Synthesis, characterization and in vitro antimicrobial screening studies of some pyridyl-coumarin compounds
Síntesis, caracterización y evaluación antimicrobiana in vitro de algunos derivados de piridil-coumarina
DOI:
https://doi.org/10.15446/rcciquifa.v46n1.67287Palabras clave:
Pyridyl-coumarin compounds, Gram positive bacteria, Gram negative bacteria, fungal strains, DMF, DMSO (en)Derivados de piridil-cumarina, bacterias Gram positivas, bacterias Gram negativas, cepas fúngicas, DMF, DMSO (es)
Descargas
La actividad antimicrobiana in vitro de algunos compuestos derivados de piridil-coumarina se evaluó frente a algunas cepas bacterianas y fúngicas en DMF y DMSO. Las piridil-cumarinas se sintetizaron en el laboratorio y sus estructuras se confirmaron por diferentes técnicas espectroscópicas, tales como IR, 1H NMR, 13C NMR y masas. Algunos de los compuestos que se obtuvieron presentaron buena actividad antibacteriana en ambos solventes.
Recibido: 11 de abril de 2016; Aceptado: 21 de abril de 2017
SUMMARY
In vitro antimicrobial screening of some pyridyl-coumarin compounds were done against some bacterial and fungal strains in DMF and DMSO. These pyridyl-coumarin compounds were synthesized in the laboratory and their structure was confirmed by different spectroscopic techniques such as IR, 1H NMR, 13C NMR and mass. Some of these compounds exhibited excellent antibacterial activity in both the solvents.
Keywords:
Pyridyl-coumarin compounds, Gram positive bacteria, Gram negative bacteria, fungal strains, DMF, DMSO.RESUMEN
La actividad antimicrobiana in vitro de algunos compuestos derivados de piridil-coumarina se evaluó frente a algunas cepas bacterianas y fúngicas en DMF y DMSO. Las piridil-cumarinas se sintetizaron en el laboratorio y sus estructuras se confirmaron por diferentes técnicas espectroscópicas, tales como IR, 1H NMR, 13C NMR y masas. Algunos de los compuestos que se obtuvieron presentaron buena actividad antibacteriana en ambos solventes.
Palabras clave:
Derivados de piridil-cumarina, bacterias Gram positivas, bacterias Gram negativas, cepas fúngicas, DMF, DMSO.INTRODUCTION
The infections causes by various microbes are dramatically increased during recent years [1]. Further, bacteria are becoming resistant to antimicrobial agents [2] so the effect of antimicrobial drugs available in the market is somewhat in doubt in future. These available antimicrobial drugs also have several drawbacks such asside effects, toxicity, low effectiveness and environmental issues [3,4]. Therefore, there is always need to develop new antimicrobials drugs for the treatment of infectious diseases [5].
Nitrogen and oxygen containing heterocyclic compounds like pyridine, coumarin etc., are always an attraction for researchers because of its efficiency towards various pharmacological usages [6, 7]. Literature survey shows that large array of coumarin derivatives possess a variety of biological activities such as antihistaminic [8], anticancer [9], anti-fungal [10], analgesic [11], anti-tubercular [12], antioxidant [13], antimicrobial [14], anti HIV [15], etc. They are also used as herbicides [16], neuroimaging agent [17], fluorescent whitening agent [18], organic sensitizers in dye sensitized solar cells [19], etc.
Owing to these interesting applications of pyridyl-coumarine derivatives, in the present work, some new pyridyl-coumarinecompoundshave been synthesized. The structure of these compounds was confirmed by different spectroscopic techniques. Further, in vitro screening of these compounds was carried out against bacterial as well as fungal strains in N,N-dimethylformamide (DMF) and dimethyl sulfoxide (DMSO).
EXPERIMENTAL AND MATERIALS
Materials
The solvents, DMF (LOBA Chemie Pvt. Ltd. & CAS No.- 68-12-2) and DMSO (LOBA Chemie Pvt. Ltd. & CAS No.- 67-68-5) used for the study of antimicrobial activity were of Analytical Reagent (AR) grade supplied by LOBA Chemie Pvt. Ltd. (Mumbai-INDIA) and were purified according to the standard reported procedure [20].
Synthesis
-
Synthesis of 2-benzylidenemalanonitrile derivatives (Int-1):
Equimolar mixture of different substituted benzaldehydes (Spectrochem Pvt. Ltd. & CAS No-100-52-7) and malanonitrile (LOBA Chemie Pvt. Ltd. & CAS No.- 109-77-3) in methanol (Allied Chemical Chemie Pvt. Ltd. & CAS No.- 67-56-1) was stirred at room temperature (RT) in presence of catalytic amount of piperidine (Sigma Aldrich & CAS No.- 110-89-4). The reaction progress was checked by analytical thin layer chromatography (TLC) (Performed on aluminum coated plates Gel 60F254 (E. Merck)) using (0.5:0.5 v/v-hexane: ethyl acetate) as mobile phase. After completion of reaction, the obtained solid was filtered, washed with cold methanol and was dried under vacuum. The obtained crude product was used in next step without further purification.
