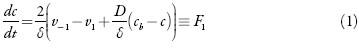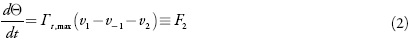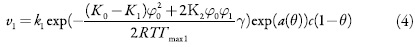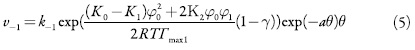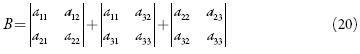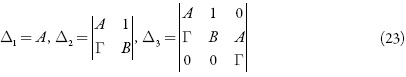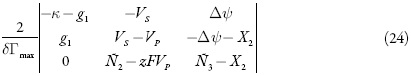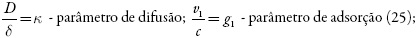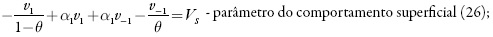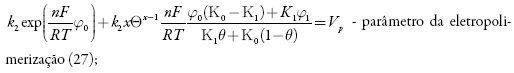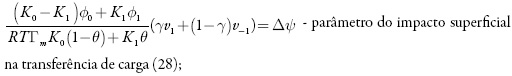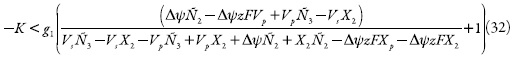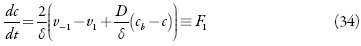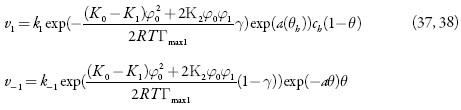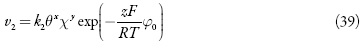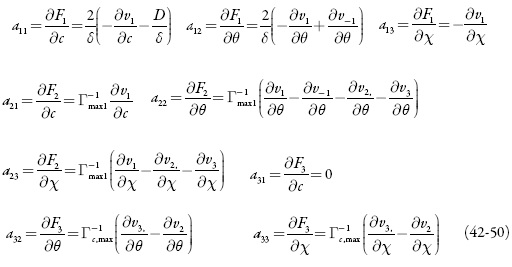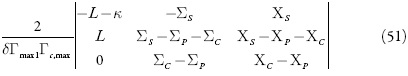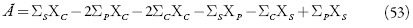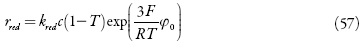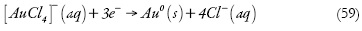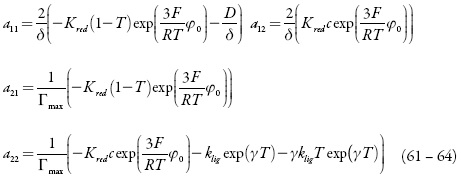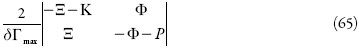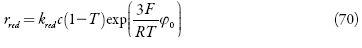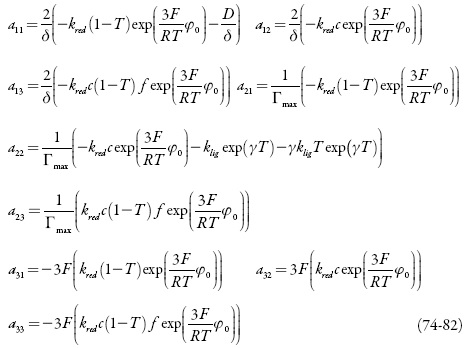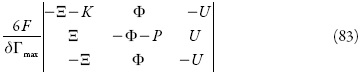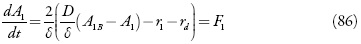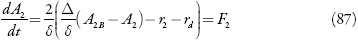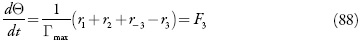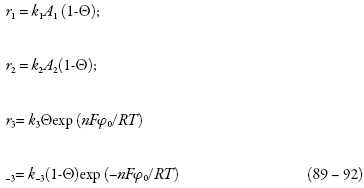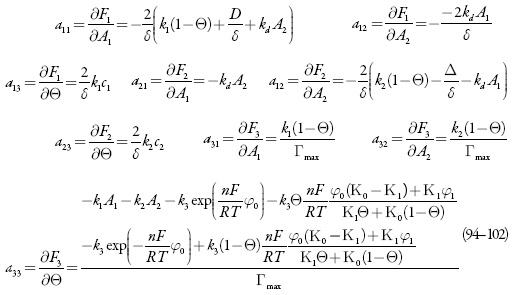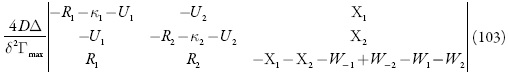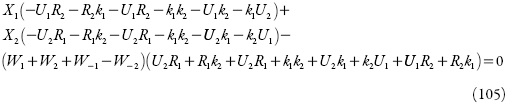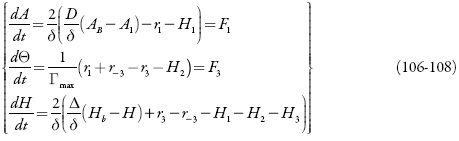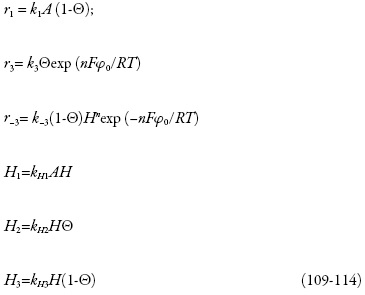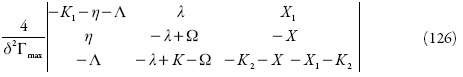Estudo de estabilidade da eletrossíntese e do desempenho eletroanalítico do compósito poli(l-metionina)-nano-Au na presença de dopamina e de ácido úrico
The stability study of the electrochemical synthesis and electroanalytical function of poly(l-methionine)-nano-Au composite in the presence of dopamine and uric acid
DOI:
https://doi.org/10.15446/rcciquifa.v45n3.62027Palabras clave:
Metionina, nanocompósitos, eletropolimerização, polieletrólitos, estado estacionário estável (pt)Methionine, nanocomposites, electropolymerization, polyelectrolytes, stable steady-state (en)
Descargas
A eletrossíntese e o desempenho eletroanalítico do compósito poli-L-metionina -nano Au foram descritos matematicamente, sendo a primeira descrita etapa por etapa. Os modelos matemáticos foram analisados por meio da teoria de estabilidade linear e da análise de bifurcações. A partir da análise foram inferidas as condições do estado estacionário estável (do modo mais confortável para a eletropolimerização, e do melhor desempenho do sensor), bem como das instabilidades oscilatória e monotônica. As conclusões da modelagem vão ao encontro do observado experimentalmente, completando também o conhecimento do processo
The electrosynthesis and the electroanalytic function of the poly-L-methioninenano-Au composite were mathematically described and the first one of the processes was described stage by stage. The mathematical models were analyzed by linear stability theory and bifurcation analysis. Basing on the analysis, the stable steady-state conditions, like also oscillatory and monotonic instability conditions were inferred.
https://doi.org/10.15446/rcciquifa.v45n3.62027
Estudo de estabilidade da eletrossíntese e do desempenho eletroanalítico do compósito poli(l-metionina)-nano-Au na presença de dopamina e de ácido úrico
The stability study of the electrochemical synthesis and electroanalytical function of poly(l-methionine)-nano-Au composite in the presence of dopamine and uric acid
Volodymyr V. Tkach1,2*, Sílvio C. de Oliveira2, Yana G. Ivanushko1, Svitlana M. Lukanova1, Reza Ojani3, Petró I. Yagodynets´1, Aline M. da Rocha4
1 Universidade Nacional de Chernivtsi, 58012, Rua de Kotsyubyns'ky, 2, Chernivtsi, Ucrânia
2 Universidade Federal de Mato Grosso do Sul, Av. Sen. Felinto. Müller, 1555, C/P. 549, 79074460, Campo Grande, MS, Brasil
3 Universidade de Mazandarã, 47416-95447, 3o km. da Rodovia de Forças Aéreas Iranianas, Babolsar, República Islâmica do Irã
4 Universidade Federal de Minas Gerais, 31270-901, Av. Pres. Antônio Carlos, 6627, Pampulha, Belo Horizonte, Brasil
* Correio eletrônico: nightwatcher2401@gmail.com
Recebido em: 22 de dezembro de 2015 Aceito em: 09 de novembro de 2016
Resumo
A eletrossíntese e o desempenho eletroanalítico do compósito poli-L-metionina -nano Au foram descritos matematicamente, sendo a primeira descrita etapa por etapa. Os modelos matemáticos foram analisados por meio da teoria de estabilidade linear e da análise de bifurcações. A partir da análise foram inferidas as condições do estado estacionário estável (do modo mais confortável para a eletropolimerização, e do melhor desempenho do sensor), bem como das instabilidades oscilatória e monotônica. As conclusões da modelagem vão ao encontro do observado experimentalmente, completando também o conhecimento do processo.
Palavras-chave: metionina, nanocompósitos, eletropolimerização, polieletrólitos, estado estacionário estável.
Summary
The electrosynthesis and the electroanalytic function of the poly-L-methioninenano-Au composite were mathematically described and the first one of the processes was described stage by stage. The mathematical models were analyzed by linear stability theory and bifurcation analysis. Basing on the analysis, the stable steady-state conditions, like also oscillatory and monotonic instability conditions were inferred.
Key words: methionine, nanocomposites, electropolymerization, polyelectrolytes, stable steady-state.
Introdução
Dopamina, que é uma das três catecolaminas naturais, desempenha um papel importante como neurotransmissor nos sistemas cardiovascular, hormonal, renal e nervoso central [1-3]. Os seus níveis extremos podem causar deficiências de metabolismo, bem como as doenças mentais, como esquizofrenia ou Parkinson. Destarte, a medição das suas concentrações faz-se tarefa importante.
Ácido úrico, além de ser o produto final de metabolismo de purinas no organismo humano, é também o produto principal de metabolismo de algumas espécies de animais, como peixes, anfíbias e répteis [4], a sua secreção pelo organismo é um processo geneticamente determinado [5]. A sua falta constitui hipouricemia, associada a síndro -mes como a doença de Wilson [6], ou a síndrome de Fanconi [7]. Já a hiperfunção de ácido úrico pode causar doenças, associadas à sua presença excessiva como doença de Lesch-Nyhan [8]. Assim sendo, a determinação da sua concentração também é uma tarefa atual.
