especially in crops with a high incidence of gray mold. Currently, the laboratory of biological control of Corpoica has a wet powder (WP) product made from Trichoderma koningiopsis Th003 named “Tricotec”. This bioinsecticide has demonstrated excellent results in vegetable crops for the pathogens control such as Rhizoctonia solani, getting a reduction of the disease of a 71% in Chonto tomato (Gómez et al., 2011; Gómez, 2012). It has also shown a high efficacy in the control of Botrytis cinerea and Oidium lycopersicum in tomato by being applied to the phyllosphere; nevertheless, studies on the valuation of technology indicated a high economic feasibility for the soil application and the foliar application (Gómez et al., 2011). According to Gómez (2012), in the trials made on lettuce with the bioinsecticide (WP) T. koningiopsis Th003 syptoms of the pathogen Botrytis spp. did not appear.
CONCLUSIONS
The best results were obtained through the following treatments: T11 (use of chemical products alternating with biological products -biocontrollers and botanic extracts-) followed by treatments T10 (chemical rotation of products according to the happening of diseases), and the treatment T3 made of Trichoderma koningiopsis (Th003); all of this considering the obtained performance/treatment, the cost of each treatment and the gross profit among others.
The treatments evaluated under experimental conditions were inefficient for the control of the downy mildew (Peronospora sp.), getting better results on the control of the gray mold and the Anthracnose of the fruit when said treatments are applied combining the opportune and adequate implementation of the cultural labors (sanitary and ventilation pruning, harvest and destruction of sick fruits, weed control, fertilization and management of pruning residues).
The products' evaluation on different grades of scaling does not allow to make the best comparison due to the possibility of having given more advantages to certain products in relation to other products that were on inferior stages of manufacture.
The low performance of the crops are explained considering that experimentally the work was carried out with only five stems per plant, and besides, the crops corresponded to the beginning of the plants' productive stage and did not reach the peak of the harvest.
REFERENCES
References
Andrade C. 2012. Evaluación del efecto de la aplicación de Trichoderma harzianum y Trichoderma viride para el control de marchitez en mora de castilla (Rubus glaucus Benth) en el cantón Pillaro, provincia de Tungurahua. Escuela Superior Politécnica de Chimborazo. Riobamba, Ecuador. 115 p.
Afanador L, Álvarez E, González A and Mejía J. 2009. Especies de Colletotrichum causantes de la Antracnosis de mora de Castilla (Rubus glaucus Benth.) in Colombia. Memorias de 29 Congreso Internacional de Fitopatología y Ciencias Afines: Patología vegetal, pasado, presente y futuro. Medellín, 107 p.
Agrobiológicos Safer and Natural Control. n.d. Technical Datasheets. Medellín, 36 p.
Arroyave G, Salazar M. n.d. Guía técnica del cultivo de mora. Manizales, Litomaster, 15 p.
Beltrán C, Sastoque L, Villamizar L, Gómez M, Grijalba E, García M, Díaz A, Smith A, Moreno C, Bosa F, Jaramillo S. and Ángel J. 2011. Trichoderma en el control biológico de la Rhizoctoniasis. Corporación colombiana de investigación agropecuaria – Corpoica CBB. Mosquera, Cundinamarca. 13 p.
BIOPROTECCION n.d. Bioprotección Beauveria bassiana, Metharhizium anisopliae, Lecanicillium lacanii, Paecilomyces lilacinus, Trichoderna sp. Especificidad, patogenicidad, permanencia, inocuidad ambiental. Plegable promocional. Manizales.
Botero MJ, Rios G, Franco G, Romero M, Pérez JC, Morales JE, Gallego JL and Echeverri DI. 2002. Identificación y espacialización de enfermedades asociadas a los cultivos de mora (Rubus glaucus Benth.), en el eje cafetero. IV Seminario Frutales de Clima Frío Moderado. Medellín, pp. 87-92. Corpoica, CDTF, UPB.