-
Synthesis of pyridyl coumarin derivatives:
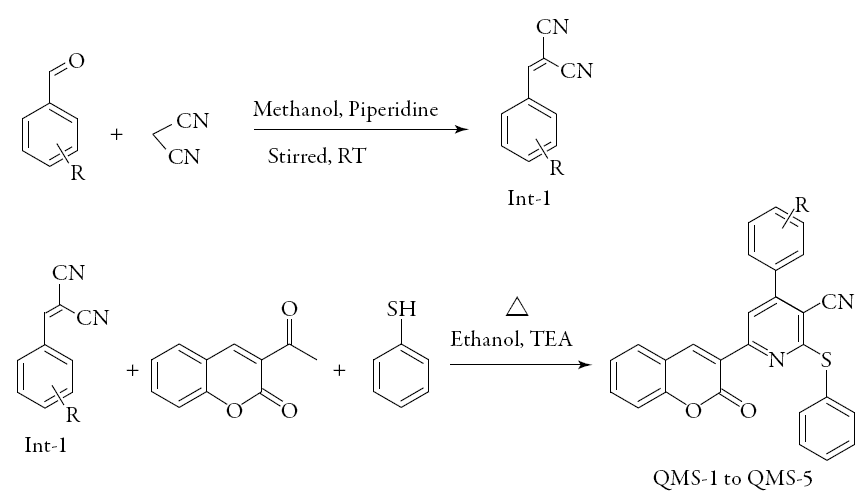
Equimolar mixture of 2-benzylidenemalanonitrile derivatives (Int-1), 3-acetylcouma-rin and thiophenol (Sigma Aldrich & CAS No.- 108-98-5) in ethanol was refluxed in presence of tri ethylamine (TEA) (Sigma Aldrich & CAS No.- 121-44-8) used as a catalyst. The progress of reaction was checked by TLC using (0.9: 0.1 v/v-chloroform: methanol) as a mobile phase. After completion of reaction, the temperature of reaction mass was allowed to decrease up to room temperature. The obtained solid was separated by filtration, washed with cold methanol and dried.
The reaction scheme is given in Figure 1. Following five pyridyl-coumarin derivatives were synthesized.
QMS-1: 4-(4-fluorophenyl)-6-(2-oxo-2H-chromen-3-yl)-2-(phenylsulfanyl)pyridine-3-carbo nitrile
QMS-2: 4-(4-bromophenyl)-6-(2-oxo-2H-chromen-3-yl)-2-(phenylsulfanyl)pyridine-3-carbo nitrile
QMS-3: 4-(2-chlorophenyl)-6-(2-oxo-2H -chromen-3-yl)-2-(phenylsulfanyl)pyridine-3-carbo nitrile
QMS-4: 4-(4-(dimethylamino)phenyl)-6-(2-oxo-2H -chromen-3-yl)-2-(phenylsulfanyl) pyridine -3-carbonitrile
QMS-5: 4-(2-hydroxyphenyl)-6-(2-oxo-2//-chromen-3-yl)-2-(phenylsulfanyl)pyri-dine-3-carbo nitrile
All the synthesized pyridyl-coumarin derivatives were crystallized from ethanol before use. The purity of these synthesized compounds was checked by GC-MS (SHI-MADZU Model-QP2010) and was found to be greater than 99.98 %.
Figure 1: Reaction scheme for the synthesis of pyridyl-coumarine compounds. Spectroscopy study
Spectroscopy study
The structure of the synthesized compounds was confirmed by FT-IR, 1H NMR, 13C NMR and mass spectral data. The IR spectra were taken on Fourier Transform InfraRed Spectrophotometer (SHIMADZU Model-IRaffinity-1S). 1H NMR and 13C NMR spectra were recorded on a Bruker AVANCE III at 400 MHzfrequency. In all the cases, NMR spectra were obtained in deuterated dimethyl sulfoxide (DMSO-d6) and in presence of tetra methyl silane used as an internal standard. The NMR signals are reported in δ ppm. Mass spectra were determined using direct inlet probe on a GC-MS (SHI-MADZU Model-QP2010) mass spectrometer.
