Evaluación de tres promotores constitutivos para la expresión GUS en arroz (Oryza sativa L., cv. J-104)
Assessment three constitutive promoters for GUS expression in rice (Oryza sativa L., var. J-104)
DOI:
https://doi.org/10.15446/rev.colomb.biote.v18n1.57716Palabras clave:
CaMV 35S, chimerical A9 promoter, ubiquitine-1 (es)Se analizó la expresión constitutiva del gen reportero de la ß-Glucuronidasa (GUS) fusionado a tres promotores: el 35S del virus del mosaico de la coliflor (CaMV), el promotor quimérico A9 que contiene la actina-1 de arroz y el promotor ubiquitina-1 de maíz. La actividad de los promotores fue analizada cualitativa y cuantitativamente en diferentes tejidos y estadíos de crecimiento de plantas de arroz (variedad J-104) transformadas mediante biobalística. Se demostró la expresión constitutiva de GUS bajo los promotores estudiados, con distintos patrones de actividad relativa en hojas, tallos y raíces de plantas in vitro y ex vitro, y en plantas de la progenie T1. Bajo el promotor quimérico A9 se lograron los mayores niveles de expresión GUS en todos los tejidos y fases de crecimiento de las plantas.
This work analyzed the constitutive expression of the ß-Glucuronidase (GUS) reporter gene fused to three promoters: the cauliflower mosaic virus (CaMV) 35S, the chimerical A9 promoter which contains rice Act1, and the Ubiquitine-1 promoter from maize. The activity of the promoters was qualitative and quantitatively obtained in different tissues and various growth stages of rice plants (cv J-104) transformed by biolistic. All the promoters were found to be active, with distinct patterns of relative activity in leaves, stem and roots from in vitro and ex vitro plants, and in plants of T1 progeny. The chimerical A9 promoter increased significantly levels of GUS expression in all the tissues and at all growth stages of the plants.
Key words: CaMV 35S, chimerical A9 promoter, ubiquitine-1.
DOI: https://doi.org/10.15446/rev.colomb.biote.v18n1.57716
ARTÍCULO DE INVESTIGACIÓN
Assessment three constitutive promoters for GUS expression in rice (Oryza sativa L., var. J-104)
Evaluación de tres promotores constitutivos para la expresión GUS en arroz (Oryza sativa L., cv. J-104)
Maylin Pérez Bernal*, Daymí Abreu Remedios**, Onel Valdivia Pérez***, Magalis Delgado Rigo****, Raúl Armas Ramos*****
* MSc Plant Biology, Research Department. Center for Genetic Engineering and Biotechnology of Sancti Spiritus. P.O.Box 83, P.C. 60200. Sancti Spiritus, Cuba, e-mail: maylin.perez@cigb.edu.cu (corresponding author)
** MSc Plant Biology, Research Department. Center for Genetic Engineering and Biotechnology of Sancti Spiritus. P.O.Box 83, P.C. 60200. Sancti Spiritus, Cuba, e-mail: daymi.abreu@cigb.edu.cu
*** MSc Plant Biology, Research Department. Center for Genetic Engineering and Biotechnology of Sancti Spiritus. P.O.Box 83, P.C. 60200. Sancti Spiritus, Cuba, e-mail: onel.valdivia@cigb.edu.cu
**** Laboratory Technician, Research Department. Center for Genetic Engineering and Biotechnology of Sancti Spiritus. P.O.Box 83, P.C. 60200. Sancti Spiritus, Cuba, e-mail: magalis.delgado@cigb.edu.cu
***** MSc Plant Biology, Research Department. Center for Genetic Engineering and Biotechnology of Sancti Spiritus. P.O.Box 83, P.C. 60200. Sancti Spiritus, Cuba, e-mail: raul.armas@cigb.edu.cu
Recibido: diciembre 14 de 2015 Aprobado: mayo 11 de 2016
Abstract
This work analyzed the constitutive expression of the ß-Glucuronidase (GUS) reporter gene fused to three promoters: the cauliflower mosaic virus (CaMV) 35S, the chimerical A9 promoter which contains rice Act1, and the Ubiquitine-1 promoter from maize. The activity of the promoters was qualitative and quantitatively obtained in different tissues and various growth stages of rice plants (cv J-104) transformed by biolistic. All the promoters were found to be active, with distinct patterns of relative activity in leaves, stem and roots from in vitro and ex vitro plants, and in plants of T1 progeny. The chimerical A9 promoter increased significantly levels of GUS expression in all the tissues and at all growth stages of the plants.
Key words: CaMV 35S, chimerical A9 promoter, ubiquitine-1.