Diante do exposto, um método analítico sensível e exato, capaz de detectar ambos os compostos será útil não só para a investigação fisiológica de organismos humanos e animais, mas também para a diagnóstica temporã na medicina humana e veterinária [9].
Para a detecção dos mencionados compostos são frequentemente usadas as técnicas voltamperométricas [10]. No entanto, a eletrooxicação simultânea de dopamina e ácido úrico sobre os elétrodos não revestidos acontece no mesmo potencial. Destarte, os picos nos voltamperogramas se coincidem, sendo impossível discriminá-los [11-13].
Poder-se-ia resolver o problema, aumentando a seletividade, usando os elétrodos, modificados química e eletroquimicamente por polímeros [14-23], especialmente sendo dopados por DNA [23], sistemas coloidais dopados por corantes [24], óxidos metálicos [25], nanotubos de carbono [26-28], heteropoliânions [29, 30] e nanopartículas metálicas [31-36]. As nanopartículas metálicas recebem atenção por terem área ativa extensa (especialmente as depositadas eletroquimicamente sobre superfícies modificas), transporte efetivo de massas e alta atividade eletrocatalítica [36-38]. Porém, as nanopartículas metálicas podem ser agir fracamente na ausência de um material condutor estabilizador, que também desempenha as funções de mediador de transferência eletrônica. Para resolver este problema, usam-se os materiais estabilizadores, como os ligantes ou polímeros, sintetizados química ou eletroquimicamente [39-45]. A estrutura porosa de polímeros condutores deixa que as nanopartículas se dispersem na matriz polimérica, formando-se os novos sítios ativos [46-50].
As nanopartículas de ouro podem aderir-se fortemente à superfície, formando ligações covalentes com os grupos funcionais como nitrila, amina ou tiólico [51-53]. Foi mostrado que os polímeros que contêm unidades tiofênicas ou metionínicas fortemente interagem com as nanopartículas de ouro [54-55]. Porém, o uso de monômeros tio-fênicos pode ser restrito haja vista o "paradoxo de politiofeno", o fenômeno da concorrência entre a eletropolimerização e sobreoxidação do polímero formado [56-58], possível também para alguns outros monômeros e que é nociva tanto para morfologia e condutividade do polímero, quanto ao desempenho do futuro sensor.
Além disso, para tal comportamento, bem como para os mais sistemas com eletropolimerização [59-66], modificação e desempenho de sensores, baseados em polímeros condutores [67-72] são possíveis as instabilidades eletroquímicas, que afetam negativamente a clareza da resposta do sensor, a resposta do equipamento eletroquímico, a morfologia do polímero condutor resultante.
A determinação da causa destas instabilidades constitui um dos problemas mais difíceis de físico-química. Anteriormente, elas obtiveram explicação fenomenológica, que, apesar de ter fundamento lógico não tinha forte base teórica e só valia para um sistema concreto. Para o melhor conhecimento do mecanismo da confecção e do desempenho de compostos sensórios, da descrição do melhor modo da confecção e das margens do melhor desempenho, bem como para o descobrimento exato da causa das possíveis desvios do padrão, causados pelas instabilidades, é preciso construir um modelo matemático, capaz de descrever adequadamente os processos no sistema.
No trabalho [73] foi descrito o sensor eletroquímico de dopamina e ácido úrico, baseado no compósito de poli-(L-metionina) e nanopartículas de ouro. A sua confecção foi feita em 2 etapas:
- Eletropolimerização de L-metionina;
- Eletrodeposição de nanopartículas de ouro.
Depois foi testado o desempenho do compósito no processo da determinação eletro-química dos mencionados compostos. Neste trabalho, os modelos matemáticos vão ser desenvolvidos para todos os três processos mencionados, sendo ele a continuação da pesquisa, descrita nos trabalhos [73-84]. Nos trabalhos [74-84] tem sido feita a tentativa da modelagem matemática dos processos da eletrossíntese e de modificação de polímeros condutores conjugados (com a ênfase no desempenho de sensores e biossensores, baseados em polímeros condutores).
O caso de metionina é diferente, pois é o polímero condutor doutro tipo (neste caso, polieletrólito, que não é conjugado e isto explica o fato de o potencial de polimerização ser superior que no caso dos compostos heterocíclicos aromáticos), que também é capaz de formar sais e complexos que dificultam a eletropolimerização. É mais uma causa da escolha do elétrodo de carbono vítreo em vez do metálico. O fato de o polímero ser um polieletrólito também explica a dependência do pH para o comportamento do compósito com respeito ao seu desempenho.
Seção I. Eletropolimerização
Caso da eletropolimerização sobre o carbono vítreo
A eletrossíntese de polímeros condutores por meio de eletropolimerização pode ser feita de três modos: potenciodinâmico, galvanostático e potenciostático.
Se a eletropolimerização for realizada no modo potenciodinâmico (como foi feito em [73]), serão obtidos os filmes finos cujas características dependem das janelas de potencial escolhidas para a obtenção do revestimento. Além disso, encontrar-se-ão muitas dificuldades. A morfologia do polímero não será satisfatória, haja vista que o polímero resultante será parcialmente sobreoxidado. Além disso, durante a eletropolimerização potenciodinâmica também são possíveis os "pontos mortos", nos quais a polimerização não ocorre durante algum tempo.
A eletropolimerização galvanostática pode ser realizada (e se realiza com bastante frequência), mas é difícil determinar a densidade da corrente mais adequada à eletropolimerização do monômero. E, ainda por cima, a corrente que circula pode sobreoxidar o polímero resultante, como no caso da eletropolimerização potenciodinâmica.
A polimerização potenciostática é a mais frequentemente usada pois é muito fácil manter o potencial do eletrodo próximo ao pico do voltamperograma cíclico feito nas condições da eletropolimerização.
Para todos os três modos, os modelos matemáticos vêm sendo propostos nos trabalhos [74, 75] e no caso do aminoácido no eletrodo de carbono vítreo, todas elas funcionam (menos o modelo do crescimento da cadeia, descrito em [74] para compostos aromáticos). Vamos mostrar o modelo em detalhes para o modo potenciodinâmico usado no trabalho [73].
Para a descrição da eletropolimerização no modo potenciodinâmico (supondo a diferença de potenciais do ânodo e do cátodo constantes), usamos três variáveis:
c - a concentração do monômero na camada pré-superficial;
- o grau do recobrimento da superfície do elétrodo pelo monômero;
q - a carga do ânodo.
Nos modelos, descritos neste artigo, para simplificar a modelagem, supõe-se que:
- o eletrólito de suporte esteja em excesso - destarte podemos menosprezar o fluxo de migração, bem como as despesas do dopante;
- o reator esteja sendo agitado intensamente - assim podemos menosprezar o fluxo causado por convecção;
- a distribuição concentracional do monômero na seção 1, dos íons metálicos na seção 2 e do analíto na seção 3 na camada pré-superficial seja linear e a espessura da camada, constante e igual a δ.
O monômero entra na camada pre-superficial difundindo-se no interior da solução e dessorvendo-se da superfície e sai da camada adsorvendo para a superfície de elétrodo. Então, a equação do balanço pode ser descrita como
Sendo v-1 e v1 as velocidades de dessorção e adsorção, D o coeficiente da difusão do monômero e cb a concentração do monômero no interior da solução.
O monômero chega até a superfície adsorvendo-se sobre ela e sai dela dessorvendo-se. Ele também é consumido durante a sua polimerização. A equação do balanço do monômero (eq. 6) na superfície do catodo pode-se descrever como.
Sendo Tt,max a concentração superficial maxima do monômero e v2 a velocidade da sua eletropolimerização.
Para o ânodo aplica-se a densidade da corrente conveniente para o processo. O valor desta corrente diminui para que ocorra a reação eletroquímica. Nas condições do modo potenciodinâmico, a equação de balanço de carga pode ser descrita como:
sendo U a diferença de potenciais do ânodo e do cátodo, A a área do eletrodo e R a resistência da superfície, iF é a corrente faradaica.
As velocidades dos processos respectivos e a corrente de Faraday podem calcular-se como:
em que k1 e k-1 são as constantes de adsorção e dessorção do monômero, K1 eK0 significam respectivamente as capacidades elétricas das regiões das camadas, pré-superficial que se referem à superfície coberta pelo monômero e à superfície livre, (φ1 é o salto de potencial relativo ao potencial de carga zero na região da dupla camada (CED), relativa à região da superfície coberta pelo monômero e φ0 significa o salto de potencial relativo ao potencial de carga zero na região da CED relativa à superfície livre e γ é a constante (γ < 1) que descreve o grau de modificação da capacidade da CED durante a adsorção, k2 a constante da reação, x a ordem da reação do monômero, z a quantidade dos elétrons transferidos, F a constante de Faraday, R é a constante universal dos gases e T é a temperatura absoluta da solução.
Resultados e discussão (subseção 1.1.)
O comportamento do sistema eletroquímico será investigado por meio da análise do sistema de equações diferenciais (1-3) reformuladas, haja visto as relações algébricas (4-7). A matriz funcional de Jacobi, cujos elementos vêm descritos para o estado estacionário, como:
sendo:
O estado estacionário estável: para determinar as condições do estado estacionário estável, vamos usar o critério de Routh e Hurwitz. A equação característica do sistema de equações (1-3) pode ser descrita como:
sendo que:
O critério de Routh e Hurwitz requer que, para um estado estacionário estável, os minores dos membros da diagonal principal da matriz de Hurwitz.
sejam positivos. Os menores dos membros da diagonal principal da matriz:
Visto que Δ3= ΓΔ2, podemos formar a condição do estado estacionário estável como Γ>0.
Para simplificar a análise de Jacobiano, é introduzido novas variáveis de modo que o jacobiano será descrito como:
sendo:
Levando em consideração o critério de estabilidade e as expressões (24-31), resolve-se a inequação Det J < 0 e obtém-se a condição de estabilidade do estado estacionário como:
Pode-se concluir que para a estabilidade do estado estacionário contribuem:
- alta velocidade da difusão (altos valores de k);
- alta velocidade-padrão de adsorção (altos valores de g1);
- alto valor da velocidade-padrão da eletropolimerização (altos valores da Vp), acompanhado pelas influências na dupla camada dos processos superficiais (valores negativos de Δψ) ou da eletropolimerização (negatividade do parâmetro Vp);
- as influências das mudanças cíclicas da superfície não dão um impacto importante à estabilidade do estado estacionário.