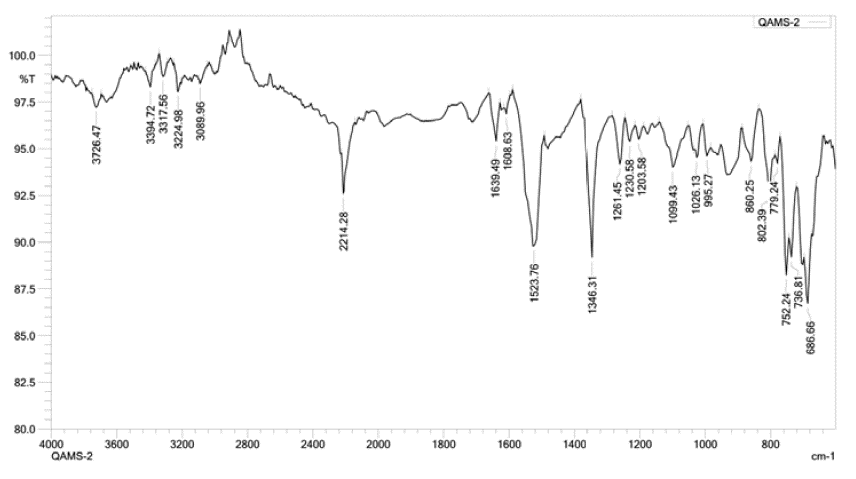
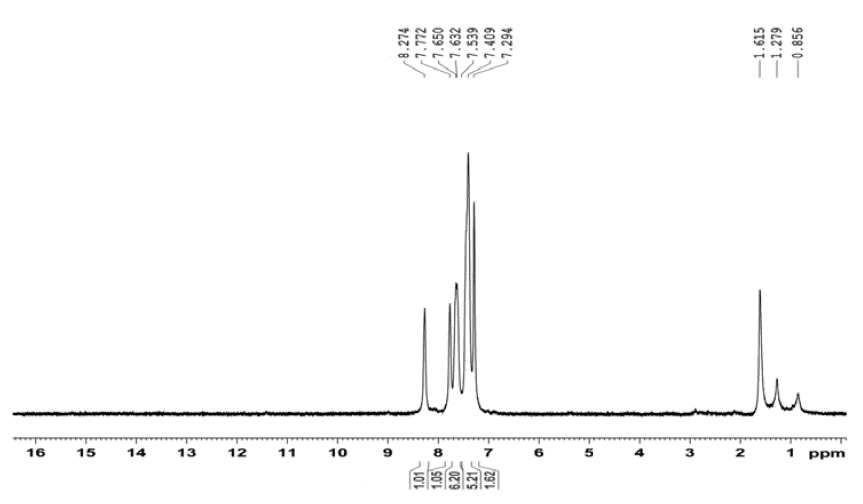
Figures 2 to 5 show FT-IR, 1H NMR, 13C NMR and mass spectra respectively for QMS-1.
Figure 2: IR spectrum of QMS-1
The melting points of compounds were measured by Differential Scanning Calorimeter (SHIMADZU Model-DSC-60) under nitrogen atmosphere (flow rate 100 ml/ min) and at 10 °C/min heating rate.
Figure 3: 1H NMR spectrum of QMS-1.
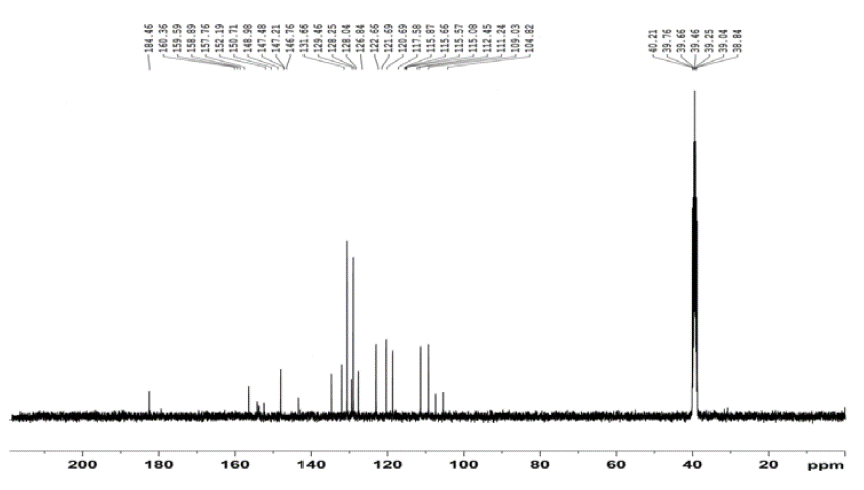
Figure 4: 13C NMR spectrum of QMS-1.