Resumen
Se analizó la expresión constitutiva del gen reportero de la ß-Glucuronidasa (GUS) fusionado a tres promotores: el 35S del virus del mosaico de la coliflor (CaMV), el promotor quimérico A9 que contiene la actina-1 de arroz y el promotor ubiquitina-1 de maíz. La actividad de los promotores fue analizada cualitativa y cuantitativamente en diferentes tejidos y estadíos de crecimiento de plantas de arroz (variedad J-104) transformadas mediante biobalística. Se demostró la expresión constitutiva de GUS bajo los promotores estudiados, con distintos patrones de actividad relativa en hojas, tallos y raíces de plantas in vitro y ex vitro, y en plantas de la progenie T1. Bajo el promotor quimérico A9 se lograron los mayores niveles de expresión GUS en todos los tejidos y fases de crecimiento de las plantas.
Palabras clave: CaMV 35S, promotor quimérico A9, ubiquitina-1.
Introduction
A constitutive promoter is able to drive gene expression in many or all tissues of a plant (Xiao et al., 2005).There are several advantages in using constitutive promoters in expression vectors used in plant biotechnology, such as: elevated production of proteins to select transgenic cells or plants; high level of expression of reporter proteins, allowing easy detection and quantification; high production of a transcription factor that is part of a regulatory transcription system; productions of compounds that requires ubiquitous activity in the plant, and compounds that are required during all stages of plant development (Park et al., 2010).
The cauliflower mosaic virus (CaMV) 35S gene promoter is the most commonly used in plant biotechnology research to drive transgene expression in a constitutive form. However, although the 35S promoter and its derivatives can drive high levels of transgene expression in dicotyledonous plants; their activities are lower in monocotyledonous plants (Park et al., 2010). Moreover, CaMV35S promoter may increase the chances of transcriptional inactivation (Stam et al., 1997), and its expression can be modulated by photoperiod, temperature and the developmental stage of the plants (Obertello et al., 2005).
Others constitutive promoters have been assessed for their capability to drive the expression of transgenes in monocots, including ZmUbi1 (Cornejo et al., 1993) from maize, and Act1 (McElroy et al., 1990), OsCc1 (Jang et al., 2002), RUBQ1 and 2 (Wang and Oard, 2003), rubi3 (Lu et al., 2008), and OsAct2 (He et al., 2009) from rice. These promoters are highly active in monocot crops, but they are also dissimilar in various ways.
The rice Act1 promoter is one of the most frequently used highly active and constitutive promoter, for the establishment of transformation procedures and expression of foreign genes in rice and other monocots. Plant actin promoters are likely to be active in all tissues because actin is a fundamental component of the plant cell cytoskeleton. In rice, there are at least eight actin-like sequences per haploid genome; four of these have been isolated and shown to differ from each other in the tissue- and stage-specific abundance of their respective transcripts. The rice actin 1 gene, Act1, was found to encode a transcript that is relatively abundant in all rice tissues and at all developmental stages examined (McElroy et al., 1990).
Another constitutive promoter that is also commonly utilized for transformation of monocots is the maize Ubi promoter. This promoter is active in many, although not all, cell types and drives strong expression in young roots and leaves, but these expression levels decrease markedly as these organs mature (Cornejo et al., 1993). In general, the maize Ubi promoter has slightly higher activity than the rice Act1 promoter in various monocot species.
Reporter genes have been used in all tissues and at all growth stages of transgenic plants and they are integral components for plant transformation studies. A widely used reporter gene is the gus gene that encodes the enzyme β-glucuronidase (GUS) (Jefferson et al., 1987). The β-glucuronidase assay is very sensitive, and it is possible to obtain both qualitative (histochemical) and quantitative (fluorometric) data. Studies on gus reporter gene fusion system have been conducted for various purposes like: (i) to understand developmental patterns of gene expression; (ii) to optimize the particle bombardment parameters; (iii) to compare different transformation methods; (iv) to identify the ideal promoter for plant species transformation and (v) to compare the best explant for plant transformation and regeneration (Basu et al., 2004). GUS reporter gene is routinely used for promoter analyses (Obertello et al., 2005; Park et al., 2010).
With the aim of obtain a highly efficient promoter that could drive constitutive transgene expression in the cuban rice cultivar J-104, three different promoters fused to the gus reporter gene were introduced in rice calli, via microprojectile bombardment. The promoters were the CaMV 35S, the chimerical A9 promoter and the Ubiquitine-1 promoter. The activity of the constitutive promoters was qualitative and quantitatively analyzed, using the GUS reporter system (Jefferson, 1987), in different tissues and at various growth stages of transgenic rice plants obtained from calli.
Materials and methods
Plant material
Mature rice (Oryza sativa L., cv J-104) seeds were manually dehulled and washed with water, following surface sterilization with a brief (30 s) rinse with 70% ethanol. The seeds were treated with 2.5% active chlorine solution for 25 min. The seeds were thoroughly washed six times with sterilized distilled water, and were shifted separately to sterilized filter papers for drying.
