Publicado
Potential fecundity, larval development, and survival of two invasive species of Arhopalus (Coleoptera: Cerambycidae) coexisting in southern South America
Fecundidad potencial, desarrollo y supervivencia larval de dos especies invasoras de Arhopalus (Coleoptera: Cerambycidae) que coexisten en el sur de Sudamérica
DOI:
https://doi.org/10.15446/caldasia.v41n2.75352Palabras clave:
Body size, Cerambycid, eggs, insect pests, larvae, long-horned beetles (en)Cerambícidos, escarabajos longicornios, huevos, insectos plaga, larvas, tamaño corporal (es)
Descargas
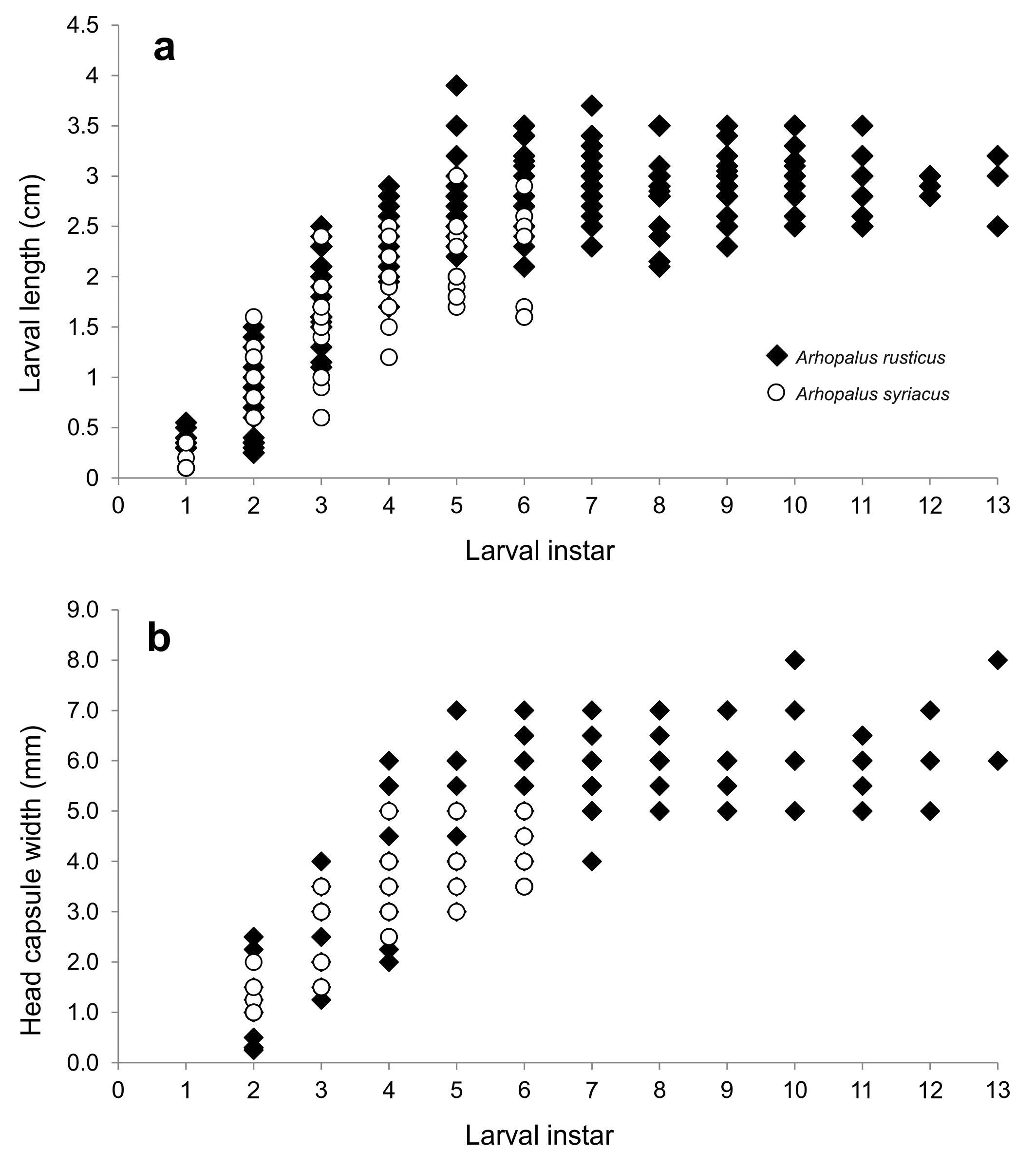
Biological invasions are one of the main threats to forest health. Although Arhopalus rusticus and Arhopalus syriacus (Cerambycidae) are alien species in South America, the ecology of these invasive species is not well studied. Here, we examined the effect of the female size on potential fecundity, size and number of mature eggs in ovaries, in both insect species. We also compared egg morphometric characteristics, larval measurements, larval development and survival between these two species. We found a positive relationship between the female size and the number of eggs in their ovaries but no relationship between the female size and egg morphometric characteristics in each of these two species. The females of A. rusticus were significantly larger and had bigger eggs than those of A. syriacus, but no significant difference was observed in the number of eggs between them. Larval body length and head capsule exuviae width were larger in A. rusticus than in A. syriacus. Arhopalus rusticus had the highest larval survival rate, but A. syriacus had the shortest larval period. Thus, female and egg size, as well as larval size and survivorship are competitive advantages to A. rusticus. However, A. syriacus has a similar number of eggs to A. rusticus and faster larval development; these could be key features to explain the coexistence of these two species.
Las invasiones biológicas son una de las principales amenazas para la salud de los bosques. Aunque Arhopalus rusticus y Arhopalus syriacus (Cerambycidae) son especies exóticas en América del Sur, la ecología de estas especies invasoras no está bien estudiada. En este estudio examinamos el efecto del tamaño de la hembra sobre la fecundidad potencial, tamaño y número de huevos maduros en ovarios