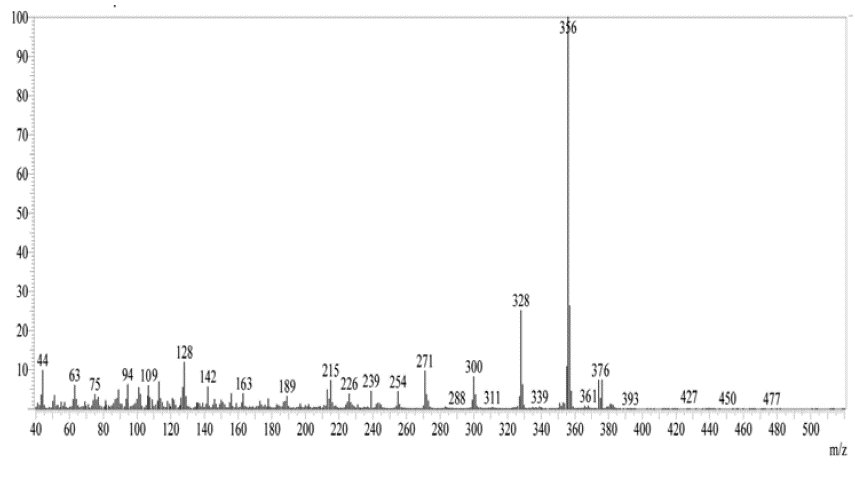
Figure 5: Mass spectrum of QMS-1.
Microorganisms tested
The studied microorganisms were obtained from National Chemical Laboratory, Pune, India and were maintained at 4°C. The selected Gram positive bacteria for the present study were Bacillus cereus ATCC11778 (BC), Corynebacterium rubrum ATCC14898 (CR), Bacillus subtilis ATCC6633 (BS) and Staphylococcus aureus ATCC29737 (SA). The Gram negative bacteria were Klebsiella pneumoniae NCIM2719(KP), Staphylo-coccus typhimurium ATCC23564 (ST), Escherichia coli NCIM2931 (EC), Pseudomo-nas aeruginosa ATCC9027 (PA). The selected fungal strains were Candida albicans ATCC2091 (CA), Candidaglabrata NCIM3448 (CG), Candida epicola NCIM3367 (CE) and Cryptococcus neoformans NCIM3542 (CN).
The agar well diffusion method [21] was used to study in vitro antimicrobial study of the synthesized compounds.For each compound in each solvent for a particular strain, the experiment was repeated three times. The average of these three values is graphically represented in Figures 6 to 8 along with uncertainty values.
RESULTS AND DISCUSSION
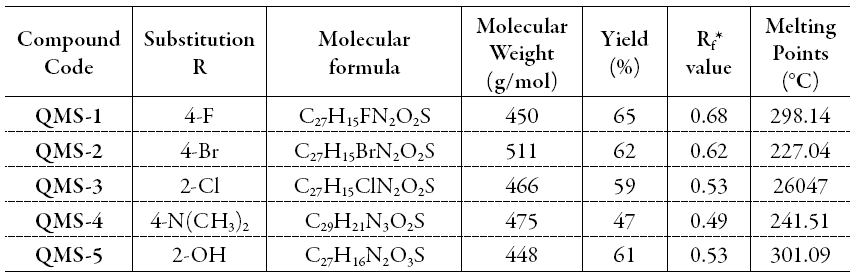
Table 1 shows the physical constant of synthesized compounds along with their side chain substitutions.
*0.9:0.1 v/v-chloroform: methanolTable 1: Physical properties of pyridyl-coumarine compounds
Spectral data
QMS-1:
IR (cm -1 ): 3394.72, 3317.56, 3224.98 (-OH stretching, H-bonded and/or -NH-stretching), 2934.67 (-CH- stretching), 2214.28 (-CN stretching), 1639.49, 1608.63 (C=Ostretching), 1523.76 (-CH- bending), 1346.31 (-CH- rock), 1261.45, 1230.58, 1203.58 (C-O stretching), 1099.43 (C-O stretching), 1026.13 (C-N stretching), 955.27, 806.25, 786.96 (substituted benzene).
1 H NMR (DMSO-d 6 , 400 MHz) (δ ppm): 7.2942-7.3183 (2H, doublet, -CH- aromatic, J= 9.64 Hz), 7409-7.539 (5H, multiplet, -CH- aromatic), 7.6324-7.6550 (6H, multiplet, -CH- aromatic), 7.7726 (1H, singlet, -CH- aromatic), 8.2743 (1H, -CH- aromatic).
13 CNMR (DMSO-d 6 , 400 MHz) (δ ppm): 104.82, 109.03, 111.24, 112.45, 115.08, 115.57, 115.66, 115.87, 117.58, 120.69, 121.69, 122.66, 126.84, 128.04, 128.25, 129.40, 131.66, 146.76, 147.21, 147.48, 148.98, 150.71, 152.59, 160.36, 184.46.
Mass (m/=): 450.
QMS-2:
IR (cm 1 ): 3352.67, 3339.37, 3228.29 (-OH stretching, H-bonded and/or -NH-stretching), 2978.09, 2885.51 (C-H stretching alkane), 2299.15, 2260.57, 2214.28 (-CN stretching), 1643.55 (C=Ostretching), 1592.61, 1481.33, 1404.18 (-CH-bending), 1369.46, 1346.31, 1327.03 (-CH- rock),1261.45, 1203.58 (C-O stretching), 1138.00, 1118.71, 1099.43 (C-C stretching), 1026.13 (C-N stretching),752.24, 671.23 (substituted benzene).