The callus induction medium consisted of salts and vitamins from N6 (Chu et al., 1975), 30 g/L sucrose, 1.0 g/L casein hydrolysate, 2.0 mg/L 2,4-D and 2.0 g/L Phytagel. The culture medium was poured into Petri dishes, with 10 seeds per dish. Cultures were maintained in darkness at 28 °C. After 30 days the calli were excised from the seeds and were used as explants for microprojectile bombardment.
Plasmids
Three plant vectors were used: p35SGUS, pUbiGUS and pA9GUS, which contained CaMV35S (cauliflower mosaic virus 35S), Ubi1 from maize (Cornejo et al., 1993) and A9 (chimerical promoter), respectively, having gus gene and the nopaline synthase terminator from Agrobacterium tumefaciens.
The chimerical A9 promoter consisted of the quadruplicated octopine synthase enhancer element from Agrobacterium tumefaciens, a version from active region of the CaMV35S promoter, and the first exon/intron/exon from rice Act1 (McElroy et al., 1990).
The plasmid DNA preparations were carried out according to the supplier instructions (QIAprep® miniprep kit, QIAGEN).
Microprojectile bombardment
Gold particles of 1.0 µm diameter were coated with the DNA plasmid as described by Taylor and Vasil (1991). Three sessions of bombardment, with 10 plates each one, were carried out. Fifteen calli of 2-3 mm in length were placed onto N6 (Chu et al., 1975) medium with 0.8 M mannitol, in each 6-cm diameter dish and bombarded with 0.154 µg gold and 0.350 µg DNA, after a vacuum of 700 mm Hg was attained. Target distance was adjusted to 6 cm from stopping plate to callus tissue.
After bombardment, calli were kept 16 h in the dark, and then were transferred to callus induction medium and they were maintained two weeks in the dark at 28°C.
Plant regeneration
The plant regeneration from calli was done using the KIBAM culture medium (Pérez Bernal et al., 2007). Cultures were maintained during a month at 25±2°C in environmentally controlled room under illumination of 1500 lux emitted by fluorescent tubes.
Regenerated plants were transferred to MS (Murashige and Skoog, 1962) medium with 3.0 g/L Phytagel. The plants with a height of 10 cm were removed from culture medium, washed their roots, placed in pots with soil and cultured in ex-vitro environment under natural conditions. T1 seeds were obtained by self-pollination of primary transformed rice (T0) plants. The seeds were used for studying the segregation of the gus gene in T1 progeny. Eighteen lines were randomly selected, six of each promoter in study. The seeds per line were germinated on MS culture medium to obtain plants of T1 progeny, which were tested by histochemical GUS assay.
Histochemical GUS assay in rice tissues
GUS assay was performed as described by Jefferson (1987). Rice tissues (leaves, stem, roots and seeds) were transferred to eppendorf tubes containing GUS staining solution (50 mM sodium phosphate at pH 7.0, 10 mM EDTA, 0.1% Triton X-100, 1 mg/ml X-Gluc, 0.1 mM potassium ferricyanide, 0.1 mM potassium ferrocyanide and 20% methanol) and incubated at 37°C until the apparition of blue spots. The staining solution was then removed, and the tissues were stored in 70% ethanol. Tissues were examined under a stereoscopic microscope.
Fluorimetric GUS assay
Leaves, stems and roots of six ex vitro rice plants were ground separately with liquid nitrogen. Protein extraction buffer (50 mM NaH2PO4, pH 7.0, 0.01% SDS, 0.01 M EDTA, pH 8.0, 0.1% Triton X-100 and 10 mM mercaptoethanol) was added to the frozen tissue powder (~100 mg) and vortexed to homogeneity. The homogenate was centrifuged at 11 000 g for 15 min at 4°C. The total soluble proteins in the supernatant were quantified by Bradford method and used for fluorimetric assay.
Fluorimetric GUS assay was performed in 500 μl of reaction, consisted of 400 µl of protein sample in extraction buffer and 100 µL of 10 mM 4-methylumbelliferylß-D-glucuronide (4-MUG). The reaction was incubated at 37°C. At zero time, an aliquot of 50 μl reaction was taken out and added to 450 μl 0.2 M sodium carbonate. The same manipulation was performed at subsequent times: 5, 10, 15, 20, 25 and 30 minutes.
MUG is cleaved by GUS to release methylumbelliferone (MU) and glucuronic acid. MU fluorescence was quantified in spectrofluorometer using excitation and emission wavelengths of 365 and 455 nm, respectively. Values were calculated according to a reference range of MU. Each sample was analyzed in triplicate. Fluorimetric assessments of GUS activity were expressed in terms of nmol of MU produced per minute per microgram of protein.
