In vitro study on the nematicidal effect of different plant extracts on Pratylenchus penetrans and Meloidogyne chitwoodi
Estudio in vitro sobre el efecto nematicida de diferentes extractos de plantas en Pratylenchus penetrans y Meloidogyne chitwoodi
DOI:
https://doi.org/10.15446/rfnam.v72n3.76070Keywords:
Brassica spp., glucosinolates, Lolium spp., Lupinus sp. (en)Brassica spp., glucosinolatos, Lolium spp., Lupinus sp. (es)
The purpose of this in vitro study was to evaluate the nematicidal effect of different glucosinolates and other secondary metabolites extracted from several plant species on the plant-parasitic nematodes Pratylenchus penetrans and Meloidogyne chitwoodi. Glucosinolate extracts from 16 species of genera Brassica, seven Lolium species and one species of Lupinus were used to investigate their nematicidal effect in vitro. From the tested extracts, the one obtained from Brassica juncea (oriental) showed the most promising results, controlling both nematode species. Lupinus sp. also showed positive results when tested against P. penetrans.
El propósito de este estudio in vitro fue evaluar el efecto nematicida de diferentes glucosinolatos y otros metabolitos secundarios extraídos de varias especies de plantas, sobre los nemátodos Pratylenchus penetrans y Meloidogyne chitwoodi que afectan negativamente diversas plantas. Extractos de glucosinolatos provenientes de 16 especies del género Brassica, siete especies de Lolium y una especie de Lupinus fueron usados para investigar su efecto nematicida in vitro. De los extractos probados, el que proviene de Brassica juncea (oriental) mostró los resultados más promisorios para el control de las dos especies de nemátodos en estudio. Lupinus sp. también mostró resultados positivos para el control de P. penetrans.
Recibido: 9 de noviembre de 2018; Aceptado: 5 de julio de 2019
ABSTRACT
The purpose of this in vitro study was to evaluate the nematicidal effect of different glucosinolates and other secondary metabolites extracted from several plant species on the plant-parasitic nematodes Pratylenchus penetrans and Meloidogyne chitwoodi. Glucosinolate extracts from 16 species of genera Brassica, seven Lolium species and one species of Lupinus were used to investigate their nematicidal effect in vitro. From the tested extracts, the one obtained from Brassica juncea (oriental) showed the most promising results, controlling both nematode species. Lupinus sp. also showed positive results when tested against P. penetrans.
Keywords:
Brassica spp., Glucosinolates Lolium spp., Lupinus sp..RESUMEN
El propósito de este estudio in vitro fue evaluar el efecto nematicida de diferentes glucosinolatos y otros metabolitos secundarios extraídos de varias especies de plantas, sobre los nemátodos Pratylenchus penetrans y Meloidogyne chitwoodi que afectan negativamente diversas plantas. Extractos de glucosinolatos provenientes de 16 especies del género Brassica, siete especies de Lolium y una especie de Lupinus fueron usados para investigar su efecto nematicida in vitro. De los extractos probados, el que proviene de Brassica juncea (oriental) mostró los resultados más promisorios para el control de las dos especies de nemátodos en estudio. Lupinus sp. también mostró resultados positivos para el control de P. penetrans.
Palabras clave:
Brassica spp., Glucosinolatos, Lolium spp., Lupinus sp..The identification of phytochemical based strategies for the control of plant pathogens is important since it can be used in sustainable production systems where there are not many possibilities to manage plant-parasitic nematodes. It can also be essential for the development of new nematicides in traditional agriculture (Chitwood, 2002). Nowadays, further research on environmentally friendly biofumigants is suggested (Devi, 2018).
Meloidogyne chitwoodi Golden, O'Bannon, Santo, & Finley is an important pathogen of potato and other crops in the western part of Europe and is also a major pest of potato in the Northwestern states of the United States (Castagnone-Sereno et al., 1999). Pratylenchus penetrans is an obligate plant parasite of a wide range of hosts, mainly in temperate climates. It is one of the principal nematodes infesting ornamental plants and causes serious losses in different crops (Peng and Moens, 2002).
Plant compounds may act as a repellent, attractant, hatching stimulants or inhibitors and nematotoxicants. They can be used as fumigants or introduced in crop rotation programs for nematode control (Chitwood, 2002).