Boyzo J, Silva HV and Rebollar A. 2015. Biorational treatments to manage dryberry of blackberry caused by Peronospora sparsa. Crop Protection. pp. 121 -126. doi: 10.1016/j.cropro.2015.03.023
Castro D and Díaz J. 2001. Alternativas para el manejo integrado del cultivo de la mora (Rubus glaucus B.). Rionegro (Antioquia), Colombia: Centro de Publicaciones, Universidad Católica de Oriente, pp. 7-44.
Diaz C. 2009. Tecnologías que contribuyan a la implementación de las BPA en el cultivo de la mora (Rubus glaucus Benth.) en Antioquia y Caldas. Project’s final report. CORPOICA, C.I La Selva, Rionegro, Antioquia. pp. 188- 205.
ECOFLORA Ltda. 2007. Technical datasheets EcoSwing®. info@ecoflora.com.
Forero de La-Rota MC. 2007. Uchuva (Physalis peruviana L.) y mora de Castilla (Rubus glaucus Benth.): enfermedades y manejo. Memorias del XXVIII Congreso Asociación Colombiana de Fitopatología y Ciencias Afines. Palmira, pp. 1-7.
Franco G and Giraldo M. 2000. El cultivo de la mora. Manizales, Corporación Colombiana de Investigación Agropecuaria (Corpoica), pp. 53, 43-55.
Gómez M, Moreno C, Cotes A, Smith A, Villamizar L, Díaz A, García M, Cruz F and Molina J. 2011. Uso de Trichoderma koningiopsis Th003 para el control de fitopatógenos en hortalizas. Corporación colombiana de investigación agropecuaria – Corpoica. Bogotá D.C. 65 p.
Gómez M. 2012. Desarrollo de protocolos de eficacia para el registro de un bioplaguicida a base de Trichoderma koningiopsis en cultivos de tomate y lechuga y de un bioplaguicida a base de Lecanicillium lecanii en berenjena. Corporación Colombiana de Investigación Agropecuaria (Corpoica). Centro de Biotecnología y Bioindustria CBB. Informe técnico final. Mosquera Cundinamarca. 79 p.
Hincapié O. 2010. Evaluación de alternativas para el manejo integrado de enfermedades en el cultivo de mora (Rubus glaucus Benth.) en Rionegro, Antioquia. Agricultural Engineering thesis. Politécnico Colombiano Jaime Isaza Cadavid, Medellín (Antioquia). 76 p.
Quinatoa N. 2015. Evaluación del control de botrytis (Botrytis cinerea) en el cultivo de mora (Rubus glaucus Benth) mediante el uso de Trichoderma yemas en la comunidad de Misquillí de la parroquia Santa Rosa, provincia de Tungurahua. Universidad Técnica de Ambato. Facultad de Ciencias Agropecuarias. Ambato Ecuador. 83 p.
Restrepo V, Hoyos L, Agudelo P and Ordúz S. 2001. Antagonismo in Vitro de aislamientos de Trichoderma y Gliocladium contra Fusarium oxysporum, Botrytis cienera y Colletotrichum gloeosporioides. XXII Congreso Asociación Colombiana de Fitopatología y Ciencias afines. Medellín, 322 p.
Rueda K. 2010. Caracterización molecular de especies de Colletotrichum prevalentes en las zonas productoras de mora de Castilla (Rubus glaucus Benth.) en Antioquia. Master’s thesis. Universidad Nacional de Colombia, Medellín, Colombia. 144 p.
Sapieha-Waszkiewicz A, Marjańska-Cichoń B and Miętkiewski R. 2011. Effect of Biopesticides on the Growth and Development of Isolates of Botrytis cinerea Pers., in vitro Obtained from Raspberry Plants. Journal Article published 1 Jan 2011 in Journal of Plant Protection Research volume 51(2): 151-156. doi: 10.2478/v10045-011-0026-8
Saldarriaga-Cardona A, Castaño-Zapata J and Arango R. 2008. Caracterización del agente causante de la Antracnosis en tomate de árbol, manzano y mora. Rev. Academia Colombiana de Ciencias Exactas, Físicas y Naturales 32(123): 145-156.