GUS activity in polyacrylamide denaturing gel
The electrophoretic run of the non heated samples (total soluble proteins extracted from leaves of regenerated plants) was performed in 10% polyacrylamide denaturing gel. After that, the gel was incubated 1h with 50 mL Triton X-100 2.5 % v/v in water, to renature the enzyme, and then was incubated with 10 mL of histochemical staining solution at 37°C, until it appeared the blue-stained bands. Total soluble proteins from leaves of tobacco transplantomic plant, with verified GUS activity in chloroplasts, were used as positive control. The tobacco transplantomic plant was provided by Molecular Biology Laboratory of Center for Genetic Engineering and Biotechnology of Sancti Spiritus, Cuba.
Statistical analysis
In the segregation analysis of gus gene to T1 progeny, the numbers of GUS positive and GUS negative plants were processed by a Chi-square goodness of fit test (p≥0.05), using the version 11.5 of the Statistical Package for Social Sciences.
Results
Qualitative GUS activity of in vitro rice tissues
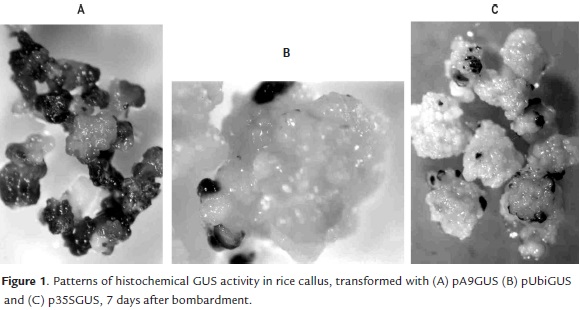
After 72 hours of the bombardment, the histochemical GUS assay did not show visible differences between the calli bombarded with three promoters under study. In all cases small and isolated blue spots on the outermost layer of calli were observed. However, the GUS staining patterns in calli cultured during 7 days after the bombardment were different, depending on the promoter used to drive the GUS expression. Callus tissues transformed with A9 promoter showed large and numerous blue-staining areas (Fig 1A), whereas with the Ubi1 promoter the blue areas were smaller and more isolated (Fig 1B), similar to the results obtained with the 35S promoter (Fig 1C).
In plants regenerated from transformed calli the constitutive expression of gus gene drove by three promoters was verified: GUS staining was confirmed in leaves, stems and roots of all the plants. But some differences were observed in the staining pattern depending on the promoter. Using A9 promoter, GUS activity was qualitatively higher in all plant organs; the roots dyed completely (Fig 2A), the blue color was more intense in leaves and stems, and the blue stain was spread to the substrate solution. The blue staining was detected in all the organs of the plants Ubi1-GUS and 35S-GUS, but it was less intense than in A9-GUS plants: the figures 2B and 2C show the blue roots after the histochemical reaction.
The blue-stained bands in polyacrylamide denaturing gel with renatured enzyme were observed after 30 minutes of gel incubation on histochemical staining solution at 37°C (Fig 3). The active form of the enzyme is a tetramer of four identical subunits (Jefferson, 1987). The wider band (lane 6) corresponded to protein extract from tobacco transplantomic plant, where the GUS activity was higher (positive control), because it is in chloroplasts. Chloroplast has been exploited as a powerful prokaryotic-like expression platform for the production of heterologous proteins (Rasala and Mayfield, 2014). The other wide band (lane 3) corresponded to the protein extract from rice plants transformed with pA9GUS, which confirm previous results that had shown increased qualitative GUS activity with A9 promoter, compared with the others two promoters.
Qualitative GUS activity in ex vitro plants
The acclimatization of plants to the ex vitro environment was successful, considering that 98% of them survived and seeds set under the established conditions.
GUS activity during ex vitro vegetative growth was examined in sections of leaves, stems and roots which had been treated with the chromogenic GUS substrate X-Gluc.
The pattern of activity conferred by the 35S promoter in ex-vitro plants was studied first and used as a reference. Histochemical staining of the aerial part of these plants revealed the staining of vascular tissues in stems and leaves. The intensity of staining increased as the leaves were older. The primary root, the root cap and the cross sections were deep blue, whereas the elongation zone was less stained.
Like the 35S, the A9-GUS plants exhibited a constitutive pattern of expression in leaves, stems and roots. The stems sections were stained deep blue; the intense staining of the leaves vascular tissue was also observable. The intensity of the color increased with the age of the leaves, suggesting a constitutive and high GUS expression. The main root exhibited reporter gene activity even in the elongation zone and the secondary roots were completely dark blue.
Transgenic plants harboring Ubi1-GUS showed a much lower blue staining unevenly distributed. Longitudinal shoot sections revealed that the indigo staining was restricted to young leaves bases. In the primary root, the ß-glucuronidase activity was confined to the root tip in a region that might correspond to the meristem and the elongation zone.