Glucosinolates are a group of allelochemicals that occur in all plants of the order Brassicales or Capparales (Cronquist, 1981), being the Brassicaceae family the most numerous and important group (Fahley et al., 2001). Glucosinolate (GLSs) have sulfur, and that explains its strong flavor (Avato et al., 2013). More than 130 glucosinolates have been identified (Fahley et al., 2001; Kirkegaard et al., 1999) and may be divided into three subclasses comprising aliphatic, phenyl, and indol-3-ylmethyl glucosinolates (Buskov et al., 2002). Isothiocyanates (ITC) and other plant compounds such as nitriles and thiocyanates (Buskov et al., 2002) are released from Brassicaceae when glucosinolates are hydrolyzed by the action of the enzyme myrosinase (Figure 1) (Kirkegaard et al., 1999). These hydrolysis products have shown various bioactive effects against some soilborne diseases and nematodes (Rosa et al., 1997).
Figure 1: Enzymatic hydrolysis of glucosinolates via myrosinase activity (Kirkegaard et al.,1999).
The glucosinolate content average is higher on field-grown B. juncea, B. napus, B. campestris, and Eruca sativa, compared to the greenhouse and high tunnel cultivation (Antonious et al., 2009).
Brassica species have shown a significant reduction in nematode numbers of P. neglectus (Potter et al., 1998) and P. penetrans (McFadden et al., 1992) when used as green manures. The same has been observed for M. chitwoodi (Mojtahedi et al., 1993). Other nematode species have shown mortality up to 100% when low doses of glucosinolates from Brassicaceaes have been applied, in vitro; such as in the case of Globodera rostochiensis (Serra et al., 2002), G. pallida (Lord et al., 2011) and M. incognita (Oliveira et al., 2010). There is also a biocidal property with the use of GLSs on Xiphinema index and Heterodera carotae (Avato et al., 2013). Width control spectrum has been found by using these species (Björkman et al., 2011). Other plant compounds as the ones released from Lupinus sp. and several kinds of grass have proved to have a nematicidal effect; mulching with Pennisetum purpureum has been used for the control of M. javanica (Matsumoto et al., 2002) and the application of Sudan's grass extracts has reduced M. hapla juveniles that penetrate lettuce roots (Wildmer and Abawi, 2007). Anastasiadis and Karanastasi (2011) also presented the effectivity of brassica and ryegrass soil amendments on M. incognita and javanica. The Fabaceae Medicago sativa has been found a good control against G. rostochiensis (D'Addabbo et al., 2011).
Considering the potential of these species as possible biocides, the purpose of this trial was to determine the nematicidal effects of plant extracts on plant-parasitic nematodes.
MATERIALS AND METHODS
Extracts
Brassicaceous accessions from a local field were tested: B. fruticulosa subsp. mauritanica, B. fruticulosa, B. tournefortii, B. tournefortii, Sinapis arvensis subsp. arvensis, B. carinata, Raphanus sativus, R. sativus, B. juncea, B. juncea (oriental), B. tournefortii, B. oxyrrhina, B. napus subsp. oleifera, Sinapis alba, Crambe abbyssinica and Crambe hispanica (identified as extract: 1, 2, 3, 4, 5, 7, 8, 9, 10, 11, 13, 15, 17, 18, 20 and 21, respectively, showed in Figure 2). The extracts of Brassicaceae and one extract of Lupinus sp. were obtained with a blend of 3 g of fresh shoot material in 6 mL of phosphate buffer (25 mM, pH 7, with 15 mg L-1 streptomycin), later sieved, centrifuged and used directly to make dilutions. Seven grasses, belonging to Lolium spp., were collected from dried meadows, and these samples were roasted for 3 hours from 160 to 260 °C before extracting the active components. Grasses were identified by letters (A, B, C, D, E, F, and G), as shown in Table 2. Grass A was not heat-treated while G was the most exposed to high temperatures. Grass extracts were made by blending 10 g of dried grass for one hour in 40 mL of extraction buffer.