en ambos insectos plaga. Asimismo, se compararon las características morfométricas del huevo, medidas de las larvas, y el desarrollo y la supervivencia larval entre ambas especies. Se observó una relación positiva entre el tamaño de la hembra y el número de huevos en sus ovarios, pero no así una relación entre el tamaño de la hembra y las características morfométricas de los huevos. Las hembras de A. rusticus fueron más grandes y tuvieron huevos más grandes que A. syriacus, pero no se observó una diferencia significativa en el número de huevos. La longitud de la larva y el ancho de la cápsula encefálica fueron mayores en A. rusticus. Arhopalus rusticus tuvo la tasa de supervivencia larval más alta, pero A. syriacus tuvo el período larval más corto. Concluimos que el tamaño de las hembras y los huevos, así como el tamaño de las larvas y la supervivencia son ventajas competitivas para A. rusticus. Sin embargo, A. syriacus tiene un número similar de huevos al de A. rusticus aunque un desarrollo larval más rápido, características que podrían ser clave para explicar la coexistencia de estas dos especies.
Referencias
Altman DG. 1991. Practical Statistics for Medical Research. London: Chapman and Hall.
Bense U. 1995. Longicorn Beetles: Illustrated Key to the Cerambycidae and Vesperidae of Europe. Weikersheim: Margraf Verlag.
Berrigan D. 1991. The allometry of egg size and number in insects. Oikos 60(3):313–321. doi: https://dx.doi.org/10.2307/3545073.
Brockerhoff EG, Hosking, GP. 2001. Arhopalus tristis (F.) [= Arhopalus ferus (Mulsant)] (Coleoptera: Cerambycidae), Burnt pine longhorn. Forest and Timber Insects in New ZealandNo. 27. Forest Research, Rotorua, New Zealand.
Cerezke HF. 1977. Characteristics of damage in tree-length white spruce logs caused by the white-spotted sawyer, Monochamus scutellatus. Can. Jour. For. Res. 7(2):232–240.
Chapman RF. 1998. The insect: Structure and Function. Cambridge: University Press.
Ciach M, Michalcewicz J. 2009. Egg morphology of Rosalia alpina (Linnaeus, 1758) (Coleoptera: Cerambycidae) from southern Poland. Entomol. News. 120(1):61–64. doi: https://doi.org/10.3157/021.120.0112.
Ciach M, Michalcewicz J. 2013. Correlation between selected biometric traits of adult Rosalia alpina (L.) (Coleoptera: Cerambycidae) and size of their exit holes: new perspectives on insect studies? Pol. J. Ecol. 61(2):349–355.