A equivalência dos efeitos favoráveis à estabilidade e à instabilidade do estado estacionário leva à aparição da instabilidade monotônica (ou instabilidade de sela e nó), cuja condição principal pode ser descrita como:
Para a instabilidade oscilatória ser realizada, é preciso (não sendo suficiente) que a diagonal principal da matriz de Jacobi contenha os elementos positivos, pois eles descrevem a positiva ligação de retorno, ou seja, autocatálise no sentido mais amplo do termo.
Na diagonal principal tem 3 elementos, capazes de ser positivos, os dois estão na célula α22, o que será no caso de:
- o elemento VS ser positivo;
- o elemento Vp ter valores negativos.
Mais um está na α33 para o caso de o elemento Ñ3 ser positivo.
O primeiro caso é o da interação lateral de partículas adsorvidas, que promove o crescimento da cadeia, mas não faz parte do mecanismo. A interação lateral pode ser promovida pela:
- interação spin-spin dos radical-cátions;
- interação eletrostática dos íons bipolares.
O segundo caso descreve as influências na dupla camada elétrica da eletropolimerização, aliás, dos processos da eletro-oxidação que a acompanham. Tais influências usualmente se manifestam quando ocorre o recarregamento da camada durante a formação do filme polifuncional, no momento da eletro-oxidação dos redutores fortes.
O terceiro caso descreve o comportamento oscilatório durante a formação do polímero passivo de alta resistência e de baixa capacidade de depositar a carga elétrica.
Desta forma em geral, o comportamento do sistema com a eletropolimerização do aminoácido sobre o carbono vítreo não difere dos outros casos de eletropolimerização, sendo porém, mais complicado.
Caso da eletropolimerização sobre metais ativos
Alguns dos monômeros (inclusive aminoácidos) podem acomplicar a sua eletropolimerização sobre certos metais por causa da formação de complexos, sendo este fenômeno observado na modificação electroquímica, bem como na electropolimerização [85, 86]. Para o processo da electromodificação este processo tem sido considerado nocivo, porém, para o caso da eletropolimerização a formação de complexos contribui muito na adesão do filme polimérico, na condutividade, na cor e nas propriedades eletrocrômicas.
Assim sendo, a escolha do carbono vítreo como o material do ânodo em vez de uma supefície metálica como aço ou alumínio é explicada também pela possível formação de complexos, cuja formação diminui a quantidade dos sítios ativos de ligação com as nanopartículas de ouro.
Para descrever matematicamente a eletropolimerização potenciostática do aminoácido sobre metais de transição, usar-se-ão as 3 variáveis:
c - a concentração do aminoácido na camada pré-superficial;
θ - o grau de recubrimento da superfície pelo monômerio;
X - o grau de recubrimento da superfície pelo complexo do monômero, supondo o suposto acima.
O monômero entra na camada pre-superficial difundindo a ele do interior da solução e dessorvendo-se da superfície e sai da camada adsorvendo para a superfície de elétrodo. Então, a equação do balanço (eq. 5) pode ser descrita conforme:
Sendo v1 e v-1 as velocidades da adsorção e da dessorção do monômero, D o coeficiente da difusão e cb a concentração do monômero no interior da solução.
O monômero entra na superfície por meio da adsorção e sai por meio da sua dessorção. Também ele participa da eletrocopolimerização com o seu próprio complexo e da formação do próprio. Destarte, a equação de balanço será descrita como:
sendo Γt,max a concentração máxima superficial do monômero, v2 a velocidade da eletropolimerização e v3 a da formação do complexo.
O complexo é formado por meio da reação entre o monômero e a superfície metálica e participa da eletrocopolimerização com o próprio monômero. Destarte, a equação de balanço será descrita como
Sendo Γx max a concentração superficial máxima do complexo.
As velocidades da adsorção e da dessorção do monômero, da sua eletropolimerização e da formação do complexo podem descrever-se como:
sendo β1 o coeficiente da interação entre o monômero e a superfície metálica, x e γ as ordens de reações, obtidas por meio da aplicação das leis de Mayo-Lewis da eletrocopolimerização.
O comportamento do sistema será estudado por meio da análise do sistema de equações (34-36), sendo tomadas em conta as relações algébricas (37-40) por meio da teoria de estabilidade lineal. A matriz funcional de Jacobi para o sistema (34-36) será descrito como:
sendo que:
Para simplificar a análise da matriz, introduzimos as novas variáveis para o determinante obter a forma
Aplicando o critério de Routh-Hurwitz obtém-se a condição da estabilidade do estado estacionário de forma seguinte:
sendo:
A condição pode ser interpretada de seguinte forma. O estado estacionário tende a ser estável no caso de a reação ser "cineticamente controlada", ou seja, quando a velocidade da difusão é superior à dos processos químicos. Outrossim, os valores mais altos das velocidades da eletropolimerização e da formação de complexos em relação às velocidades de adsorção e dessorção favorecem a estabilidade.
Ao serem iguais as influências mencionadas, realiza-se a bifurcação de sela e nó e realiza-se a instabilidade monotônica, a condição de cuja aparição é:
Usando a metodologia apresentada acima, pode-se ver que o comportamento oscilatório pode ser descrito pela positividade de seguintes elementos da diagonal principal da matriz e causado pelos seguintes fatores:
- Σs nas condições da atração entre partículas adsorvidas,
- Σp no caso do rearranjo da dupla camada durante a eletropolimerização (φ0<0)
- Xc no caso da atração entre o monômero e a superfície metálica.
Se as duas primeiras são características para todos os processos da eletropolimeriza-ção, a terceira é específica para os sistemas com a formação de complexos na superfície. Assim, o fator mencionado é responsável pelo comportamento oscilatório aquando da eletropolimerização, acompanhada pela formação de complexos.
Seção II. Eletrodeposição
Modo potenciostático
Segundo o artigo [73], na síntese do revestimento sensitivo, após a eletropolimerização do monômero, neste caso do aminoácido metionina, ocorreu a deposição catódica de nanopartículas de ouro.
Outro jeito de modificar poderia ser a intercalação delas para dentro da matriz polimérica por meio da dopagem. Porém, se tal processo fosse feito a matriz polimérica bloquearia parcialmente o contato de partículas intercaladas com o analito. Destarte ocorreu a deposição catódica de nanopartículas de ouro com a próxima ligação delas com o polieletrólito depositado.
Embora as instabilidades ocorram mais frequentemente nos processos anódicos, nos processos catódicos [87] também podem ocorrer oscilações eletroquímicas, multiplicidade de estados estacionários e outros. Assim sendo, isso leva a pensar que no caso da eletrodeposição descrita em [73], sob algumas condições, talvez não mencionadas no trabalho, podem ocorrer as instabilidades de estados estacionários.
Como a eletrodeposição ocorreu sem adsorção do íon complexo de ouro, para a descrição matemática do processo no modo potenciostático (sendo o valor concreto do potencial catódico mencionado no trabalho referente) usam-se as duas variáveis (usar-se-iam as 3, caso a eletrorredução fosse precedida pela adsorção):
c - a concentração dos íons complexos AuCl4-;
T - o grau do revestimento da superfície do eletrodo pelas nanopartículas de ouro, haja vista o pressuposto acima.
Os íons complexos difundem-se para a camada pré-superficial e, após isso, são eletrorreduzidos formando o metal sob a forma de nanopartículas. Posto assim, a equação de balanço dos íons complexos de ouro (III) será descrita como:
sendo rred a velocidade de deposição catódica, c0 a concentração dos íons complexos no interior da solução.
As nanopartículas metálicas formam-se aquando da eletrorredução do ânion AuCLf e, depois, formam ligações covalentes com o polieletrólito depositado. Destarte, a equação de balanço será descrita como:
sendo Γmax a concentração máxima de partículas metálicas sobre o polieletrólito e rlig a velocidade da formação de ligações entre elas e o polieletrólito.
As velocidades das reações podem ser calculadas como:
sendo kred a constante da eletrorredução, klig da formação do compósito. A aparição do número 3 na equação (57) é explicada pelo fato de se transferirem, aquando da reação elementar, os 3 elétrons:
γ é o coeficiente que descreve a interação entre o polieletrólito e as nanopartículas metálicas.
Analisar-se-á o sistema de equações (55, 56), tendo em conta as relações (57, 58) por meio da teoria de estabilidade linear e da análise de bifurcações. A matriz funcional de Jacobi, sendo os seus elementos calculados para o estado estacionário, ver-se-á como:
Sendo:
Depois da simplificação, feita por meio da introdução de novas variáveis, o determinante da matriz descrever-se-á como:
Como a condição de Tr J < 0 é satisfeita para a maioria dos sistemas (e, pelo que se vê, para este também), a principal será Det J >0, que se satisfará em condição de:
Esta condição confirma a ausência da possibilidade de quaisquer instabilidades no sistema, descrito em [73]. Manter-se-á, nele, o estado estacionário estável.
O K, parâmetro de difusão, é sempre positivo e os parâmetros Ξ e Φ, também. Assim, o único parâmetro, capaz de ser negativo, pode ser P, mas isso só acontecerá no caso da interação repelente, o que não existe no sistema em destaque. Assim, não serão satisfeitas, para ele, as condições de Det J = 0 (com a positividade do parâmetro P, o determinante será positivo) para a instabilidade monotônica, e de Tr J = 0 para a oscilatória (o traço da matriz terá valores negativos). Assim, matematicamente é confirmada a razoabilidade da escolha de modo da eletrodeposição catódica de nanopartículas.
Em geral, porém, ambas as instabilidades podem ocorrer, mas só no caso da ocorrência da interação repelente entre o revestimento e as nanopartículas.
Modo galvanostático
Como foi descrito na subseção 2.1, a razoabilidade da escolha do modo da eletrode-posição para o processo do artigo [73] (potenciostático), foi comprovada pela modelagem matemática. Para o sistema concreto não acontecem quaisquer instabilidades eletroquímicas, mantendo-se o estado estacionário estável.
Para os modos galvanostático e potenciodinâmico o comportamento é mais complicado, pois há mais fatores que o definem.
Assim, para analisá-lo matematicamente, introduzir-se-ão as três variáveis:
c - a concentração dos íons complexos AuCl4-;
T - o grau do revestimento da superfície do elétrodo pelas nanopartículas de ouro,
Q - a carga do elétrodo.