Quantitative GUS expression
To support the differences observed in qualitative GUS expression in plants transformed with three promoters, fluorimetric activity was quantified and more accurate comparable results were achieved.
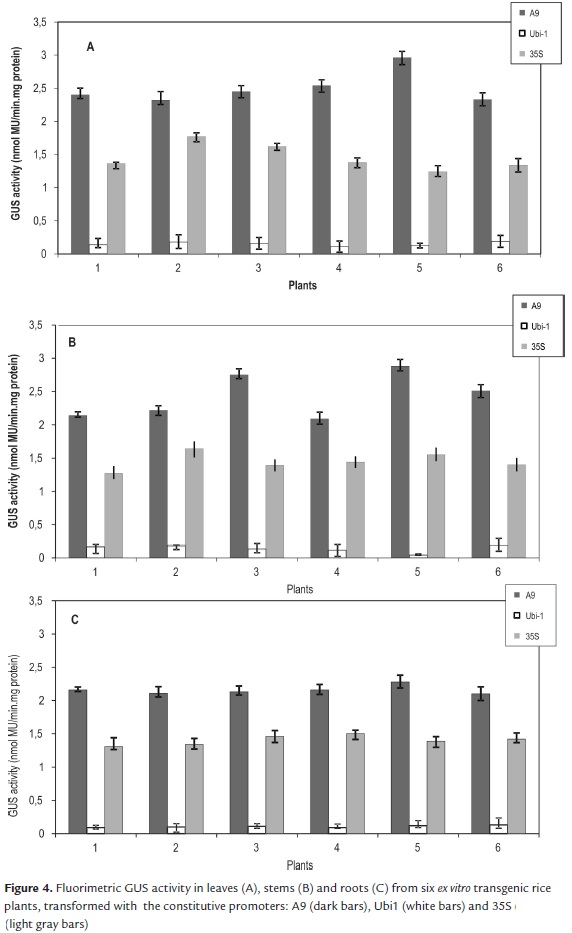
In agreement with the results of the qualitative GUS activity, the highest values of fluorimetric activity were found with the gus gene drove by A9 promoter. In leaves the fluorimetric GUS activity had the maximum average, 2.5 nmol MU/min.mg protein. In stems the activity ranged from 2.09 to 2.88 nmol MU/min.mg protein, and in roots ranged from 2.1 to 2.28 nmol MU/min.mg protein (Fig. 4).
An average of 1.43 nmol MU/min.mg protein was calculated for the fluorimetric GUS activity among leaves, stems and roots of plants transformed with 35S promoter was. Qualitatively, there were not visible differences in GUS expression in plants transformed with the A9 and 35S promoters. However, quantitative assay confirmed that A9 promoter effectiveness was 1.74 fold higher than the 35S in the three tissues.
GUS activity recorded in leaves and stems transformed with the Ubi1 promoter averaged 2-fold lesser activity than that of the A9 promoter. As in qualitative assay, GUS expression in roots was slight in the six plants, and the fluorimetric activity ranged only between 0.09 and 0.13 nmol MU/min.mg protein (Fig 4).
Segregation of gus gene to T1 progeny
All plants were capable to produce healthy and fertile seed of T1 progeny three months after the transplant to ex vitro environment. The GUS assay was conducted in manually dehulled seeds and in longitudinal sections of the seeds.
The penetration of the substrate in the intact seeds was limited, so only small blue spots were observed in the external coating of them. The blue staining was detected rapidly in the embryos from seeds harboring the pA9GUS. Embryos transformed with p35SGUS y pUbiGUS were stained one hour later. The histochemical staining was completely distributed in the starchy endosperm of the longitudinal sections of the seeds (Fig 5).
Segregation analysis of the gus gene was performed in eighteen independent lines of T1 progeny. Plants germinated on MS culture medium were subjected to histochemical GUS assays. Results indicated that 17 of 18 evaluated lines segregated the gus gene to T1 progeny in a Mendelian form. Only the line 35S-4 had not complied with a ratio 3:1 GUS positive/GUS negative (Table 1).
Discussion
The promoter plays the most important role in determining the temporal and spatial expression pattern and gene transcript, although the final amount of gene product is determined at both the transcriptional and post-transcriptional levels (Qu et al., 2008). To date, some strong constitutive promoters, such as the cauliflower mosaic virus 35S promoter and maize Ubiquitin-1, are widely used in plant biotechnology research.
The gus gene is commonly used to evaluate the activity of gene promoters. Qu et al. (2008) examined the activity of six glutelin promoters fused to gus gene, in order to develop diverse promoters to drive multigene expression while avoiding the problem of homology-based gene silencing in transgenic rice. Xiao et al. (2005) used gus gene for the isolation and characterization of a novel plant promoter that directs strong constitutive expression of transgenes in plants. The authors demonstrated that the promoter could lead to GUS expression at various developmental stages and in different tissues and organs, similar results to those obtained in the present work.