Ciesla W. 2011. Forest Entomology: A global perspective. Oxford: John Wiley & Sons. Carle P. 1969. Milieux artificiels pour l’élevage des larves de Pissodes notatus L. (Col. Curculionidae) et autres xylophages du Pin maritime. Ann. des Sci. Forest 26(3):397–406. doi: https://dx.doi.org/10.1051/forest/19690304.
De Groot P, Nott R. 2001. Evaluation of traps of six different designs to capture pine sawyer beetles (Coleoptera: Cerambycidae). Agric. For. Entomol. 3(2):107–111. doi: https://dx.doi.org/10.1046/j.1461-9563.2001.00087.x.
Di Iorio OR. 2004. Especies exóticas de Cerambycidae (Coleoptera) introducidas en Argentina. Parte 2. Nuevos registros, plantas huéspedes, periodos de emergencia, y estado actual. Agrociencia 38(6):663–678.
Di Rienzo JA, Casanoves F, Balzarini MG, Gonzalez L, Tablada M, Robledo CW. c2017. InfoStat versión 2017. Grupo InfoStat, FCA, Universidad Nacional de Córdoba, Argentina. [last ac-cessed: 10 Apr 2017]. http://www.infostat.com.ar
De Viedma MG, Notario A, Baragano JR. 1985. Laboratory rearing of lignicolous Coleoptera (Cerambycidae). J. Econ. Entomol. 78(5):1149–1150. doi: https://dx.doi.org/10.1093/jee/78.5.1149.
Ernsting G, Isaaks A. 2000. Ectotherms, temperature, and trade offs: size and number of eggs in a carabid beetle. Am. Nat. 155(6):804–813. doi: https://dx.doi.org/10.1086/303361.
Fachinetti R, Pedemonte ML, Grilli MP. 2015. Tiempo de desarrollo y supervivencia de Arhopalus syriacus (Reitter) (Coleoptera: Cerambycidae), una plaga potencialmente perjudicial de Pinus sp. en Argentina. AgriScientia 32(2):95–105. doi: https://dx.doi.org/10.31047/1668.298x.v32.n2.16560.
Fox CW. 1993. Multiple mating, lifetime fecundity and female mortality of the bruchid beetle Callosobruchus maculatus (Coleoptera: Bruchidae). Funct. Ecol. 7(2):203–208. doi: https://dx.doi.org/10.2307/2389888.
Fox CW. 1994. The influence of egg size on offspring performance in the seed beetle, Callosobruchus maculatus. Oikos 71(2):321– 325. doi: https://dx.doi.org/10.2307/3546280.
Fox CW, Czesak ME. 2000. Evolutionary ecology of progeny size in arthropods. Annu. Rev. Entomol. 45(1):341–369. doi: https://dx.doi.org/10.1146/annurev.ento.45.1.341.
Galford JR. 1969. Artificial rearing of 10 species of wood-boring insects. Research Note NE-102. Upper Darby, PA: U.S. Department of Agriculture, Forest Service, Northeastern Forest Exper-iment Station.
Hernández JM. 1990. Descripción del huevo de las especies del género Iberodorcadion (Breunning, 1943) endémicas de la Sierra de Guadarrama (España) (Coleoptera, Cerambycidae). Bol. R. Soc. Esp. Hist. Nat., Secc. Biol. 86(1–4):161–179.
Honek A. 1993. Intraspecific variation in body size and fecundity in insects: a general relationship. Oikos 66(3):483–492. doi: https://dx.doi.org/10.2307/3544943.
Hosking GP, Bain J. 1977. Arhopalus ferus (Coleoptera: Cerambycidae); its biology in New Zealand. N. Z. J. For. Sci. 7(1):3–15.
Iglesias C, Notario A, Baragaño JR. 1989. Evaluación de las condiciones de cría y datos bionómicos de coleópteros lignícolas de tocón de pino. Bol. San. Veg. Plagas 15(1):9–16.
Ito K, Matsumoto K, Sato T. 1992. Effects of female adult size on progeny size and survival of the cryptomeria bark borer, Semanotus japonicus Lacordaire. Trans. Kansai Br. Jpn. For. Soc. 1:261–264.