Como no caso da subseção anterior, os íons complexos difundem-se para a camada pré-superficial e, após isso, são eletrorreduzidos, formando o metal sob a forma de nanopartículas. Posto assim, a equação de balanço dos íons complexas de ouro (III) será descrita como:
sendo rred a velocidade de deposição catódica e c0 a concentração dos íons complexos no interior da solução.
As nanopartículas metálicas formam-se aquando da eletrorredução do ânion AuCl4- e, depois formam ligações covalentes com o polieletrólito depositado. Destarte, a equação de balanço será descrita como:
Sendo Tmax a concentração máxima de partículas metálicas por sobre o polieletrólito e rig a velocidade da formação de ligações entre elas e o polieletrólito.
A equação de balanço de carga do elétrodo, para o modo galvanostático será descrita como
E, como na subseção anterior,
sendo a aparição do número 3 explicada acima.
Analisamos o comportamento do sistema da eletrodeposição galvanostática de nano-partículas de ouro por meio do uso da metodologia anteriormente descrita com o sistema de equações diferenciais (67-69). Os elementos estacionários da matriz funcional de Jacobi podem calcular-se como:
Sendo:
Após a introdução de novos parâmetros, que descrevem vários impactos dos processos ao estado estacionário, o determinante do jacobiano ver-se-á como:
Haja vista a não negatividade do coeficiente na expressão (83), a condição de estabilidade do estado estacionário, aplicando-se o critério Routh-Hurwitz, obter-se-á sob a forma de:
que se reduz até
Como não há, no sistema em destaque, forças repelentes entre as nanopartículas e o polieletrólito, bem como a diminuição da capacidade de deposição de carga por parte do compósito formado, comparado com o polímero, não vai haver possibilidade de instabilidades oscilatória e monotônica e a condição (85) se satisfaz sempre. Assim sendo, no modo galvanostático a eletrodeposição de nanopartículas de ouro sobre o poliami-noácido também corre sem instabilidades.
Em geral, porém o comportamento oscilatório pode ser causado pela possível interação repelente entre as nanopartículas metálicas (negatividade de P) e diminuição da possibilidade de depositar carga elétrica pelo compósito (negatividade de U).
A instabilidade monotônica pode ser causada pela igualdade dos impactos da atração entre as nanopartículas metálicas e o polieletrólito aos da sua eletrodeposição (nulidade de P).
Seção III. desempenho do sensor
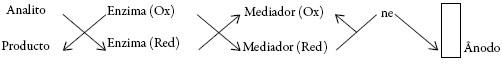
Os PC são amplamente usados em sensores e biossensores eletroquímicos [14-58]. Como é conhecido, o polímero condutor pode desempenhar, no sensor, o papel de
- substância ativa (sendo o exemplo, o polímero condutor, substituído pelo fragmento do fator ou cofator da enzima-esquema I);
- mediador (esquema-II)
- catalisador.
A descrição matemática do desempenho de compósitos, baseados em polímeros condutores, pode ser feita de dois jeitos:
- considerando o compósito como um todo (menosprezando a distribuição de papéis dos seus componentes no desempenho do sensor), ou seja, tendo em base o Esq. I;
- tendo em conta os papéis dos componentes do compósito (neste caso, as nanopar-tículas são substância ativa e o polímero condutor, o mediador), ou seja, tendo em base o Esq. II.
Ambas as possibilidades foram usadas nos nossos trabalhos anteriores.
Embora o primeiro jeito possa ser criticado pela possível inexatidão, tal "junção" pode simplificar a modelagem, especialmente quando é preciso descrever matematicamente a determinação de dois compostos diferentes ou de investigar, por meio da modelagem, a influência da presença doutras substâncias ao desempenho do sensor.
Na subseção 3.1. será modelado e investigado teoricamente o desempenho do sensor, descrito em [73]. Na subseção 3.2. será descrita a influência de outras substâncias ao seu desempenho.
A descrição matemática da detecção simultânea de dopamina e ácido úrico no pH neutro por meio do sensor, baseado no compósito nano-Au-poliaminoácido.
Para tal, serão introduzidas as três variáveis:
A1 - a concentração da dopamina na camada pré-superficial;
A2 - a concentração do ácido úrico na camada pré-superficial;
Θ - o grau do recobrimento do compósito reduzido, sendo que a forma oxidada, a princípio, cobre toda a superfície,
Supondo o exposto acima.
Os analitos entram na camada pré-superficial por meio da difusão e depois reagem com o compósito, sendo oxidadas. Além disso, eles voltam ao seu estado inicial mudando-se o potencial do elétrodo, pois o comportamento eletroquímico do compósito, segundo [73] foi reversível. Outrossim, eles reagem um com outro destarte, as equações de balanço da concentração dos analitos na camada pré-superficial serão descritas como:
Sendo D e Δ os seus coeficientes de difusão, A1B e A2B as concentrações dos analitos no interior da solução, r1, r2 e rd as velocidades das reações da oxidação química entre o compósito e os analitos e da possível reação da dopamina com o ácido úrico.
A forma reduzida do compósito é formada por meio da oxidação de analitos, sendo depois eletroquimicamente oxidada. Como a reação do compósito é reversível, a forma reduzida também é formada aquando do processo catódico de redução. Destarte, a equação de seu balanço será descrita como:
com as variáveis análogas às descritas nas seções anteriores.
As velocidades das respectivas reações podem ser calculadas como:
Investigando-se o comportamento do sensor, analisa-se o sistema (86-88) por meio da teoria de estabilidade linear. Os elementos estacionários da matriz funcional de Jacobi serão:
sendo:
Com a introdução de novas variáveis, o determinante simplificar-se-á até:
Aplicando-se o critério de estabilidade mencionado acima, obter-se-á a condição de estabilidade do estado estacionário sob a forma:
Mesmo que se mantenham na expressão (104) os parênteses, pode-se ver que as expressões que os coeficientes que acompanham os parâmetros da concentração dos analitos (X1 eX2) são negativos, assim o aumento da velocidade da difusão do analito (aumento de valores de parâmetros k), e das suas reações (aumento de valores dos parâmetros t/ e R) faz com que o estado estacionário seja mais estável, sendo confirmado o controle de reação pela difusão, relatado em [73]. A aceleração das reações eletroquímicas (aumento de W1 e de W2) sem influências à dupla camada (positividade de W2 e a negatividade de W-2). Posto assim, a região do melhor desempenho (trecho linear da curva parâmetro eletroquímico-concentração do analito) para o sensor deve ser vasta, o que está de acordo com os dados experimentais [73].
Aquando da igualdade dos efeitos da dupla camada às outras, ocorre a instabilidade monotônica, a condição de cuja aparição pode ser descrita como:
Como nos casos anteriores do comportamento dos sensores eletroquímicos, baseados em polímeros condutores [74-84], o comportamento oscilatório pode ser causado pela influência das etapas eletroquímicas à dupla camada (neste caso, a negatividade de W2 e a positividade de W-2). Porém agora, haja vista a reversibilidade da reação, o "ciclo de limite" vai ser acompanhado pelo arranjo-rearranjo desta camada e as mudanças cíclicas das suas capacitâncias. Esta causa do comportamento oscilatório é a única, pois as propriedades dos analitos não preveem autocatálise.
Destarte, o modelo confirma o observado experimentalmente em [73], sendo também possível a sua aplicação para casos análogos.
A influência dopHe dopOHao desempenho do sensor
Em [73] foi visto que o desempenho do sensor era melhor sob pH=7, o que se deve à melhor estabilidade do aminoácido no meio neutro.
As diferentes inclinações da curva de relações de correntes de picos com o pH aos lados básico e ácido (Fig. 4b de [73]) podem ser explicadas pelas diferentes capacidades dos analitos de reagirem com os ácidos e as bases fortes.
Para explicar a diferença do comportamento do sensor nos valores do pH, diferentes de 7, desenvolvemos um modelo matemático, capaz de descrever adequadamente os processos no sistema que ocorrem aquando da análise da concentração de cada analito dos mencionados.
O modelo, descrito em [84] pode aplicar-se ao sensor, descrito em [73], sendo as conclusões da modelagem aplicadas. Mas, como o modelo [84] é generalista, é preciso adaptá-lo, aliás, aplicá-lo para o caso particular destacado, o que se fará nesta subseção.
Para avaliar matematicamente a influência do pH ao desempenho do sensor, descrito em [73] para cada um dos analitos, podemos introduzir 3 variáveis:
- A - a concentração do analito na camada pré-superficial;
- Θ - o grau do recobrimento do compósito reduzido;
- H - a concentração dos prótons na camada pré-superficial. supondo o suposto acima.
Analogamente à subseção 3.1 e aos artigos [83, 84] e haja vista a reversibilidade do comportamento eletroquímico do compósito, descrevem-se as equações de balanço. Destarte, o sistema de equações se apresenta como:
sendo os parâmetros H as velocidades das reações dos prótons com o analito, o compósito reduzido e o oxidado correspondentemente. Se o ácido for selênico, haverá a destruição do compósito eliminando-se as nanopartículas de ouro.
As velocidades das respectivas reações podem ser calculadas como:
tomando em conta que o polímero reage diferentemente com o compósito reduzido e oxidado.
Para analisar o comportamento do sistema com o desempenho do sensor em meio fortemente ácido e compará-lo para o caso de meio neutro, investigamos o sistema de equações diferenciais (106-108) por meio da teoria de estabilidade lineal e da análise de bifurcações. Os elementos estacionários da sua matriz funcional de Jacobi veem-se como:
sendo:
Já pela análise dos elementos da diagonal principal da matriz funcional de Jacobi se vê que o comportamento pode ser causado pelos fatores mencionados da dupla camada, mas a amplitude de oscilações vai crescer haja vista a aparição dos elementos que descrevem o comportamento de prótons, introduzimos as novas variáveis para simplificar o Jacobiano e descrevê-lo de forma:
Que, após a junção dos parâmetros que descrevem o comportamento superficial num só, vai simplificar-se até:
A condição da estabilidade de estado estacionário, obtida do requisito de (126) <0, pode ser expressa como:
Comparada à inequação (104), a (127) tem mais elementos positivos à esquerda. Assim sendo, pode ser confirmado que a diminuição do pH torna menor a faixa de exatidão do sensor e a intensidade do pico, haja vista o comportamento dos prótons.
Como pode ser observado, a estabilidade de estado estacionário mantém-se pelo aumento da velocidade da difusão do analito (aumento de k1) e das velocidades da saída de prótons. O aumento da concentração de prótons, bem como a rapidez da sua difusão não favorece a exatidão do sensor de aminas.