Saldarriaga A. 2006. Caracterización del agente causante de la Antracnosis en tomate de árbol, manzana y mora. Manizales. 191p. Phytopathology Master’s thesis. Universidad de Caldas, Facultad de Ciencias Agropecuarias.
Saldarriaga A and Bernal J. 2000. Enfermedades asociadas a los cultivos de mora (Rubus glaucus Benth.) en el departamento de Antioquia. p. 132-135. In: Tercer Seminario Frutales de Clima Frío Moderado (2000: Manizales. Colombia). Memorias. Manizales: CDTF.
Tamayo P and Peláez A. 2000. Caracterización de daños y pérdidas causadas por enfermedades del fruto de la mora de castilla (Rubus glaucus Benth.) en Antioquia. In: Seminario Frutales de Clima Frío Moderado (2000: Manizales). Memorias del Tercer Seminario de Frutales de Clima Frío Moderado. Manizales: Litoas, 2000. pp. 174-178.
Tamayo P. 2003. Principales enfermedades del tomate de árbol, la mora y el lulo en Colombia. Technical bulletin 20. Rionegro, Antioquia, Corpoica. pp. 16-25.
Tamayo P. 2009. Enfermedades. In: Seminario de Actualización Tecnológica: Cultivo, Agroindustria y comercialización de la mora de Castilla. (2009: Rionegro (Antioquia), Colombia). Memorias CD. Rionegro: Corpoica.
Velosa M, Ramírez S, Miño J, Uricoechea J, Ramírez J and González C. 2003. Manejo biológico de la pudrición del fruto (Botrytis cinerea Pers. Ex. Fr.), en la mora (Rubus glaucus Benth) en condiciones de laboratorio. p. 3. In: XXIV Congreso Asociación Colombiana de Fitopatología y Ciencias afines. ASCOLFI Palmira.
Velosa M, Ramírez S, Miño J, Uricoechea J, Ramírez J and González C. 2002. Manejo biológico mediante el uso de extractos de plantas de la pudrición del fruto (Botrytis cinerea Pers. Ex. Fr.), en la mora (Rubus glaucus Benth) en condiciones de laboratorio. p. 118- 125. In: IV Seminario Frutales de Clima Frio Moderado (2002: Medellín). Memorias. Medellín: CORPOICA, CDTF, UPB.
Información general de Burkholderia Cepacia 2009. In: Wikipedia, http://es.wikipedia.org/wiki/Burkholderia_cepacia; accessed: November 2009.
Información general de Botrycid 2014. In: Natural Control, http://www.naturalcontrol.com.co/site/index.php/bioinsumos/botrycid: Agosto 2014.
Información general de Agrobiologicos Safer. 2009. In: http://www.agrobiologicossafer.com: Marzo 2016.
How to Cite
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Download Citation
CrossRef Cited-by
1. Yessica Camargo Piñeres, Gisela Zambrano Montenegro, Mailen Ortega Cuadros, Deivis Jhoan Gutierrez Montero, Jayr Alfredo Yepes Escorcia. (2021). Actividad antifúngica in vitro del aceite esencial de Swinglea glutinosa Merr sobre Colletotrichum sp. patógeno de mango (Mangifera indica L.).. Revista Colombiana de Biotecnología, 23(1), p.62. https://doi.org/10.15446/rev.colomb.biote.v23n1.88025.
2. Alessandrina Ascencio-Arteaga, Silvia Luna-Suárez, Jeanette G. Cárdenas-Valdovinos, Ernesto Oregel-Zamudio, Guadalupe Oyoque-Salcedo, José A. Ceja-Díaz, María V. Angoa-Pérez, Hortencia G. Mena-Violante. (2022). Shelf Life of Blackberry Fruits (Rubus fruticosus) with Edible Coatings Based on Candelilla Wax and Guar Gum. Horticulturae, 8(7), p.574. https://doi.org/10.3390/horticulturae8070574.
3. Luis Gabriel Bautista-Montealegre, Leidy Yibeth Deantonio-Florido, William Andrés Cardona, Martha Marina Bolaños-Benavides, Gerhard Fischer. (2022). Mineral nutrition and tolerance to Colletotrichum spp. of Andean blackberry (Rubus glaucus Benth.) in nursery. Agronomía Mesoamericana, , p.48655. https://doi.org/10.15517/am.v33i3.48655.