1 H NMR (DMSO-d 6 , 400 MHz) (δppm): 6.9637-6.9783 (2H, doublet, -CH- aromatic,), 7.3734-7.5493 (3H, multiplet, -CH- aromatic), 7.5538-7.6132 (2H, doublet, -CH - aromatic), 7.6263-7.7743 (6H, multiplet -CH- aromatic), 8.0958 (1H, singlet, -CH-aromatic), 8.2769 (1H, singlet, -CH- aromatic).
13 CNMR (DMSO-d 6 , 400 MHz) (δ ppm): 104.38, 109.63, 113.68, 112.96, 115.21, 115.54, 115.98, 127.36, 120.48, 121.13, 122.67, 126.38, 128.09, 129.48, 146.28, 147.79, 147.73, 148.29, 150.68, 153.26, 157.69, 158.26, 159.18, 160.09, 185.56.
Mass (m/(): 511.
QMS-3:
IR (cm 1 ): 3356.28, 3356.09, 3223.67 (-OH stretching, H-bonded and/or -NH-stretching), 2981.95, 2881.65 (C-H stretching alkane), 2349.30, 2299.15, 2214.28 (-CN stretching), 1631.78, 1604.77 (C=Ostretching), 1550.77, 1523.76, 1504.48 (n-H bending), 1481.33, 1442.75 (-CH- bending), 1369.46 (-CH- rock), 1296.16, 1222.87 (C-O stretching), 1157.29, 1099.43, 1026.13 (C-N stretching),779.24, 686.66 (substituted benzene).
1 HNMR (DMSO-d6, 400 MHz) (δ ppm): 7.2942-7.3372 (2H, doublet, -CH- aromatic), 7.4932-7.3721 (5H, multiplet, -CH- aromatic), 7.6348-7.6529 (6H, multiplet, -CH- aromatic), 7.2834 (1H, singlet, -CH- aromatic), 8.2758 (1H, -CH- aromatic).
13 CNMR (DMSO-d 6 , 400 MHz) (δ ppm): 104.48, 109.49, 113.29, 112.60, 115.02, 115.35, 115.89, 127.39, 120.67, 121.59, 122.27, 126.29, 127.46, 128.27, 129.58, 146.37, 147.68, 147.28, 148.48, 150.79, 153.26, 157.47, 158.79, 159.39, 160.29, 185.49.
Mass (m/( ): 466. QMS-4:
IR (cm 1 ): 3378.98, 3312.67, 3221.45 (-OH stretching, H-bonded and/or -NH-stretching), 2978.09 (C-H stretching alkane), 2349.30, 2299.15, 2210.42 (-CN stretching), 1708.93, 1639.49 (C=Ostretching), 1546.91, 1523.76, 1442.75 (-CH- bending), 1273.02, 1203.58, 1118.71, 1099.43 (C-O stretching), 1080.14, 1026.13 (C-N stretching), 756.10, 686.66, 671.23 (substituted benzene).
1 HNMR (DMSO-d 6 , 400 MHz) (δppm): 3.4657 (3H, singlet -CH3), 3.5783 (3H, singlet -CH3), 7.2958-7.3349 (2H, doublet, -CH- aromatic), 7.4939-7.3749 (5H, multiplet, -CH- aromatic), 7.6359-7.6527 (6H, multiplet, -CH- aromatic), 7.2583 (1H, singlet, -CH- aromatic), 8.2402 (1H, -CH- aromatic).
13 C NMR (DMSO-d 6 , 400 MHz) (δ ppm): 23.67, 24.38,104.84, 109.63, 113.28, 112.47, 115.02, 115.35, 115.78, 127.36, 120.48, 121.59, 122.62, 126.29, 128.27, 129.90, 146.37, 147.68, 147.28, 148.69, 150.39, 153.28, 157.90, 158.38, 159.18, 160.29, 185.49.
Mass (m/( ): 475.
QMS-5:
IR (cm 1 ): 3378.65, 3324.56, 3222.78 (-OH stretching, H-bonded and/or -NH-stretching), 2978.09, 2885.51 (C-H stretching alkane), 2384.02, 2349.30, 2299.15, 2214.28 (-CN stretching), 1708.93 (C=Ostretching), 1512.19 (N-H bending), 1442.75 (-CH- bending), 1369.46, 1346.31 (-CH- rock), 1273.02, 1234.44, 1199.72 (C-O stretching), 1118.71, 1068.56, 1026.13 (C-N stretching), 763.81, 740.67, 682.80 (substituted benzene).
1 HNMR (DMSO-d6, 400 MHz) (δ ppm): 7.2948-7.3338 (2H, doublet, -CH- aromatic), 7.4958-7.3782 (5H, multiplet, -CH- aromatic), 7.6359-7.6924 (6H, multiplet, -CH- aromatic), 7.2548 (1H, singlet, -CH- aromatic), 8.2423 (1H, -CH- aromatic).