A instabilidade monotônica para este sistema é realizada no caso de:
A mesma analise pode ser feita também para o meio fortemente básico. Tal meio também não favorece a exatidão do sensor (não favorecendo a estabilidade de estado estacionário), mas, como as bases participam de menos reações, pois se comportam de maneira diversa, a zona de estabilidade é mais vasta que no caso do meio ácido (há menos elementos positivos à esquerda da inequação, análoga à inequação 127). Daí a assimetria do gráfico da dependência: "corrente de pico vs. pH".
Conclusões
1. A modelagem matemática dos processos da eletropolimerização do aminoácido, da eletrodeposição de nanopartículas de ouro e do desempenho do sensor, baseado no compósito obtido, indo ao encontro dos dados experimentais, confirmou a presença de estruturas dissipativas temporais no primeiro e no terceiro processo (para o caso do sensor experimentalmente descrito). No caso destacado, no segundo processo não há estruturas dissipativas temporais, mas no caso geral da eletrodeposição de nanopartículas metálicas sobre o eletrólito elas podem ocorrer.
2. Aquando da eletropolimerização, a estabilidade de estado estacionário é favorecida pelos fatores abaixo.
no caso do elétrodo de carbono vítreo:
- alta velocidade da difusão (altos valores de k);
- alta velocidade-padrão da adsorção (altos valores deg1);
- alto valor da velocidade-padrão da eletropolimerização (altos valores da Vp), acompanhado pelas influências na dupla camada dos processos superficiais (valores negativos de ΔΨ) ou da eletropolimerização (negatividade do parâmetro Vp);
no caso de elétrodo de metal ativo:
- reação cineticamente controlada.
3. A eletrodeposição potenciostática de nanopartículas de ouro sobre o polieletró-lito ocorre no estado estacionário estável. Obter-se-á o mesmo resultado ao se usar o modo galvanostático.
4. A comparação de modelos de desempenho do sensor nos modos neutro, fortemente ácido e básico confirma o fato de o sensor ser mais exato e ter resposta mais clara em meio neutro.
5. Os modelos podem ser usados não só para o sensor descrito, mas também para todos os sensores semelhantes.
Conflito de interesses
Os autores declaram que não há conflito de interesses.
Referências
1. J.M. Fellous, R.E. Suri, The Roles of Dopamine, URL: http://128.196.98.170/pubs/Dopamine-HBTNN2e-preprint.pdf, acesso aos 26 de outubro de 2016.
2. L.C. Triarhou, Dopamine and Parkinson Disease, http://www.ncbi.nlm.nihgov/books/NBK6271/, acesso aos 28 de setembro de 2016.
3. P. Seeman, Glutamate and dopamine components in schizophrenia, J. Psych. Neurosci., 34, 143 (2009).
4. M.A. Singer, Do mammals, birds, reptiles and fish have similar nitrogen conserving systems?, Comp. Biochem. Physiol., 134, 543 (2003).
5. K. Ichida, M. Hosoyamada, N. Kamatani et al., Age and origin of the G774A mutation in SLC22A12 causing renal hypouricemia in Japanese, Clin. Genet., 74, 243 (2008).
6. E.A. Roberts, M.L. Schilsky, Diagnosis and treatment of Wilson disease: An update, Hepatology, 47, 2089 (2008).
7. Y. Moriwaki, T. Yamamoto, S. Takahashi et al., Uric acid transport in Fanconi syndrome with marked renal hypouricemia, Nephron, 74, 452 (1996).
8. R.J. Torres, J. Puig, Hypoxanthine-guanine phosophoribosyltransferase (HPRT) deficiency: Lesch-Nyhan syndrome, Orph. J. Rare Dis., 2, 48 (2007).
9. R. Bravo, C.C. Hsueh, A. Jaramillo, A. Brajter-Toth, Possibilities and limitations in miniaturized sensor design for uric acid., Analyst, 123, 1625 (1998).
10. G. Rivas, M.D. Rubianes, M.C. Rodrigues et al., Carbon nanotubes for electrochemical biosensing, Talanta, 74, 291 (2007).
11. J.C. Giddings, F.J. Yang, M.N. Myers, Theoretical and experimental characterization of flow field-flow fractionation, Anal. Chem., 48, 1126 (1976).
12. J.A. Stamford, J.B. Justice, Probing brain chemistry, Anal. Chem., 68, 359A (1996).
13. F. Gonon, M. Buda, R. Cespuglio et al., In vivo electrochemical detection of catechols in the neostriatum of anaesthetized rats: dopamine or DOPAC?, Nature, 286, 902 (1980).
14. G.Y. Jin, Y. Zhang, W.X. Cheng, Poly(p-aminobenzene sulfonic acid)-modified glassy carbon electrode for simultaneous detection of dopamine and ascorbic acid, Sens. Act. B, 107, 528 (2005).
15. H. Zhao, Y. Zhang, Z. Yan, Study on the electrochemical behavior of dopamine with poly(sulfosalicylic acid) modified glassy carbon electrode, Anal. Chim. Acta, 441, 117 (2001).
16. M. Li, H. Li, W. Zhong et al., Stretchable conductive polypyrrole/polyurethane (PPy/PU) strain sensor with netlike microcracks for human breath detection, Appl. Mat. Int., 6, 1313 (2014).
17. J.D. Nickels, C.E. Schmidt, Surface modification of the conducting polymer, polypyrrole, via affinity peptide, J. Biomed. Mater. Res. A, 101, 1464 (2013).
18. J.W. Park, S.J. Park, O.S. Kwon, Polypyrrole nanotube embedded reduced graphene oxide transducer for field-effect transistor-type H2O2 biosensor, Anal. Chem., 86, 1822 (2014).
19. Z. Yao, X. Hu, B. Huang et al., Halochromism of a polythiophene derivative induced by conformational changes and its sensing application of carbon dioxide, Appl. Mat., 5, 5783 (2013).
20. G. Fukuhara, Y. Inoue, Chirality-sensing binaphthocrown ether-polythiophene conjugate, Chem. Eur. J., 18, 11459 (2012).
21. V. Raj, S. Kunnetheeri, Nonconjugated polyelectrolyte as efficient fluorescence quencher and their applications as biosensors: Polymer-polymer interaction, ISRN Anal. Chem., ID: 841857 (2014).
22. N. Atta, M.F. El-Kady, A. Galal, Palladium nanoclusters-coated polyfuran as a novel sensor for catecholamine neurotransmitters and paracetamol, Sens. Act. B, 141, 566 (2009).
23. L. Lu, X. Lin, Selective determination of uric acid with DNA doped polymers modified carbon fiber microelectrode, Electrochem. Comm., 10, 704 (2008).
24. S.B. Khoo, F. Chen, Studies of sol-gel ceramic film incorporating methylene blue on glassy carbon: An electrocatalytic system for the simultaneous determination of ascorbic and uric acids, Anal. Chem., 74, 5734 (2002).
25. P. Shakkthivel, S.M. Chen, Simultaneous determination of ascorbic acid and dopamine in the presence of uric acid on ruthenium oxide modified electrode, Biosens. Bioelectr, 22, 1680 (2007).
26. A.A. Ensafi, H. Bahrami, H. Karimi-Maleh, Sh. Mallakpour, Carbon paste electrode prepared from chemically modified multiwall carbon nanotubes for the voltammetric determination of isoprenaline in pharmaceutical and urine samples, Chin. J. Cat, 33, 1919 (2012).
27. A. Liu, I. Honma, H. Zhou, Simultaneous voltammetric detection of dopamine and uric acid at their physiological level in the presence of ascorbic acid using poly(acrylic acid)-multiwalled carbon-nanotube composite-covered glassy-carbon electrode, Biosens. Bioelectr., 23, 74 (2007).
28. A.N. Shipway, M. Lahav, I. Willner, Nanostructured gold colloid electrodes, Adv. Mater., 12, 993 (2000).
29. A.A. Al-Suhybani, A.M. Al-Mayouf, M.S.A. Saleh, A. Aouissi, Development of a 12-tungstosilicate-modified carbon electrode for cyclohexane electrocatalytic oxidation, Int. J. Electrochem. Sci., 9, 1547 (2014).
30. R. Ojani, M.A. Rahmaninafar, P. Naderi, Electrocatalytic reduction of nitrite by phosphotungstic heteropolyanion. Application for its simple and selective determination, Electroanal., 20, 1092 (2008).
31. A. Sivanesan, P. Kannan, J.S. Abraham, Electrocatalytic oxidation of ascorbic acid using a single layer of gold nanoparticles immobilized on 1,6-hexanedithiol modified gold electrode, Electrochim. Acta, 52, 8118 (2007).
32. C.R. Raj, T. Okajima, T. Ohsaka, Gold nanoparticle arrays for the voltammetric sensing of dopamine, J. Electroanal. Chem., 543, 127 (2003).
33. L.Y. Zhang, R. Yuan, Y.Q. Chai, Z.L. Li, Investigation of the electrochemical and electrocatalytic behavior of positively charged gold nanoparticle and L-cysteine film on an Au electrode, Anal. Chim. Acta, 596, 99 (2007).
34. M.A. Sheikh-Mohseni, S. Pirsa, Simultaneous determination of dopamine and acetaminophen by a carbon paste electrode doubly modified with poly (pyrrole) and CuO nanoparticles, Anal. Bioanal. Electrochem., 8, 777 (2017).
35. J. Hernández-Ferrer, "Caracterización electroquímica de nanopartículas de oro. Relacion de la forma tridimensional de la partícula y su orientación superficial", Tesis Doctoral. Universidad de Alicante, 2008.
36. E. Arkan, M. Shamsipur, R. Saber et al., A novel electrochemical sensor based on a silver nanoparticle modified carbon ionic liquid electrode for selective and sensitive determination of levetiracetam in pharmaceutical tablets and blood plasma samples, Anal. Meth., 6, 2197 (2014).
37. S. Gupta, M. Tiwari, R. Prakash, Gold nanoparticles incorporated 3-(trimethoxysilyl)propyl methacrylate modified electrode for non-enzymatic electro-sensing of urea, J. Nanosci. Nanotechnol, 14, 2786 (2014).
38. G. Yin, L. Xing, X.J. Ma, J. Wan, Non-enzymatic hydrogen peroxide sensor based on a nanoporous gold electrode modified with platinum nanoparticles, Chem. Papers, 68, 435 (2014).