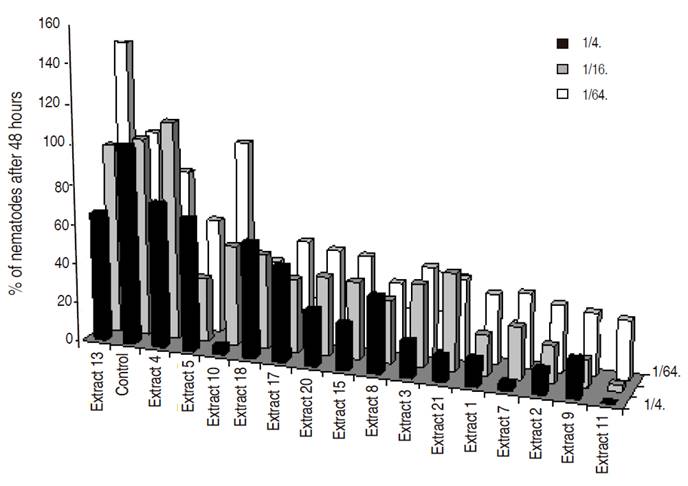
Figure 2: Percentage over the control of total P. penetrans found after 48 hours for different Brassicaceae extracts at different concentrations.
Dilutions
Extract dilutions were made to test the dose-response of nematode migration activity. The dilutions show doses that could be obtained by practical quantities of crop residue on field. 6 mL from each extract of the stock-solution was used. This volume was diluted to 1/3, where 6 mL was used to make the dilutions 1/4, 1/16, and 1/64. The final dilutions were prepared with the same phosphate buffer (pH 7) as the original extracts. These dilutions were used for the filter plate experiments.
Filter plate experiments
The filter plate experiment was used to test the dose-response of nematode migration activity. There were two plates placed one on top of each other. The upper plate was a 96-wells high filter plate with a small tube underneath. The lower plate was a standard 96-wells plate. The upper plates were filled with cigarette filters. It was pipetted 700 µL of the plant extract solution on these filters, then 200 µL of the nematode suspension (200 P. penetrans and 100 J2 M. chitwoodi nematodes). This proportion was selected because Meloidogyne migrates faster, so there would be more nematodes per well. The plates were covered and stored at 20 °C for 24 hours. After 24 hours, the upper plate was transferred to a new lower plate, and 200 µL of buffer was added. The whole set was place at 20 °C for 24 hours again, so the rest of the nematodes could migrate. The plates were counted again after 48 hours. Four replications per dilution were scored, and four wells with 900 µL of buffer per plate were used as a control. For each well, the number of nematodes was scored. Juveniles and adults P. penetrans were counted separately.
Nematode counting for each dilution was compared and statistically analyzed with STATISTIX for Windows. All data were log(x+1) transformed for an appropriate analysis. Tukey tests were used to compare treatments with significant differences (P<0.05).
RESULTS AND DISCUSSION
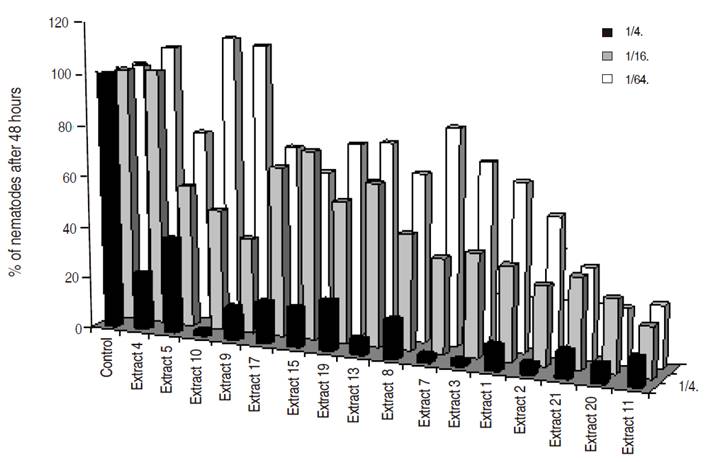
All the Brassicaseous and grass extracts showed nematicidal or repellent effect at a high concentration (1/4 dilution), whereas at lower concentrations (dilutions 1/16 and 1/64) differences were not always observed (Figures 2, 3 and 4). This nematicidal effect was a function of the dilution and time of exposure.
Figure 3: Percentage over the control of total P. penetrans found after 48 hours for the different grass extracts and Lupinus sp. at different concentrations.
Figure 4: Percentage over the control of total M. chitwoodi found after 48 hours for different Brassicaceae extracts at different concentrations.
Glucosinolates relative slight structural differences could confer deeply different nematicidal effects, confirming that biological activity was a function of the concentration of the product and the chemical properties of the R chain (Serra et al., 2002; Zasada and Ferris, 2003).