13 CNMR (DMSO-d 6 , 400 MHz) (δppm): 104.58, 109.39, 113.68, 112.29, 115.89, 115.37, 115.69, 127.39, 120.97, 121.58, 122.69, 126.38, 127.36, 128.79, 129.90, 136.78, 146.36, 147.48, 147.68, 148.12, 150.45, 153.78, 157.90, 158.23, 159.56, 160.78, 185.12.
Mass (m/(): 448.
IR spectra
The IR spectrum of QMS-1 is given in Figure 2. The peaks observed around 32003500 cm-1 are due to stretching of -OH (H-bounded) and/or -NH- groups. The peak around 2929-2978 cm-1 is of -CH stretching of aromatic ring. The-CN stretching is observed around 2300-2200 cm-1. The peaks for -C=O and C-H stretching are obtained around 1600-1700 cm-1 and 1550-1600 cm-1 respectively whereas alkane C-H bending peak is observed around 1469-1490 cm-1. The peaks observed around 1300-1334 cm-1 are due to C-O stretching of ester group and/or ether group. The -CN stretching is observed around 1250-1050 cm-1.
1 HNMR spectra
The 1H NMR spectrum of QMS-1 is shown in Figure 3. For aromatic protons, peaks are between 7.2940 to 7.6500 δ ppm with their appropriate multiplicity. Two singlet peaks of aromatic proton (=CH-) are observed at 7.7120 and 8.2740.
All the 1H NMR peaks suggests that compounds are synthesized successfully.
13 CNMR spectra
Figure 4 shows the 13C NMR spectrum of compound QMS-1. The aromatic carbons of phenyl rings are shown between 104.82 to 184.46 δ ppm with their appropriate multiplicity.
Mass spectra
Figure 5 shows the mass spectrum of compound QMS-1. From mass fragmentation, the structures of synthesized compounds are confirmed.
Antimicrobial activity
Figure 6 shows the zone of inhibition for the studied compounds against Gram positive bacteria in DMF and DMSO along with two standard antibiotics. It is observed that against Bacillus cereus, all the studied compounds exhibited inhibition (except QMS-4 in DMSO) in both DMF and DMSO. However, in DMF, QMS-5 showed maximum inhibition and this value is higher than tetracyclin but lower than Chloramphenicol.
Figure 6: Antibacterial activity of synthesized compounds against Gram positive bacteria in [A] DMF and [B] DMSO. [QMS-1, (); QMS-2, (); QMS-3, (); QMS-4, (); QMS-5, (); Chloram-phenicol (); Tetracyclin ()].
However in DMSO, QMS-1 showed maximum inhibition against Bacillus cereus but lower than both the antibiotics.
In DMF, QMS-1, QMS-3 and QMS-5 showed inhibition against Corynebacterium rubrum whereas in DMSO only QMS-1 was found to be effective against this bacterial strain. Against Bacillus subtilis in DMF, all the studied compounds exhibited significant inhibition. However in DMSO, QMS-1, QMS-2 and QMS-3 exhibited inhibition. In DMF, against Staphylococcus aureus only QMS-1 showed inhibition whereas in DMSO, none of compounds was found to be effective.
This suggests that inhibition depends on solvent, structure of compound and bacterial strain. In the present work, all the studied compounds have the same central moiety but different substitution groups as listed in Table 1. QMS-5 contains 2-hydroxy group which shows maximum inhibition against Bacillus cereus in DMF than other substitutions. However in DMSO, 4-fluoro (as in QMS-1) group showed significant inhibition against this bacterial strain. In DMF, against Bacillus subtilis and Staphylococcus aureus, again 4-fluoro group (as in QMS-1) is most effective. However, in DMF, against Bacillus subtilis, 4-bromo (as in QMS-2) and 4-N,N-dimethylamine (as in QMS-4) groups are also found to be effective almost up to same extent. None of the groups are found to be effective against Staphylococcus aureus in DMF and DMSO except QMS-1 containing 4-fluoro group in DMF.
Thus, for the studied compounds, DMF is better solvent against selected Gram positive bacteria.
Figure 7 shows the zone of inhibition against Gram negative bacteria in both DMF and DMSO. In DMF, against Klebsiella pneumoniae, compounds QMS-1 containing 4-fluoro, QMS-4 containing 4-VJVT-dimethylamine and QMS-5 containing 2-hydroxy groups exhibited significant inhibition then antibiotic chloramphenicol and inhibition is maximum for QMS-1. In DMSO, compounds QMS-1, QMS-2 and QMS-3 showed significant inhibition against Klebsiella pneumonia and inhibition of QMS-1 is almost to the same extent as chloramphenicol. Thus, in DMF and DMSO, 4-fluoro group is found to be most effective against Klebsiella pneumonia.