4. Germán Franco, Jorge Alonso Bernal Estrada, Luz Adriana Váquez Gallo, Alegría Saldarriaga Cardona, Juan Camilo Henao Rojas, John Díaz Montaño, Ana Elizabeth Díaz Montilla, Germán Andrés Aguilera Arango, María Cristina García Muñoz, Jorge Humberto Argüelles Cárdenas, Mónica Betancourt Vásquez, Érika Patricia Sánchez Betancourt, Yimmy Alexander Zapata Narváez, Camilo Rubén Beltrán Acosta, Alba Marina Cotes Prado, Germán Alonso Antía Londoño, Carlos Eduardo Orrego Alzate, Natalia Salgado Alzate, Lady Joana Rodríguez, Yeimy Alejandra Rodríguez Restrepo, Álvaro de Jesús Tamayo Vélez, Cipriano Arturo Díaz Díez, Pablo Julián Tamayo Molano. (2020). Tecnología para el cultivo de la mora (Rubus glaucus Benth).. https://doi.org/10.21930/agrosavia.manual.7403251.
5. Nathalia Cardona Hurtado, Gloria Edith Guerrero Álvarez, Ana María López Gutiérrez. (2019). Identificación de Peronospora sparsa y evaluación del contenido de fenoles en frutos de mora de castilla afectados por este microorganismo. Revista Ceres, 66(1), p.11. https://doi.org/10.1590/0034-737x201966010002.
6. Karen Johana Ortega Villalba, Carlos Velez-Pasos, Pablo E. Rodriguez-Fonseca, Fabrice Vaillant-Barka. (2023). Unleashing the potential of flash vacuum expansion: an innovative approach for Andean Blackberry (Rubus glaucus Benth) Processing. Ingeniería y Competitividad, 25(4) https://doi.org/10.25100/iyc.v25i4.13132.
7. W. Viera, M. Noboa, A. Martínez, R. Jácome, L. Medina, T. Jackson. (2020). Trichoderma application increases yield and individual fruit weight of blackberries grown by small farmers in Ecuador. Acta Horticulturae, (1277), p.287. https://doi.org/10.17660/ActaHortic.2020.1277.42.
8. William Andrés Cardona. (2019). Manual de nutrición del cultivo de mora de Castilla (Rubus glaucus Benth.) bajo un esquema de buenas prácticas en fertilización integrada. https://doi.org/10.21930/agrosavia.manual-18.
9. Nathalia Cardona-Hurtado, Gloria E Guerrero-Alvarez, Ana M López-Gutiérrez. (2020). Changes in phenolic compounds and antioxidant capacity of Andean raspberries in response to Peronospora sparsa. Universitas Scientiarum, 25(2), p.299. https://doi.org/10.11144/Javeriana.SC25-2.cipc.
Dimensions
PlumX
Article abstract page views
Downloads
License
Copyright (c) 2017 Revista Facultad Nacional de Agronomia

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
The journal allows the author(s) to maintain the exploitation rights (copyright) of their articles without restrictions. The author(s) accept the distribution of their articles on the web and in paper support (25 copies per issue) under open access at local, regional, and international levels. The full paper will be included and disseminated through the Portal of Journals and Institutional Repository of the Universidad Nacional de Colombia, and in all the specialized databases that the journal considers pertinent for its indexation, to provide visibility and positioning to the article. All articles must comply with Colombian and international legislation, related to copyright.
Author Commitments
The author(s) undertake to assign the rights of printing and reprinting of the material published to the journal Revista Facultad Nacional de Agronomía Medellín. Any quotation of the articles published in the journal should be made given the respective credits to the journal and its content. In case content duplication of the journal or its partial or total publication in another language, there must be written permission of the Director.
Content Responsibility
The Faculty of Agricultural Sciences and the journal are not necessarily responsible or in solidarity with the concepts issued in the published articles, whose responsibility will be entirely the author or the authors.