Best nematicidal effects at 1/4 dilution were observed with Extracts 11, grass G and Lupinus for P. penetrans (Tables 1 and 2), and with extracts 10, 3, and 7 for M. chitwoodi (Table 3). At 1/16 dilution the extracts with higher effect were 11, 9, and Lupinus sp. for P. penetrans and extract 11 for M. chitwoodi. At the lower concentration of 1/64 most effective extracts were 2, 11, 9, and Lupinus for P. penetrans, and 20 and 11 for M. chitwoodi.
Table 1: Mean of total nematodes present after 48 hours for different Brassicaceae extracts at different concentrations in P. penetrans corrected over the control.

Table 2: Mean of total nematodes present after 48 hours for different plant grass extracts at different concentrations in P. penetrans corrected over the control.
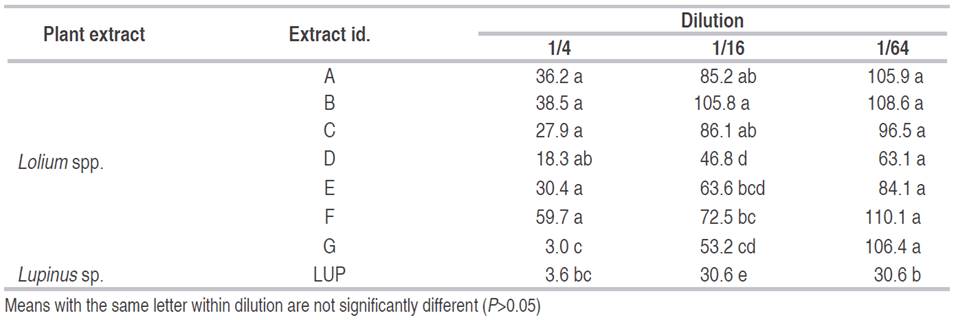
Table 3: Mean of total nematodes present after 48 hours for different Brassicaceae extracts at different concentrations in M. chitwoodi corrected over the control.

In cases where the metabolite had a better effect when used in a lower concentration (e.g., 1/64) could be assumed that its effect did not only depend on the concentration of the plant extract, but it was also related to the optimal dilution at which the compound gave positive results for nematode management.
For P. penetrans control the principal compounds involved are sinigrine and gluconapin according to preliminary studies performed by the Plant Research International in Wageningen. The first one was found in high concentrations in extracts 7 and 11 (Raphanus sativus and Brassica juncea oriental, respectively), while the latter was in excess in extracts 1 and 2 (Brassica fructiculosa both species) (data not shown). In the experience of Lazzeri et al. (1993) with a concentration of 0.05%, sinigrin and gluconapin showed nematicidal effect after 96 hours and 114 hours, respectively, for the control of Heterodera schachtii. Brassica juncea products have also shown a reduction in Globodera pallida (Lord et al., 2011), P. penetrans and M. incognita (Zasada et al., 2009). Ngala et al. (2014) also found nematicidal effects for the same nematode with B. juncea and Raphanus sativus deterred its multiplication.
For M. chitwoodi control showed a clear correlation between the type of glucosinolate so its control effectiveness could not be done easily because most of the extracts controlled the nematode effectively, at least for the lowest concentration (Table 3). However, it could be assumed that several isothiocyanates or the combination of them have detrimental effects on M. chitwoodi populations; therefore, its use could be highly appreciated in crops where this nematode causes economic losses. Further chemical studies of the most promissory plant species are necessary in order to determine specific compounds that have nematicidal or nematostatic effects.
CONCLUSIONS
Glucosinolates present in Brassica juncea and Lupinus sp. showed the most significant nematicidal effect with the lowest dilution (1/4). It seemed that the high concentration of these glucosinolates in the plant tissue is the direct responsible for the biocidal effect of these plants. Given the high effectiveness of most of the tested Brassica spp. for the control of M. chitwoodi and P. penetrans.
ACKNOWLEDGEMENTS
The author is grateful to the staff from Plant Research International - Wageningen for any input given. This study was part of a graded project presented in February 2004 for the course Ecological Aspects of Biointeractions. For the MSc in Plant Sciences, at Wageningen University in The Netherlands.
REFERENCES
References
Anastasiadis I and Karanastasi E. 2011. Factors affecting the efficacy of Brassica species and ryegrass (Lolium perenne L.) on root-knot nematode infestation of tomato. Communications in Agricultural and Applied Biological Sciences 76(3): 333-340.