39. D.N. Muraviev, Inter-matrix synthesis of polymer stabilised metal nanoparticles for sensor applications, Contributions to Science, 3, 19 (2005).
40. Y. Ohnuki, T. Ohsaka, H. Matsuda, N. Oyama, Permselectivity of films prepared by electrochemical oxidation of phenol and amino-aromatic compounds, J. Electroanal. Chem. Interf. Electrochem., 158, 55 (1983).
41. J. Roncali, Conjugated poly(thiophenes). Synthesis, functionalization, and applications, Chem. Rev., 92, 711 (1992).
42. R. Ojani, J.B. Raoof, A. Ahmady, S.R. Hosseini, High improved methanol oxidation onto carbon paste electrode modified by nickel particles dispersed into poly(2,5-dimethylamine) film, Casp. J. Chem., 2, 45 (2013).
43. R. Ojani, J.B. Raoof, S.R. Hosseini, Electrocatalytic oxidation of methanol on carbon paste electrode modified by nickel ions dispersed into poly (1,5-diami-nonaphthalene) film, Electrochim. Acta, 53, 2402 (2008).
44. R. Ojani, J.B. Raoof, P.S. Afagh, Electrocatalytic oxidation of some carbohydrates by poly(1-naphthylamine)/nickel modified carbon paste electrode, J. Electroanal. Chem, 571, 1 (2004).
45. Z. Bagheryan, J.B. Raoof, R. Ojani, E. Hamidi-Asl, Introduction of ketamine as a G-quadruplex-binding ligand using platinum nanoparticle modified carbon paste electrode, Electroanal., 25, 2659 (2013).
46. J. Raoof, R. Ojani, S.R. Hosseini, An electrochemical investigation of methanol oxidation on nickel hydroxide nanoparticles, South Afr. J. Chem., 66, 47 (2013).
47. R. Ojani, J.B. Raoof, R. Valliolahi, Pt nanoparticles/graphene paste electrode for sodium borohydride electrooxidation, J. Solid. State. Electrochem., 17, 217 (2013).
48. S.J. Tian, J.Y. Liu, T. Zhu, W. Knoll, Polyaniline/gold nanoparticle multilayer films: Assembly, properties, and biological applications, Chem. Mater., 16, 4103 (2004).
49. L.J. Zhang, M.X. Wan, Polyaniline/TiO2 composite nanotubes, J. Phys. Chem. B, 107, 6748 (2003).
50. R. Davies, G.A. Schurr, P. Meenan et al. Engineered particle surfaces, Adv. Mat., 10, 1264 (1998).
51. C.S.C. Bose, K. Rajeshwar, Efficient electrocatalyst assemblies for proton and oxygen reduction: the electrosynthesis and characterization of polypyrrole films containing nanodispersed platinum particles, J. Electroanal. Chem., 333, 235 (1992).
52. J. Li, X.Q. Lin, Electrocatalytic reduction of nitrite at polypyrrole nanowire-platinum nanocluster modified glassy carbon electrode, Microchem. J., 87, 41 (2007).
53. Y.C. Liu, H.T. Lee, S.J. Yang, Strategy for the syntheses of isolated fine silver nanoparticles and polypyrrole/silver nanocomposites on gold substrates, Electrochim. Acta, 51, 3441 (2006).
54. S. Roux, G.J. Soler-Illia, A.A. Demoustier-Champagne et al., Titania/polypyrrole hybrid nanocomposites built from in-situ generated organically functionalized nanoanatase building blocks, Adv. Mat., 15, 217 (2003).
55. W. Chen, C.M. Li, P. Chen, C.Q. Sun, Electrosynthesis and characterization of polypyrrole/Au nanocomposite, Electrochim. Acta, 52, 2845 (2007).
56. B. Krische, M. Zagorska, The polythiophene paradox, Synth. Met., 28, 263 (1989).
57. K. Aoki, I. Mukoyama, J. Chen, Polymerization and dissolution of polythiophene films, Russ. J. Electrochem., 40, 319 (2004).
58. J. Mostany, B.R Scharifker, Impedance spectroscopy of undoped, doped and overoxidized polypyrrole films, Synth. Met., 87, 179 (1997).
59. I. Das, N.R. Agrawal, S.A. Ansari, S.K. Gupta, Pattern formation and oscillatory electropolymerization of thiophene, Indian J. Chem. A, 47A, 1798 (2008).
60. I. Das, N. Goel, S.K. Gupta, N.R. Agrawal, Electropolymerization of pyrrole: Dendrimers, nano-sized patterns and oscillations in potential in presence of aromatic and aliphatic surfactants, J. Electroanal. Chem., 670, 1 (2012).
61. I. Das, N. Goel, N.R. Agrawal, S. K. Gupta, Growth patterns of dendrimers and electric potential oscillations during electropolymerization of pyrrole using mono and mixed surfactants, J. Phys. Chem., 114, 12888 (2010).
62. M.S. Ba-Shammakh, "Electropolymerization of pyrrole on mild steel for corrosion protection", Ph. D. Thesis, King Fahd University of Petroleum and Minerals, Dharan, Saudi Arabia, 2002.
63. D. Sazou, The dynamical behavior of the electrochemical polymerization of indole on Fe in acetonitrile-water mixtures, Synth. Met., 130, 45 (2002).
64. A.S. Liu, M.A.S. Oliveira, Electrodeposition of polypyrrole films on aluminum from tartrate aqueous solution, J. Braz. Chem. Soc., 18, 143 (2007).
65. K.R. Lemos-Castagno, "Eletropolimerização de pirrol sobre liga de alumínio 1100", Tese D. Sc., Universidade Federal do Rio Grande do Sul, Porto Alegre, 2007.
66. M. Bazzaoui, E.A. Bazzaoui, L. Martins, J.I. Martins, Electropolymerization of pyrrole on zinc-lead-silve ralloys' electrodes in neutral and acid organic media, Synth. Met, 130, 73 (2002).
67. V.M. de Andrade, "Confecção de biossensores através da imobilização de bio-componentes por eletropolimerização de pirrol", Tese M. Eng., Universidade Federal do Rio Grande do Sul, Porto Alegre, 2006.
68. J.P. Tosar-Rovira, "Estudio de la inmovilización de oligonucleótidos a elétrodos modificados de oro: polipirrol, y detección electroquímica de secuencias complementarias", Tesis Lic. Bioquím., Universidad de la República, Montevideo, 2008.
69. D. Zane, G. B. Appetecchi, C. Bianchini et al., An impedimetric glucose biosensor based on overoxidized polypyrrole thin film, Electroanal., 23, 1134 (2011).
70. Y. Oztekin, A. Ramanaviciene, Z. Yazicigil et al., Direct electron transfer from glucose oxidase immobilized on polyphenanthroline-modified glassy carbon electrode, Biosens. Bioelectr., 26, 2541 (2011).
71. R. Singh, Prospects of organic conducting polymer modified electrodes: Enzymosensors, Int. J. Electrochem., N 502707(2012).
72. O. Korostynska, K. Arshak, E. Gill, A. Arshak, Review on state-of-the-art in polymer based pH sensors, Sensors, 7, 3027 (2008).
73. R. Ojani, J.-B. Raoof, A.A. Maleki, S. Safshekan, Simultaneous voltammetric determination of ascorbic acid and dopamine at the surface of electrodes modified with selfassembled gold nanoparticle films, Chin. J. Cat., 35, 423 (2014).
74. V. Tkach, V. Nechyporuk, P. Yagodynets, Eletropolimerização dos compostos heterocíclicos. Modelos matemáticos, Ciênc. Tecn. Mat., 24, 54 (2012).
75. V. Tkach, V. Nechyporuk, P. Yagodynets', As eletropolimerizações galvanostática e potenciodinâmica (em voltagem constante), dos compostos heterocíclicos e a sua descrição matemática, Tecn. Met. Mat. Min., 10, 249 (2013).
76. V.V. Tkach, V.V. Nechyporuk, P.I. Yagodynets', The mathematical description of "the polythiophene paradox" for soluble polymers, Geor. Chem. J., 11, 387 (2011).
77. V. Tkach, V. Nechyporuk, P. Yagodynets, Estudio matemático de las condiciones de estabilidad en la polimerización electroquímica de compuestos heterocíclicos en presencia de oxidantes, Afinidad, 70, 73 (2013).
78. V. Tkach, V. Nechyporuk, P. Yagodynets, Descripción matemática de la síntesis electroquímica de polímeros conductores en la presencia de surfactantes, Avances en Química, 8, 9 (2013).
79. V.V. Tkach, V.V. Nechyporuk, P.I. Yagodynets', V. Meslyuk-Yu, Mathematical model of the work of the mediated conducting polymer sensors with the use of the polymers of heterocyclic compounds, Revista de la Sociedad Química del Perú, 77, 259 (2011).
80. V. Tkach, V. Nechyporuk, P. Yagodynets', I. Rusnak, Los biosensores basados en los polímeros conductores, y la descripción matemática de su desempeño, Orbital Electr. J. Chem, 4, 136 (2012).
81. V. Tkach, B. Kumara Swamy, R. Ojani et al., Mecanismo de la oxidación de omeprazol sobre el electrodo de carbono vitroso, modificado por polializarina, y su descripción matemática, Orbital Electr. J. Chem., 7, 1 (2015).
82. V. Tkach, B. Kumara Swamy, R. Ojani et al., Comportamento de paracetamol durante a sua oxidação eletrocatalítica sobre poli(azul da anilina) e a sua descrição matemática, Rev. Colomb. Cienc. Quím. Farm., 44, 148 (2015).
83. V. Tkach, V. Nechyporuk, P. Yagodynets, El estudio de estabilidad en el proceso del desempeño de los sensores y biosensores, basados en los polímeros conductores en medio fuertemente ácido, Rev. Colomb. Cienc. Quím. Farm., 42, 30 (2013).
84. V. Tkach, V. Nechyporuk, P. Yagodynets', A descrição matemática do processo da detecção de duas substâncias com um grupo funcional comum pelos sensores e biossensores eletroquímicos, baseados em polímeros condutores, Rev. Colomb. Cienc. Quím. Farm., 42, 215 (2013).
85. X. Bao, Metallopolymer electrochromic film prepared by oxidative electropolymerization of a Fe(II) complex with arylamine functionalized terpyridine ligand, Inorg. Chem. Commun., 38, 88 (2013).