Figure 7: Antibacterial activity of synthesized compounds against Gram negative bacteria in [A] DMF and [B] DMSO. [QMS-1, (); QMS-2, (); QMS-3, (); QMS-4, (); QMS-5, (); Chloram-phenicol (); Tetracyclin ()].
Against Staphylococcus typhimurium, only QMS-1 and QMS-5 having 4-fluoro and 2-hydroxy groups respectively showed inhibition in DMF whereas in DMSO except QMS-2, other compounds exhibited inhibition. Thus, in DMSO 4-bromo group is not effective against this strain. Against Escherichia coli in DMF, only QMS-1 showed inhibition whereas in DMSO, compounds QMS-3 and QMS-5 containing 2-chloro and 2-hydroxy groups respectively showed inhibition. Against Pseudomonas aeruginosa, none of the studied compounds are effective in DMF whereas in DMSO, only 4-bromo (as in QMS-2) group showed inhibition and up to same extent with tetracyclin.
Hence, the synthesized compounds showed better activity in DMSO against Gram negative bacteria.
Figure 8 shows the zone of inhibition for the studied compounds and two antibiotics such as nystatin and itroconazolagainst selected fungal strain in DMF and DMSO. Against Candida albicans, in DMF except QMS-3, other compounds exhibited significant inhibition and QMS-5 containing 2-hydroxy group showed maximum inhibition. However, in DMSO none of the studied compounds are found to be effective against this fungal strain. In DMF, none of compound was found to inhibit Candida glabrata and Candida epicola. Whereas in DMSO, only QMS-3 showed some inhibitionagainst Candida glabrata. Against Candida epicola, there was no inhibition by any of the compound in DMSO. Against Cryptococcus neoformans in DMF, only QMS-1 having 4-fluoro group showed inhibition whereas in DMSO, none of the studied compounds was effective.
Figure 8: Antifungal activity of synthesized compounds in [A] DMF and [B] DMSO. [QMS-1, (); QMS-2, (); QMS-3, (); QMS-4, (); QMS-5, (); Nystatin (); Itroconazol ()].
Overall, Staphylococcus aureus, Candida albicans, Candida epicola and Cryptococcus neoformans are most resistant strains.
CONCLUSIONS
The inhibition against bacterial and fungal strains depends upon the solvent, structures of compound and strain. For the selected Gram positive bacterial and fungal strains, DMF is better solvent whereas for Gram negative bacteria, DMSO is better solvent. Compounds having halogen groups are more effective against selected microbial strains. Staphylococcus aureus, Candida albicans, Candida epicola and Cryptococcus neoformans are the most resistant strains.
REFERENCES
Referencias
(1) A.L. Demain, S. Sanchez, Microbial drug discovery: 80 years of progress (review
article), J. Antibiotics, 62, 5 (2009).
(2) U. Kalidhar, A. Kaur, An overview on some benzimidazole and sulphonamide
derivatives with anti-microbial activity, Res. J. Pharm. Biol. Chem. Sci., 2, 1116
(2011).
(3) R.J. Fair, Y. Tor, Antibiotics and bacterial resistance in the 21st century, Perspect
Med. Chem., 6, 25 (2014).
(4) S.B. Singh, Confronting the challenges of discovery of novel antibacterial agents,
Bioorg. Med. Chem. Lett., 24, 3683 (2014).
(5) S.S. Kanj, Z.A. Kanafani, Current concept in antimicrobial therapy against
resistant Gram negative organisms: extended spectrum ß-lactamase producing
enterobacteriaceae, carbapenem resistant enterobacteriaceae and multidrug
resistant Pseudomonus aeruginosa, Mayo Clin. Proc., 86, 250 (2011).
(6) N.C. Desai, H.M. Satodiya, K.M. Rajpara, V.V. Joshi, H.V. Vaghani, Microwave
assisted synthesis of new coumarin based 3-cyanopyridine scaffolds bearing sulphonamide
group having antimicrobial activity, Ind. J. Chem., 52B, 904 (2013).
(7) J. Sahoo, S.K. Mekap, P.S. Kumar, Synthesis, spectral characterization of some
new 3-heteroaryl azo 4-hydroxy coumarin derivatives and their antimicrobial
evaluation, J. Taibuh Uni. Sci., 9, 187 (2015).
(8) D.R. Buckie, D.J. Outred, H. Smith, B.A. Spicer, N-benzylpiperazino derivatives
of 3-nitro-4-hydroxycoumarin with H1 antihistamine and mast cell stabilizing
properties, J. Med. Chem., 27, 1452 (1984).
(9) T. Nasr, S. Bondock, M. Youns, Anticancer activity of new coumarin substituted
hydrazide-hydrazone derivatives, Eur. J. Med. Chem., 76, 539 (2014).