The promoters CaMV35S, A9 and Ubi1 were found to be active in the whole J-104 rice plant body or in various tissues and/or stages of plant growth, with distinct patterns of relative activity. Qualitative and quantitative analysis of GUS activity demonstrated that GUS expression is higher under A9 promoter. Although it is considered to be a strong constitutive promoter, the expression of genes regulated by CaMV-35S promoter can be modulated by photoperiod, temperature and the developmental stage (Obertello et al., 2005). That could be a reason for which the GUS expression under 35S in ex vitro plants was minor in some organs, compared with the GUS expression under A9 promoter that was better in whole plant and growth stages.
Since the ubiquitin genes are expressed in almost all plant tissues in their native context, their promoters should drive constitutive gene expression in transgenic plants especially of the same species. Characterization of transgenic plants containing different ubiquitin promoters to date have revealed that these promoters drive gene expression preferentially in young tissues, vascular tissues and pollen grains (Plesse et al., 2001).
Histochemical localization of GUS activity revealed that Ubi1 was most active in rapidly dividing cells. This promoter is expressed in many, but not all, rice tissues and undergoes important changes in activity during the development of transgenic rice plants (Cornejo et al., 1993). We verified that, in ex vitro conditions, transgenic rice plants harboring Ubi1-GUS showed a much lower blue staining that was, in addition, irregularly distributed. It was not detected the constitutive GUS expression in old ex vitro plants, in which the reduction or absence of GUS activity in leaves and roots in this growth stage was demonstrated.
Hernandez et al. (2009) have reported that the ubiquitin promoters typically show high activity in young tissues, like the blue staining detected in all in the organs of the Ubi1-GUS plants during in vitro regeneration process, or in active dividing tissues like the primary root, where the ß-glucuronidase activity was confined to the root tip in a region that might correspond to the meristem and the elongation zone.
Differing with the Ubi1 promoter, the A9 not only achieved high constitutive GUS activity in vitro and in young tissues, but also in ex vitro plants with old leaves, mature roots and reproductive status, which is convenient if is necessary to express agronomically important traits.
The penetration of GUS staining solution in the starchy endosperm of dehulled seeds was limited, due to the hardness of the external coating of the seeds. Only small blue spots were observed in areas where may have been wounded during the manual shelling. In the longitudinal sections of the seeds, the blue product occupied the entire area subjected to the histochemical reaction. It was interesting to note that A9, Ubi1 and 35S showed the same expression pattern and activity in the endosperm and aleurone layer of the seeds, but the embryos from seeds transformed with the A9 promoter were deep blue rapidly after the incubation at 37°C with the substrate. For these reasons, we concluded that the A9 promoter is very active in the whole rice grain: embryo, endosperm and aleurone layer. A similar result was described by Park et al., in 2010, whose R1G1B promoter was reported as the first constitutive gene promoter that was active in the entire rice grain.
The Chi-square goodness of fit test allowed to assess between expected and observed segregation patterns. In the majority of the lines, gus gene was inherited in a expected 3:1 Mendelian ratio. Satoto et al. (2008) demonstrated that if the gene is dominant and inserted in one locus, it will follow a 3:1 Mendelian segregation pattern for a single dominant gene in the subsequent generation, since rice is primarily self-pollinated plant. For further use in the breeding programs, it is important to ensure that the transgene is dominant and inherits in a stable and predictable manner.
Conclusions
The promoters CaMV35S, A9 and Ubi1 were found to be active in the whole J-104 rice plant body or in various tissues and/or stages of plant growth, with distinct patterns of relative activity.
The A9 promoter direct significantly higher levels of GUS expression in J-104 rice tissues and growth stages.
References
Basu, C., Kausch, A.P., Chandlee, J.M. (2004). Use of beta-glucuronidase reporter gene for gene expression analysis in turfgrasses. Biochemical and Biophysical Research Communications, 320, 7-10.
Cornejo, M.J., Luth, D., Blankenship, K.M., Anderson, O.D., Blechl, A.E. (1993). Activity of a maize ubiquitin promoter in transgenic rice. Plant Molecular Biology, 23, 567-81.
Chu, C.C., Wang, C.C., Sun, C.S., Chen, H., Yin, K.C., Chu, C.Y. (1975). Establishment of an efficient medium for anther culture of rice through comparative experiments on the nitrogen sources. Scientia Sinica, 18, 659-68.
He, C., Lin, Z., McElroy, D., Wu, R. (2009). Identification of a rice actin 2 gene regulatory region for high-level expression of transgenes in monocots. Plant Biotechnology Journal, 7, 227-39.
Hernandez, C.M., Martinelli, A.P., Bouchard, R.A., Finer, J.J. (2009). A soybean (Glycine max) polyubiquitin promoter gives strong constitutive expression in transgenic soybean. Plant Cell Reports, 28, 837-49.