86. M. Sousa, É. J. Dallan, R. Bertazolli, Obtenção de um material adsorvente para íons de metais pesados, através da eletropolimerização de 2-mercapto- benzimidazol à superfície de carbono vítreo reticulado, Quím. Nova, 23, 326 (2000).
87. D.N. Gritsan, "As investigações do processos eletródicos e da eletrodeposição dos metais. Cádmio" (publicado em russo), Kharkov, Vyshcha shkola, 1974, 144 páginas.
Como citar este artigo
Volodymyr V. Tkach, Sílvio C. de Oliveira, Yana G. Ivanushko, Svitlana M. Lukaniova, Reza Ojani, Petró I. Yagodynets', Aline M. da Rocha, Estudo de estabilidade da ele-trossíntese e do desempenho eletroanalítico do compósito poli(l-metionina)-nano-Au na presença de dopamina e de ácido úrico, Rev. Colomb. Cienc. Quím. Farm., 45(3), 385-421 (2016).
Referencias
(1) J.M. Fellous, R.E. Suri, The Roles of Dopamine, URL: http://128.196.98.170/pubs/Dopamine-HBTNN2e-preprint.pdf , acesso aos 26 de outubro de 2016.
(2) L.C. Triarhou, Dopamine and Parkinson Disease, http://www.ncbi.nlm.nih.gov/books/NBK6271/ , acesso aos 28 de setembro de 2016.
(3) P. Seeman, Glutamate and dopamine components in schizophrenia, J. Psych. Neurosci., 34, 143 (2009).
(4) M.A. Singer, Do mammals, birds, reptiles and fish have similar nitrogen conserving systems?, Comp. Biochem. Physiol., 134, 543 (2003).
(5) K. Ichida, M. Hosoyamada, N. Kamatani et al., Age and origin of the G774A mutation in SLC22A12 causing renal hypouricemia in Japanese, Clin. Genet., 74, 243 (2008).
(6) E.A. Roberts, M.L. Schilsky, Diagnosis and treatment of Wilson disease: An update, Hepatology, 47, 2089 (2008).
(7) Y. Moriwaki, T. Yamamoto, S. Takahashi et al., Uric acid transport in Fanconi syndrome with marked renal hypouricemia, Nephron, 74, 452 (1996).
(8) R.J. Torres, J. Puig, Hypoxanthine-guanine phosophoribosyltransferase (HPRT) deficiency: Lesch-Nyhan syndrome, Orph. J. Rare Dis., 2, 48 (2007).
(9) R. Bravo, C.C. Hsueh, A. Jaramillo, A. Brajter-Toth, Possibilities and limitations in miniaturized sensor design for uric acid., Analyst, 123, 1625 (1998).
(10) G. Rivas, M.D. Rubianes, M.C. Rodrigues et al., Carbon nanotubes for electrochemicalbiosensing, Talanta, 74, 291 (2007).
(11) J.C. Giddings, F.J. Yang, M.N. Myers, Theoretical and experimental characterization of flow field-flow fractionation, Anal. Chem., 48, 1126 (1976).
(12) J.A. Stamford, J.B. Justice, Probing brain chemistry, Anal. Chem., 68, 359A (1996).
(13) F. Gonon, M. Buda, R. Cespuglio et al., In vivo electrochemical detection of catechols in the neostriatum of anaesthetized rats: dopamine or DOPAC?, Nature, 286, 902 (1980).
(14) G.Y. Jin, Y. Zhang, W.X. Cheng, Poly(p-aminobenzene sulfonic acid)-modified glassy carbon electrode for simultaneous detection of dopamine and ascorbic acid, Sens. Act. B, 107, 528 (2005).
(15) H. Zhao, Y. Zhang, Z. Yan, Study on the electrochemical behavior of dopamine with poly(sulfosalicylic acid) modified glassy carbon electrode, Anal. Chim. Acta, 441, 117 (2001).
(16) M. Li, H. Li, W. Zhong et al., Stretchable conductive polypyrrole/polyurethane (PPy/PU) strain sensor with netlike microcracks for human breath detection, Appl. Mat. Int., 6, 1313 (2014).
(17) J.D. Nickels, C.E. Schmidt, Surface modification of the conducting polymer, polypyrrole, via affinity peptide, J. Biomed. Mater. Res. A, 101, 1464 (2013).
(18) J.W. Park, S.J. Park, O.S. Kwon, Polypyrrole nanotube embedded reduced graphene oxide transducer for field-effect transistor-type H2O2 biosensor, Anal. Chem., 86, 1822 (2014).
(19) Z. Yao, X. Hu, B. Huang et al., Halochromism of a polythiophene derivative induced by conformational changes and its sensing application of carbon dioxide, Appl. Mat., 5, 5783 (2013).
(20) G. Fukuhara, Y. Inoue, Chirality-sensing binaphthocrown ether-polythiophene conjugate, Chem. Eur. J., 18, 11459 (2012).
(21) V. Raj, S. Kunnetheeri, Nonconjugated polyelectrolyte as efficient fluorescence quencher and their applications as biosensors: Polymer-polymer interaction, ISRN Anal. Chem., ID: 841857 (2014).
(22) N. Atta, M.F. El-Kady, A. Galal, Palladium nanoclusters-coated polyfuran as a novel sensor for catecholamine neurotransmitters and paracetamol, Sens. Act. B, 141, 566 (2009).
(23) L. Lu, X. Lin, Selective determination of uric acid with DNA doped polymers modified carbon fiber microelectrode, Electrochem. Comm., 10, 704 (2008).
(24) S.B. Khoo, F. Chen, Studies of sol-gel ceramic film incorporating methylene blue on glassy carbon: An electrocatalytic system for the simultaneous determination of ascorbic and uric acids, Anal. Chem., 74, 5734 (2002).
(25) P. Shakkthivel, S.M. Chen, Simultaneous determination of ascorbic acid and dopamine in the presence of uric acid on ruthenium oxide modified electrode, Biosens. Bioelectr., 22, 1680 (2007).
(26) A.A. Ensafi, H. Bahrami, H. Karimi-Maleh, Sh. Mallakpour, Carbon paste electrode prepared from chemically modified multiwall carbon nanotubes for the voltammetric determination of isoprenaline in pharmaceutical and urine samples, Chin. J. Cat., 33, 1919 (2012).
(27) A. Liu, I. Honma, H. Zhou, Simultaneous voltammetric detection of dopamine and uric acid at their physiological level in the presence of ascorbic acid using poly(acrylic acid)-multiwalled carbon-nanotube composite-covered glassy-carbon electrode, Biosens. Bioelectr., 23, 74 (2007).
(28) A.N. Shipway, M. Lahav, I. Willner, Nanostructured gold colloid electrodes, Adv. Mater., 12, 993 (2000).
(29) A.A. Al-Suhybani, A.M. Al-Mayouf, M.S.A. Saleh, A. Aouissi, Development of a 12-tungstosilicate-modified carbon electrode for cyclohexane electrocatalytic oxidation, Int. J. Electrochem. Sci., 9, 1547 (2014).
(30) R. Ojani, M.A. Rahmaninafar, P. Naderi, Electrocatalytic reduction of nitrite by phosphotungstic heteropolyanion. Application for its simple and selective determination, Electroanal., 20, 1092 (2008).
(31) A. Sivanesan, P. Kannan, J.S. Abraham, Electrocatalytic oxidation of ascorbic acid using a single layer of gold nanoparticles immobilized on 1,6-hexanedithiol modified gold electrode, Electrochim. Acta, 52, 8118 (2007).
(32) C.R. Raj, T. Okajima, T. Ohsaka, Gold nanoparticle arrays for the voltammetric sensing of dopamine, J. Electroanal. Chem., 543, 127 (2003).
(33) L.Y. Zhang, R. Yuan, Y.Q. Chai, Z.L. Li, Investigation of the electrochemical and electrocatalytic behavior of positively charged gold nanoparticle and L-cysteine film on an Au electrode, Anal. Chim. Acta, 596, 99 (2007).
(34) M.A. Sheikh-Mohseni, S. Pirsa, Simultaneous determination of dopamine and acetaminophen by a carbon paste electrode doubly modified with poly (pyrrole) and CuO nanoparticles, Anal. Bioanal. Electrochem., 8, 777 (2017).
(35) J. Hernandez-Ferrer, “Caracterizacion electroquimica de nanoparticulas de oro. Relacion de la forma tridimensional de la particula y su orientacion superficial”, Tesis Doctoral. Universidad de Alicante, 2008.
(36) E. Arkan, M. Shamsipur, R. Saber et al., A novel electrochemical sensor based on a silver nanoparticle modified carbon ionic liquid electrode for selective and sensitive determination of levetiracetam in pharmaceutical tablets and blood plasma samples, Anal. Meth., 6, 2197 (2014).
(37) S. Gupta, M. Tiwari, R. Prakash, Gold nanoparticles incorporated 3-(trimethoxysilyl) propyl methacrylate modified electrode for non-enzymatic electro-sensing of urea, J. Nanosci. Nanotechnol., 14, 2786 (2014).
(38) G. Yin, L. Xing, X.J. Ma, J. Wan, Non-enzymatic hydrogen peroxide sensor based on a nanoporous gold electrode modified with platinum nanoparticles, Chem. Papers, 68, 435 (2014).
(39) D.N. Muraviev, Inter-matrix synthesis of polymer stabilised metal nanoparticles for sensor applications, Contributions to Science, 3, 19 (2005).
(40) Y. Ohnuki, T. Ohsaka, H. Matsuda, N. Oyama, Permselectivity of films prepared by electrochemical oxidation of phenol and amino-aromatic compounds, J. Electroanal. Chem. Interf. Electrochem., 158, 55 (1983).
(41) J. Roncali, Conjugated poly(thiophenes). Synthesis, functionalization, and applications, Chem. Rev., 92, 711 (1992).
(42) R. Ojani, J.B. Raoof, A. Ahmady, S.R. Hosseini, High improved methanol oxidation onto carbon paste electrode modified by nickel particles dispersed into poly(2,5-dimethylamine) film, Casp. J. Chem., 2, 45 (2013).
(43) R. Ojani, J.B. Raoof, S.R. Hosseini, Electrocatalytic oxidation of methanol on carbon paste electrode modified by nickel ions dispersed into poly (1,5-diaminonaphthalene) film, Electrochim. Acta, 53, 2402 (2008).
(44) R. Ojani, J.B. Raoof, P.S. Afagh, Electrocatalytic oxidation of some carbohydrates by poly(1-naphthylamine)/nickel modified carbon paste electrode, J. Electroanal. Chem., 571, 1 (2004).