Antonious GF, Bomford M and Vincelli P. 2009. Screening Brassica species for glucosinolate content. Journal of Environmental Science and Health 44(3): 311-316. doi: 10.1080/03601230902728476
Avato P, D’Addabbo T, Leonetti P and Argentieri M. 2013. Nematicidal potential of Brassicaceae. Phytochemistry Reviews 12(4): 791-802. doi: 10.1007/s11101-013-9303-7
Björkman M, Klingen I, Birch ANE, Bones AM, Bruce TJA, Johansen TJ, Meadow R, Mølmann J, Seljåsen R, Smart LE and Stewart D. 2011. Phytochemicals of Brassicaceae in plant protection and human health – influences of climate, environment and agronomic practice. Phytochemistry 72(7): 538-556. doi: 10.1016/j.phytochem.2011.01.014
Buskov S, Serra B, Rosa E, Sørensen H, Sørensen J. 2002. Effect of intact glucosinolates and products produced from glucosinolates in myrosinase-catalyzed hydrolysis on the potato cyst nematode (Globodera rostochiensis Cv. Woll). Journal of Agricultural and Food Chemistry
(4): 690-695. doi: 10.1021/jf010470s
Castagnone-Sereno P, Leroy F, Bongiovanni M, Zijlstra C and Abad P. 1999. Specific diagnosis of two root-knot nematodes, Meloidogyne chitwoodi and M. fallax, with satellite DNA probes. Phytopathology 89(5): 380-384. doi: 10.1094/PHYTO.1999.89.5.380
Chitwood DJ. 2002. Phytochemical based strategies for nematode control. Annual Review of Phytopathology 40: 221-249. doi: 10.1146/annurev.phyto.40.032602.130045
Cronquist A. 1981. An integrated system of classification of flowering plants. Columbia University Press, New York. 1262p.
D’Addabbo T, Carbonara T, Leonetti P, Radicci V, Tava A and Avato P. 2011. Control of plant parasitic nematodes with active saponins and biomass from Medicago sativa. Phytochemistry Reviews 10(4): 503-519. doi: 10.1007/s11101-010-9180-2
Devi G. 2018. Biofumigation: A potential aspect for suppression of plant-parasitic nematodes. International Journal of Environment, Agriculture and Biotechnology (3)4: 1285-1291. doi: 10.22161/ijeab/3.4.20
Fahley JW, Zalcmann AT and Talay P. 2001. The chemical diversity and distribution of glucosinolates and isothyocyanates among plants. Phytochemistry 56(1): 5-51. doi: 10.1016/S0031-9422(00)00316-2
Kirkegaard J, Matthiessen J, Wong P, Mead A, Sarwar M and Smith B. 1999. Exploiting the biofumigation potential of Brassicas in farming systems. pp 1-7. In: Proceedings of the 10th International Rapeseed Congress. The regional Institute Ltd. Canberra.
Lazzeri L, Tacconi R and Palmieri S. 1993. In vitro activity of some glucosinolates and their reaction products toward a population of the nematode Heterodera schachtii. Journal of Agricultural and Food Chemistry 41(5): 825-829. doi: 10.1021/jf00029a028
Lord J, Lazzeri L, Atkinson H and Urwin P. 2011. Biofumigation for control of pale potato cyst nematodes: Activity of brassica leaf extracts and green manures on Globodera pallida in vitro and in soil. Journal of Agricultural and Food Chemistry 59(14): 7882-7890. doi: 10.1021/jf200925k
Matsumoto MN, Goulart AMC, Homechin M, Sakamoto MMY and Santiago DC. 2002. Mulching with Pennisetum purpureum and other cultural practices for management of Meloidogyne javanica on tomato under greenhouse conditions. Nematologia Brasileira 26: 101-104.
McFadden W, Potter J and Brandle J. 1992. Use of Brassica spp. as green manure for biological control of Pratylenchus penetrans. Phytopathology 82(2): 246.
Mojtahedi H, Santo GS, Wilson JH and Hang AN. 1993. Managing Meloidogyne chitwoodi on potato with rapeseed as green manure. Plant Disease 77(1): 42-46. doi: 10.1094/PD-77-0042
Ngala M, Haydock P, Woods S and Back M. 2015. Biofumigation with Brassica juncea, Raphanus sativus, and Eruca sativa for the management of field populations of the potato cyst nematode Globodera pallida. Pest Management Science 71(5): 759-769. doi: 10.1002/ps.3849
Oliveira RDL, Dhingra OD, Lima AO, Jham GN, Berhow MA, Holloway RK and Vaughn S. 2010. Glucosinolate content and nematicidal activity of Brazilian wild mustard tissues against Meloidogyne incognita in tomato. Plant and Soil 341(1-2): 155-164. doi: 10.1007/s11104-010-0631-8
Peng Y and Moens M. 2002. Tolerance of Rosa rootstocks and species to Pratylenchus penetrans.