Jang, I.C., Choi, W.B., Lee, K.H., Song, S.I., Nahm, B.H., Kim, J.K. (2002). High-level and ubiquitous expression of the rice cytochrome c gene OsCc1 and its promoter activity in transgenic plants provides a useful promoter for transgenesis of monocots. Plant Physiology, 129, 1473-81.
Jefferson, R.A. (1987). Assaying chimeric genes in plants: The GUS gene fusion system. Plant Molecular Biology Reporter, 5, 387-405.
Lu, J., Sivamani, E., Li, X., Qu, R. (2008). Activity of the 5' regulatory regions of the rice polyubiquitin rubi3 gene in transgenic rice plants as analysed by both GUS and GFP reporter genes. Plant Cell Reports, 27, 1587-1600.
McElroy, D., Zhang, W., Cao, J., Wu, R. (1990). Isolation of an efficient actin promoter for use in rice transformation. The Plant Cell, 2, 163-71.
Murahige, T. and Skoog, F. (1962). A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiologia Plantarum, 15, 473-97.
Obertello, M., Santi, C., Mame-Oureye, S., Laplaze, L., Auguy, F., Bogusz, D. (2005). Comparison of four constitutive promoters for the expression of transgenes in the tropical nitrogen-fixing tree Allocasuarina verticillata. Plant Cell Reports, 24, 540-48.
Park, S.H., Yi, N., Kim, Y.S., Jeong, M.H., Bang, S.W., Choi, Y.D. (2010). Analysis of five novel putative constitutive gene promoters in transgenic rice plants. Journal of Experimental Botany, 61, 2459-67.
Plesse, B., Criqui, M.C., Durr, A., Parmentier, Y., Fleck, J., Genschik, P. (2001). Effects of the polyubiquitin gene Ubi.u4 leader intron and first ubiquitin monomer on reporter gene expression in Nicotiana tabacum. Plant Molecular Biology, 45, 655-67.
Pérez Bernal, M., Delgado, M., Hernández, C., & Armas, R. (2007). Morphological evaluation of shoots regenerated from hygromycin-resistant rice callus (cv. IACuba-28). Colombian Journal of Biotechnology, 9(1), 35-40.
Qu, L.Q., Xing, Y.P., Liu, W.X., Xu, X.P., Song, Y.R. (2008). Expression pattern and activity of six glutelin gene promoters in transgenic rice. Journal of Experimental Botany, 59, 2417-24.
Rasala, B.A. and Mayfield, S.P. (2014). Photosynthetic biomanufacturing in green algae: production of recombinant proteins for industrial, nutritional, and medical uses. Photosynthesis Research, 123, 1-13.
Satoto, J.S., Hartana, A., Slamet-Leodin, I.H. (2008). The segregation pattern of insect resistance genes in the progenies and crooses of transgenic Rojolele rice. Indonesian Agricultural Journal, 9, 35-43.
Stam, M., Mol, J.N.M., Kooter, J.M. (1997). The silence of genes in transgenic plants. Annals of Botany, 79, 3-12.
Taylor, M.G. and Vasil, I.K. (1991). Histology of, and physical factors affecting, transient GUS expression in pearl millet (Pennisetum glaucum (L.) R.BR.) embryos following microprojectile bombardment. Plant Cell Reports, 10, 120-25.
Wang, J. and Oard, J.H. (2003). Rice ubiquitin promoters: deletion analysis and potential usefulness in plant transformation systems. Plant Cell Reports, 22, 129-34.
Xiao, K., Zhang, C., Harrison, M., Wang, Z.Y. (2005). Isolation and characterization of a novel plant promoter that directs strong constitutive expression of transgenes in plants. Molecular Breeding, 15, 221-231.
Referencias
Basu, C., Kausch, A.P., Chandlee, J.M. (2004). Use of beta-glucuronidase reporter gene for gene expression analysis in turfgrasses. Biochemical and Biophysical Research Communications, 320, 7-10.
Cornejo, M.J., Luth, D., Blankenship, K.M., Anderson, O.D., Blechl, A.E. (1993). Activity of a maize ubiquitin promoter in transgenic rice. Plant Molecular Biology, 23, 567-81.
Chu, C.C., Wang, C.C., Sun, C.S., Chen, H., Yin, K.C., Chu, C.Y. (1975). Establishment of an efficient medium for anther culture of rice through comparative experiments on the nitrogen sources. Scientia Sinica, 18, 659-68.
He, C., Lin, Z., McElroy, D., Wu, R. (2009). Identification of a rice actin 2 gene regulatory region for high-level expression of transgenes in monocots. Plant Biotechnology Journal, 7, 227-39.