(10) A.A. Al-Amiery, A.A.H. Kadhum, A.B. Mohamad, Antifungal activities of new
coumarins, Molecules, 17, 5713 (2012).
(11) B.S. Jayashree, S. Nigam, A. Pai, P.V.R. Chowdary, Overview on the recently
developed coumarinyl heterocycles as useful therapeutic agents, Arabian J.
Chem., 7, 885 (2014).
(12) A. Manvar, A. Malde, J. Verma, V. Virsodia, A. Mishra, K. Upadhyay, H. Achrya,
E. Coutinho, A. Shah, Synthesis anti-tubercular active and 3D QSAR study
of coumarin-4-acetic acid benzylidene hydrazides, Eur. J. Med. Chem., 43, 2395
(2008).
(13) R.K. Arora, N. Kaur, Y. Bansal, G. Bansal, Novel coumarin-benzimidazole derivatives
as antioxidant and safer anti-inflammatory agent, Acta Pharm. Sinica B, 4,
(2014).
(14) J. Sahoo, S.K. Mekap, P.S. Kumar, Synthesis, spectral characterization of some
new 3-heteroaryl azo 4-hydroxy coumarin derivatives and their antimicrobial
evaluation, J. Taibah Univ. Sci., 9, 187 (2015).
(15) D. Yu. M. Suzuki, L. Xie, S. L. Morris-Natschke, K. H. Lee, Recent progress in
the development of coumarin derivatives as potent anti-HIV agent, Med. Res.
Rev., 23, 322 (2003).
(16) U.C. Mashelkar, S. S. Tungare, S. Bhagat, Studies on synthesis of various N-substituted
derivatives with different heterocycles and their herbicidal activity, Ind.
J. Chem., 50B, 315 (2011).
(17) P.A. Vadola, D. Sames, Catalytic coupling of arene C-H bonds and alkynes for
the synthesis of coumarins: substrate scope and application to the development
of neuroimaging agents, J. Org. Chem., 77, 7804 (2012).
(18) R. Rajagopal, V.U. Shenoy, S. Padmanabhan, S. Sequeria, S. Seshadri, Synthesis
of fluorescent 2,3-fused coumarin derivatives, Dyes Pigments, 13, 167 (1990).
(19) R. Sanchez-de-Armas, M.A.S. Miguel, J. Oviedo, J.F. Sanz, Coumarin derivatives
for dye sensitized solar cells: a TD-DFT study, Phys. Chem. Chem. Phys., 14,
, (2012).
(20) J.A. Riddick, W.B. Bunger, T. Sakano, “Organic solvents-physical properties
and methods of purification, techniques of Chemistry”, II, A Wiley-Interscience
Publication, John Wiley, New York, 1986.
(21) J. Parekh, P. Inamdar, R. Nair, S. Baluja, S. Chanda, Synthesis and antibacterial
activity of some Schiff bases derived from 4-aminobenzoic acid, J. Serb. Chem.
Soc., 70, 1155 (2005).
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
Licencia
Derechos de autor 2017 Revista Colombiana de Ciencias Químico-Farmacéuticas

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
El Departamento de Farmacia de la Facultad de Ciencias de la Universidad Nacional de Colombia autoriza la fotocopia de artículos y textos para fines de uso académico o interno de las instituciones citando la fuente. Las ideas emitidas por los autores son responsabilidad expresa de estos y no de la revista.
Todo el contenido de esta revista, excepto dónde está identificado, está bajo una Licencia Creative Commons de Atribución 4.0 aprobada en Colombia. Consulte la normativa en: http://co.creativecommons.org/?page_id=13




![Antibacterial activity of synthesized compounds against Gram positive bacteria in [A] DMF and [B] DMSO. [QMS-1, (); QMS-2, (); QMS-3, (); QMS-4, (); QMS-5, (); Chloram-phenicol (); Tetracyclin ()].](https://revistas.unal.edu.co/index.php/rccquifa/article/download/67287/version/53327/63235/358357/0034-7418-rccqf-46-01-00005-gf6.png)
![Antibacterial activity of synthesized compounds against Gram negative bacteria in [A] DMF and [B] DMSO. [QMS-1, (); QMS-2, (); QMS-3, (); QMS-4, (); QMS-5, (); Chloram-phenicol (); Tetracyclin ()].](https://revistas.unal.edu.co/index.php/rccquifa/article/download/67287/version/53327/63235/358358/0034-7418-rccqf-46-01-00005-gf7.png)
![Antifungal activity of synthesized compounds in [A] DMF and [B] DMSO. [QMS-1, (); QMS-2, (); QMS-3, (); QMS-4, (); QMS-5, (); Nystatin (); Itroconazol ()].](https://revistas.unal.edu.co/index.php/rccquifa/article/download/67287/version/53327/63235/358362/0034-7418-rccqf-46-01-00005-gf8.png)