Jikumaru S, Togashi K, Taketsune A, Takahashi E. 1994. Oviposition biology of Monochamus saltuarius (Coleoptera: Cerambycidae) at a constant temperature. Appl. Entomol. Zool. 29(4):555–561. doi: https://dx.doi.org/10.1303/aez.29.555.
Juliano SA. 1985. The effects of body size on mating and reproduction in Brachinus lateralis (Coleoptera: Carabidae). Ecol. Entomol. 10(3):271–280. doi: https://dx.doi.org/10.1111/j.1365-2311.1985.tb00724.x.
Kato K, Yamada H, Shibata E. 2000. Role of female adult size in reproductive fitness of Semanotus japonicus (Coleoptera: Cerambycidae). Appl. Entomol. Zool. 35(3):327–331. doi: https://dx.doi.org/10.1303/aez.2000.327.
Keena MA. 2005. Pourable artificial diet for rearing Anoplophora glabripennis (Coleoptera: Cerambycidae) and methods to optimize larval survival and synchronize development. Ann. Entomol. Soc. Am. 98(4):536–547. doi: http://dx.doi.org/10.1603/0013-8746(2005)098%5B0536:PADFRA%5D2.0.CO;2.
Keena MA. 2017. Laboratory rearing and handling of cerambycids. In: Wang Q, editor. Cerambycidae of the world: biology and pest management. CRC Press. p. 253–289.
Kojima W. 2015. Variation in body size in the giant rhinoceros beetle Trypoxylus dichotomus is mediated by maternal effects on egg size. Ecol. Entomol. 40(4):420–427. doi: https://doi.org/10.1111/een.12205.
Kolk A, Starzyk JR. 1996. Long-horn beetle Arhopalus rusticus (L.). The Atlas of Forest Insect Pests. Multico Warszawa. Poland: The Polish Forest Research Institute.
Lander TA, Klein EK, Oddou-Muratorio S, Candau JN, Gidoin C, Chalon A, Roig, A, Fallour D, Auger-Rozenberg MA, Boivin T. 2014. Reconstruction of a windborne insect invasion using a particle dispersal model, historical wind data, and Bayesian analysis of genetic data. Ecol. Evol. 4(24):4609–4625. doi: https://dx.doi.org/10.1002/ece3.1206.
Lawrence WS. 1990. Effects of body size and repeated matings on female milkweed beetle (Coleoptera: Cerambycidae) reproductive success. Ann. Entomol. Soc. Am. 83(6):1096–1100. doi: https://dx.doi.org/10.1093/aesa/83.6.1096.
Liebhold AM, Halverson JA, Elmes GA. 1992. Gypsy moth invasion in North America: a quantitative analysis. J. Biogeogr. 19(5):513–520. doi: https://dx.doi.org/10.2307/2845770.
López A, García J, Demaestri M, Di Iorio O, Magris R. 2008. The genus Arhopalus Serville, 1834 (Insecta: Coleoptera: Cerambycidae: Aseminae) in association to Sirex noctilio in Argentina. Bol. San. Veg. Plagas 34(4):529–531.
Mack RN, Simberloff D, Lonsdale WM, Evans H, Clout M, Bazzaz FA. 2000. Biotic invasions: causes, epidemiology, global consequences, and control. Ecol. Appl. 10(3):689–710. doi: https://dx.doi.org/10.2307/2641039.
Notario A, Baragaño R. 1978. Ergates faber Linnaeus (Col. Cerambycidae): descripción, cría artificial y estudio cariológico. Anales Inst. Nac. Invest. Agrarias (I.N.l.A.) Serie: Protección Vegetal 8:45–57.
Pedemonte ML. 2014. Efecto de la conectividad del paisaje, el tamaño y la calidad del parche sobre la abundancia poblacional de dos especies de insectos herbívoros. [Tesis]. [Córdoba]: Universidad Nacional de Córdoba, Argentina.
Rassati D, Toffolo EP, Battisti A, Faccoli M. 2012. Monitoring of the pine sawyer beetle Monochamus galloprovincialis by pheromone traps in Italy. Phytoparasitica 40(4):329–336. doi: https://dx.doi.org/10.1007/s12600-012-0233-5.