(45) Z. Bagheryan, J.B. Raoof, R. Ojani, E. Hamidi-Asl, Introduction of ketamine as a G-quadruplex-binding ligand using platinum nanoparticle modified carbon paste electrode, Electroanal., 25, 2659 (2013).
(46) J. Raoof, R. Ojani, S.R. Hosseini, An electrochemical investigation of methanol oxidation on nickel hydroxide nanoparticles, South Afr. J. Chem., 66, 47 (2013).
(47) R. Ojani, J.B. Raoof, R. Valliolahi, Pt nanoparticles/graphene paste electrode for sodium borohydride electrooxidation, J. Solid. State. Electrochem., 17, 217 (2013).
(48) S.J. Tian, J.Y. Liu, T. Zhu, W. Knoll, Polyaniline/gold nanoparticle multilayer films: Assembly, properties, and biological applications, Chem. Mater., 16, 4103 (2004).
(49) L.J. Zhang, M.X. Wan, Polyaniline/TiO2 composite nanotubes, J. Phys. Chem. B, 107, 6748 (2003).
(50) R. Davies, G.A. Schurr, P. Meenan et al., Engineered particle surfaces, Adv. Mat., 10, 1264 (1998).
(51) C.S.C. Bose, K. Rajeshwar, Efficient electrocatalyst assemblies for proton and oxygen reduction: the electrosynthesis and characterization of polypyrrole films containing nanodispersed platinum particles, J. Electroanal. Chem., 333, 235 (1992).
(52) J. Li, X.Q. Lin, Electrocatalytic reduction of nitrite at polypyrrole nanowire– platinum nanocluster modified glassy carbon electrode, Microchem. J., 87, 41 (2007).
(53) Y.C. Liu, H.T. Lee, S.J. Yang, Strategy for the syntheses of isolated fine silver nanoparticles and polypyrrole/silver nanocomposites on gold substrates, Electrochim. Acta, 51, 3441 (2006).
(54). S. Roux, G.J. Soler-Illia, A.A. Demoustier-Champagne et al., Titania/polypyrrole hybrid nanocomposites built from in-situ generated organically functionalized nanoanatase building blocks, Adv. Mat., 15, 217 (2003).
(55) W. Chen, C.M. Li, P. Chen, C.Q. Sun, Electrosynthesis and characterization of polypyrrole/Au nanocomposite, Electrochim. Acta, 52, 2845 (2007).
(56) B. Krische, M. Zagorska, The polythiophene paradox, Synth. Met., 28, 263 (1989).
(57) K. Aoki, I. Mukoyama, J. Chen, Polymerization and dissolution of polythiophene films, Russ. J. Electrochem., 40, 319 (2004).
(58) J. Mostany, B.R Scharifker, Impedance spectroscopy of undoped, doped and overoxidized polypyrrole films, Synth. Met., 87, 179 (1997).
(59) I. Das, N.R. Agrawal, S.A. Ansari, S.K. Gupta, Pattern formation and oscillatory electropolymerization of thiophene, Indian J. Chem. A, 47A, 1798 (2008).
(60) I. Das, N. Goel, S.K. Gupta, N.R. Agrawal, Electropolymerization of pyrrole: Dendrimers, nano-sized patterns and oscillations in potential in presence of aromatic and aliphatic surfactants, J. Electroanal. Chem., 670, 1 (2012).
(61) I. Das, N. Goel, N.R. Agrawal, S. K. Gupta, Growth patterns of dendrimers and electric potential oscillations during electropolymerization of pyrrole using mono and mixed surfactants, J. Phys. Chem., 114, 12888 (2010).
(62) M.S. Ba-Shammakh, “Electropolymerization of pyrrole on mild steel for corrosion protection”, Ph. D. Thesis, King Fahd University of Petroleum and Minerals, Dharan, Saudi Arabia, 2002.
(63) D. Sazou, The dynamical behavior of the electrochemical polymerization of indole on Fe in acetonitrile-water mixtures, Synth. Met., 130, 45 (2002).
(64) A.S. Liu, M.A.S. Oliveira, Electrodeposition of polypyrrole films on aluminum from tartrate aqueous solution, J. Braz. Chem. Soc., 18, 143 (2007).
(65) K.R. Lemos-Castagno, “Eletropolimerizacao de pirrol sobre liga de aluminio 1100”, Tese D. Sc., Universidade Federal do Rio Grande do Sul, Porto Alegre, 2007.
(66) M. Bazzaoui, E.A. Bazzaoui, L. Martins, J.I. Martins, Electropolymerization of pyrrole on zinc-lead-silve ralloys’ electrodes in neutral and acid organic media, Synth. Met., 130, 73 (2002).
(67) V.M. de Andrade, “Confeccao de biossensores atraves da imobilizacao de biocomponentes por eletropolimerizacao de pirrol”, Tese M. Eng., Universidade Federal do Rio Grande do Sul, Porto Alegre, 2006.
(68) J.P. Tosar-Rovira, “Estudio de la inmovilizacion de oligonucleotidos a eletrodos modificados de oro:polipirrol, y deteccion electroquimica de secuencias complementarias”, Tesis Lic. Bioquim., Universidad de la Republica, Montevideo, 2008.
(69) D. Zane, G. B. Appetecchi, C. Bianchini et al., An impedimetric glucose biosensor based on overoxidized polypyrrole thin film, Electroanal., 23, 1134 (2011).
(70) Y. Oztekin, A. Ramanaviciene, Z. Yazicigil et al., Direct electron transfer from glucose oxidase immobilized on polyphenanthroline-modified glassy carbon electrode, Biosens. Bioelectr., 26, 2541 (2011).
(71) R. Singh, Prospects of organic conducting polymer modified electrodes: Enzymosensors, Int. J. Electrochem., N 502707(2012).
(72) O. Korostynska, K. Arshak, E. Gill, A. Arshak, Review on state-of-the-art in polymer based pH sensors, Sensors, 7, 3027 (2008).
(73) R. Ojani, J.-B. Raoof, A.A. Maleki, S. Safshekan, Simultaneous voltammetric determination of ascorbic acid and dopamine at the surface of electrodes modified with selfassembled gold nanoparticle films, Chin. J. Cat., 35, 423 (2014).
(74) V. Tkach, V. Nechyporuk, P. Yagodynets, Eletropolimerizacao dos compostos heterociclicos. Modelos matematicos, Cienc. Tecn. Mat., 24, 54 (2012).
(75) V. Tkach, V. Nechyporuk, P. Yagodynets’, As eletropolimerizacoes galvanostatica e potenciodinamica (em voltagem constante), dos compostos heterociclicos e a sua descricao matematica, Tecn. Met. Mat. Min., 10, 249 (2013).
(76) V.V. Tkach, V.V. Nechyporuk, P.I. Yagodynets´, The mathematical description of “the polythiophene paradox” for soluble polymers, Geor. Chem. J., 11, 387 (2011).
(77) V. Tkach, V. Nechyporuk, P. Yagodynets, Estudio matematico de las condiciones de estabilidad en la polimerizacion electroquimica de compuestos heterociclicosen presencia de oxidantes, Afinidad, 70, 73 (2013).
(78) V. Tkach, V. Nechyporuk, P. Yagodynets, Descripcion matematica de la sintesis electroquimica de polimeros conductores en la presencia de surfactantes, Avances en Quimica, 8, 9 (2013).
(79) V.V. Tkach, V.V. Nechyporuk, P.I. Yagodynets´, V. Meslyuk-Yu, Mathematical model of the work of the mediated conducting polymer sensors with the use of the polymers of heterocyclic compounds, Revista de la Sociedad Quimica del Peru, 77, 259 (2011).
(80) V. Tkach, V. Nechyporuk, P. Yagodynets´, I. Rusnak, Los biosensores basados en los polimeros conductores, y la descripcion matematica de su desempeno, Orbital Electr. J. Chem., 4, 136 (2012).
(81) V. Tkach, B. Kumara Swamy, R. Ojani et al., Mecanismo de la oxidacion de omeprazol sobre el electrodo de carbono vitroso, modificado por polializarina, y su descripcion matematica, Orbital Electr. J. Chem., 7, 1 (2015).
(82) V. Tkach, B. Kumara Swamy, R. Ojani et al., Comportamento de paracetamol durante a sua oxidacao eletrocatalitica sobre poli(azul da anilina) e a sua descricao matematica, Rev. Colomb. Cienc. Quim. Farm., 44, 148 (2015).
(83) V. Tkach, V. Nechyporuk, P. Yagodynets, El estudio de estabilidad en el proceso del desempeno de los sensores y biosensores, basados en los polimeros conductores en medio fuertemente acido, Rev. Colomb. Cienc. Quim. Farm., 42, 30 (2013).
(84) V. Tkach, V. Nechyporuk, P. Yagodynets´, A descricao matematica do processo da deteccao de duas substancias com um grupo funcional comum pelos sensores e biossensores eletroquimicos, baseados em polimeros condutores, Rev. Colomb. Cienc. Quim. Farm., 42, 215 (2013).
(85) X. Bao, Metallopolymer electrochromic film prepared by oxidative electropolymerization of a Fe(II) complex with arylamine functionalized terpyridine ligand, Inorg. Chem. Commun., 38, 88 (2013).
(86) M. Sousa, E. J. Dallan, R. Bertazolli, Obtencao de um material adsorvente para ions de metais pesados, atraves da eletropolimerizacao de 2-mercapto- benzimidazol a superficie de carbono vitreo reticulado, Quim. Nova, 23, 326 (2000).
(87) D.N. Gritsan, “As investigacoes do processos eletrodicos e da eletrodeposicao dos metais. Cadmio” (publicado em russo), Kharkov, Vyshcha shkola, 1974.
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
Licencia
Derechos de autor 2016 Revista Colombiana de Ciencias Químico-Farmacéuticas

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
El Departamento de Farmacia de la Facultad de Ciencias de la Universidad Nacional de Colombia autoriza la fotocopia de artículos y textos para fines de uso académico o interno de las instituciones citando la fuente. Las ideas emitidas por los autores son responsabilidad expresa de estos y no de la revista.
Todo el contenido de esta revista, excepto dónde está identificado, está bajo una Licencia Creative Commons de Atribución 4.0 aprobada en Colombia. Consulte la normativa en: http://co.creativecommons.org/?page_id=13