Nematology 4(3): 395-401. doi: 10.1163/156854102760199231
Potter M, Davies K and Rathjen A. 1998. Suppressive impact of glucosinolates in Brassica vegetative tissues on root lesion nematode Pratylenchus neglectus. Journal of Chemical Ecology 24(1): 67-80. doi: 10.1023/A:1022336812240
Rosa EAS, Heaney RK, Fenwick GR and Portas CAM. 1997. Chapter 3: Glucosinolates in crop plants. pp. 99-215. In: Janick J (ed). Horticultural Review. Vol. 19. John Wiley & Sons, Inc. 441p.
Serra B, Rosa E, Iori R, Barillari J, Cardoso A, Abreu C and Rollin, P. 2002. In vitro activity of 2-phenylethyl glucosinolate, and its hydrolysis derivatives on the root-knot nematode Globodera rostochiensis (Woll.). Scientia Horticulturae 92(1): 75-81. doi: 10.1016/S0304-4238(01)00277-1
Wildmer TL and Abawi GS. 2007. Mechanism of suppression of Meloidogyne hapla and its damage by a green manure of Sudan grass. Plant Disease 84: 562-568. doi: 10.1094/PDIS.2000.84.5.562
Zasada I and Ferris H. 2003. Sensitivity of Meloidogyne javanica and Tylenchulus semipenetrans to isothiocyanates in laboratory assays. Nematology 93(6): 747-750. doi: 10.1094/PHYTO.2003.93.6.747
Zasada IA, Meyer SLF and Morra MJ. 2009. Brassicaceous seed meals as soil amendments to suppress the plant-parasitic nematodes Pratylenchus penetrans and Meloidogyne incognita. The Journal of Nematology 41 (3): 221-227.
How to Cite
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Download Citation
CrossRef Cited-by
1. Donald Riascos-Ortiz, Ana T. Mosquera-Espinosa, Francia Varón de Agudelo, Claudio Marcelo Gonçalves Oliveira, Jaime Eduardo Muñoz Flórez. (2022). Sustainable Management of Nematodes in Agriculture, Vol.1: Organic Management. Sustainability in Plant and Crop Protection. 18, p.381. https://doi.org/10.1007/978-3-031-09943-4_15.
2. Ece Börteçine KASAPOĞLU ULUDAMAR. (2023). Nematicidal activity of various aqueous extracts against root-knot nematodes (Meloidogyne chitwoodi). Tarım Bilimleri Dergisi, https://doi.org/10.15832/ankutbd.1329956.
3. Ana María Mesa Vanegas, Jessica Wagner Arenas, Omar Ocampo Jiménez, O, Zulma Monsalve Fonnegra, I. (2023). Nematicidal activity and in vitro radical scavenging from Piper cumbricola and Piper eriopodon. Biocatalysis and Agricultural Biotechnology, 47, p.102595. https://doi.org/10.1016/j.bcab.2022.102595.
Dimensions
PlumX
Article abstract page views
Downloads
License
Copyright (c) 2019 Revista Facultad Nacional de Agronomía Medellín

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License.
The journal allows the author(s) to maintain the exploitation rights (copyright) of their articles without restrictions. The author(s) accept the distribution of their articles on the web and in paper support (25 copies per issue) under open access at local, regional, and international levels. The full paper will be included and disseminated through the Portal of Journals and Institutional Repository of the Universidad Nacional de Colombia, and in all the specialized databases that the journal considers pertinent for its indexation, to provide visibility and positioning to the article. All articles must comply with Colombian and international legislation, related to copyright.
Author Commitments
The author(s) undertake to assign the rights of printing and reprinting of the material published to the journal Revista Facultad Nacional de Agronomía Medellín. Any quotation of the articles published in the journal should be made given the respective credits to the journal and its content. In case content duplication of the journal or its partial or total publication in another language, there must be written permission of the Director.
Content Responsibility
The Faculty of Agricultural Sciences and the journal are not necessarily responsible or in solidarity with the concepts issued in the published articles, whose responsibility will be entirely the author or the authors.