Hernandez, C.M., Martinelli, A.P., Bouchard, R.A., Finer, J.J. (2009). A soybean (Glycine max) polyubiquitin promoter gives strong constitutive expression in transgenic soybean. Plant Cell Reports, 28, 837-49.
Jang, I.C., Choi, W.B., Lee, K.H., Song, S.I., Nahm, B.H., Kim, J.K. (2002). High-level and ubiquitous expression of the rice cytochrome c gene OsCc1 and its promoter activity in transgenic plants provides a useful promoter for transgenesis of monocots. Plant Physiology, 129, 1473-81.
Jefferson, R.A. (1987). Assaying chimeric genes in plants: The GUS gene fusion system. Plant Molecular Biology Reporter, 5, 387-405.
Lu, J., Sivamani, E., Li, X., Qu, R. (2008). Activity of the 5’ regulatory regions of the rice polyubiquitin rubi3 gene in transgenic rice plants as analysed by both GUS and GFP reporter genes. Plant Cell Reports, 27, 1587–1600.
McElroy, D., Zhang, W., Cao, J., Wu, R. (1990). Isolation of an efficient actin promoter for use in rice transformation. The Plant Cell, 2, 163-71.
Murahige, T. and Skoog, F. (1962). A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiologia Plantarum, 15, 473-97.
Obertello, M., Santi, C., Mame-Oureye, S., Laplaze, L., Auguy, F., Bogusz, D. (2005). Comparison of four constitutive promoters for the expression of transgenes in the tropical nitrogen-fixing tree Allocasuarina verticillata. Plant Cell Reports, 24, 540-48.
Park, S.H., Yi, N., Kim, Y.S., Jeong, M.H., Bang, S.W., Choi, Y.D. (2010). Analysis of five novel putative constitutive gene promoters in transgenic rice plants. Journal of Experimental Botany, 61, 2459-67.
Plesse, B., Criqui, M.C., Durr, A., Parmentier, Y., Fleck, J., Genschik, P. (2001). Effects of the polyubiquitin gene Ubi.u4 leader intron and first ubiquitin monomer on reporter gene expression in Nicotiana tabacum. Plant Molecular Biology, 45, 655-67.
Pérez Bernal, M., Delgado, M., Hernández, C., & Armas, R. (2007). Morphological evaluation of shoots regenerated from hygromycin-resistant rice callus (cv. IACuba-28). Colombian Journal of Biotechnology, 9(1), 35-40.
Qu, L.Q., Xing, Y.P., Liu, W.X., Xu, X.P., Song, Y.R. (2008). Expression pattern and activity of six glutelin gene promoters in transgenic rice. Journal of Experimental Botany, 59, 2417-24.
Rasala, B.A. and Mayfield, S.P. (2014). Photosynthetic biomanufacturing in green algae: production of recombinant proteins for industrial, nutritional, and medical uses. Photosynthesis Research, 123, 1-13.
Satoto, J.S., Hartana, A., Slamet-Leodin, I.H. (2008). The segregation pattern of insect resistance genes in the progenies and crooses of transgenic Rojolele rice. Indonesian Agricultural Journal, 9, 35-43.
Stam, M., Mol, J.N.M., Kooter, J.M. (1997). The silence of genes in transgenic plants. Annals of Botany, 79, 3–12.
Taylor, M.G. and Vasil, I.K. (1991). Histology of, and physical factors affecting, transient GUS expression in pearl millet (Pennisetum glaucum (L.) R.BR.) embryos following microprojectile bombardment. Plant Cell Reports, 10, 120-25.
Wang, J. and Oard, J.H. (2003). Rice ubiquitin promoters: deletion analysis and potential usefulness in plant transformation systems. Plant Cell Reports, 22, 129-34.
Xiao, K., Zhang, C., Harrison, M., Wang, Z.Y. (2005). Isolation and characterization of a novel plant promoter that directs strong constitutive expression of transgenes in plants. Molecular Breeding, 15, 221–231.
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. Ryo Maruyama, Yasuyoshi Mayuzumi, Jun Morisawa, Shinya Kawai. (2021). Transgenic rice plants expressing the α-L-arabinofuranosidase of Coprinopsis cinerea exhibit strong dwarfism and markedly enhanced tillering. Plant Biotechnology, 38(3), p.379. https://doi.org/10.5511/plantbiotechnology.21.0616a.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2016 Revista Colombiana de Biotecnología

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
Esta es una revista de acceso abierto distribuida bajo los términos de la Licencia Creative Commons Atribución 4.0 Internacional (CC BY). Se permite el uso, distribución o reproducción en otros medios, siempre que se citen el autor(es) original y la revista, de conformidad con la práctica académica aceptada. El uso, distribución o reproducción está permitido desde que cumpla con estos términos.
Todo artículo sometido a la Revista debe estar acompañado de la carta de originalidad. DESCARGAR AQUI (español) (inglés).