Rogers DJ, Lewthwaite SE, Dentener PR. 2002. Rearing huhu beetle larvae, Prionoplus reticularis (Coleoptera: Cerambycidae) on artificial diet. New Zeal. J. Zool. 29(4):303–310. Doi: https://dx.doi.org/10.1080/03014223.2002.9518314.
Smith CC, Fretwell SD. 1974. The optimal balance between size and number of offspring. Am. Nat. 108(962):499–506. doi: https://dx.doi.org/10.1086/282929.
Suckling DM, Gibb AR, Daly JM, Chen X, Brockerhoff EG. 2001. Behavioral and electrophysiological responses of Arhopalus tristis to burnt pine and other stimuli. J. Chem. Ecol. 27(6):1091–1104. doi: https//dx.doi.org/10.1023/A:1010355710509.
Togashi K. 1997. Lifetime fecundity and body size of Monochamus alternatus (Coleoptera: Cerambycidae) at a constant temperature. Jpn. J. Entomol. 65(3):458–470.
Togashi K. 2007. Lifetime fecundity and female body size in Paraglenea fortunei (Coleoptera: Cerambycidae). Appl. Entomol. Zool. 42(4):549–556. doi: https://dx.doi.org/10.1303/aez.2007.549.
Togashi K, Akita Y, Nakane I, Shibata Y, Nakai I. 1997. Relatively larger eggs produced by smaller females of Monochamus alternatus (Coleoptera: Cerambycidae). Appl. Entomol. Zool. 32(1):264–266. doi: https://dx.doi.org/10.1303/aez.32.264.
Webb GA, Eldridge RH. 1997. Arhopalus syriacus (Reitter) (Coleoptera: Cerambycidae): a potential economic pest of Pinus in Australia, with notes on its biology and distribution. Aust. For. 60(2):125–129. doi: https://dx.doi.org/10.1080/00049158.1997.10674707.
Yanagi S, Tuda M. 2012. Female size constrains egg size via the influence of reproductive organ size and resource storage in the seed beetle Callosobruchus chinensis. J. Insect Physiol. 58(11):1432–1437. doi: https://dx.doi.org/10.1016/j.jinsphys.2012.08.007.
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. Romina Fachinetti, Mariano Pablo Grilli. (2022). Biological traits and field distribution of introduced Arhopalus species in Central Argentina. International Journal of Pest Management, 68(3), p.274. https://doi.org/10.1080/09670874.2020.1818871.
2. Qiang Huang, Sheina B Sim, Scott M Geib, Anna Childers, Junfeng Liu, Xiuxiu Wei, Wensu Han, Francisco Posada-Florez, Allen Z Xue, Zheng Li, Jay D Evans. (2022). Identification of sex chromosomes and primary sex ratio in the small hive beetle, a worldwide parasite of honey bees. GigaScience, 12 https://doi.org/10.1093/gigascience/giad056.
3. Juliano de Bastos Pazini, Anderson Dionei Grützmacher, Enio Júnior Seidel, Aline Costa Padilha, Fernando Felisberto da Silva, Daniel Bernardi, Michael Orrin Way, José Francisco da Silva Martins, Frank Peairs. (2022). Field Assessment of Oryzophagus oryzae (Coleoptera: Curculionidae) Preference and Performance on Selected Rice Cultivars. Journal of Economic Entomology, 115(2), p.671. https://doi.org/10.1093/jee/toac003.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2019 Caldasia

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
Aquellos autores/as que tengan publicaciones con esta revista, aceptan los términos siguientes:
- Los autores/as conservarán sus derechos de autor y garantizarán a la revista el derecho de primera publicación de su obra, el cual estará simultáneamente sujeto a la Licencia de reconocimiento de Creative Commons que permite a terceros compartir la obra siempre que se indique su autor y su primera publicación esta revista.
- Los autores/as podrán adoptar otros acuerdos de licencia no exclusiva de distribución de la versión de la obra publicada (p. ej.: depositarla en un archivo telemático institucional o publicarla en un volumen monográfico) siempre que se indique la publicación inicial en esta revista.
- Se permite y recomienda a los autores/as difundir su obra a través de Internet (p. ej.: en archivos telemáticos institucionales o en su página web) antes y durante el proceso de envío, lo cual puede producir intercambios interesantes y aumentar las citas de la obra publicada. (Véase El efecto del acceso abierto).























