Sistema endocanabinoide: modificando los factores de riesgo cardiovascular
Endocannabinoid system: modifying cardiovascular risk factors
Palabras clave:
endocanabinoides, tabaquismo, obesidad, factores de riesgo, receptores canabinoides, leptina, rimonabant. (es)endocannabinoid, smoking, obesity, risk factor, receptors cannabinoid, leptin, rimonabant (en)
La necesidad de alcanzar un tratamiento óptimo para el tabaquismo, la obesidad y sus comorbilidades, conocidos factores de riesgo cardiovascular, ha fomentado la búsqueda de objetivos terapéuticos novedosos. Es el caso del sistema endocanabinoide, involucrado en diversos fenómenos fisiológicos entre los que se encuentran el refuerzo de ciertos comportamientos y la regulación del apetito.
La sobreactivación de este sistema altera la homeostasis corporal predisponiendo a dependencias o a un aumento en la ingesta alimentaria, lo que puede traducirse en tabaquismo u obesidad. La intervención farmacológica sobre el sistema endocanabinoide puede contribuir al manejo de estos factores de riesgo cardiovascular, teniendo en cuenta que a tales beneficios se suman otros independientes de la suspensión del tabaquismo o la reducción de peso, como el aumento del colesterol de alta densidad, la disminución de triglicéridos y la mejoría del control glucémico en pacientes con diabetes.
Ensayos clínicos controlados aleatorizados adelantados en poblaciones con diferentes características, han evaluado la utilidad de la regulación farmacológica del sistema endocanabinoide; confirmando su eficacia en personas con factores de riesgo cardiovascular establecidos.
The need for an optimal treatment for smoking, obesity and their comorbidities, well-known cardiovascular risk factors; has prompted the search for novel therapeutic targets. This is the case of the endocannabinoid system, involved in several physiological phenomena including the reinforcement of certain behaviors and the regulation of appetite.
ACTUALIZACION
John Edwin Feliciano Alfonso 1 , Carlos Olimpo Mendivil Anaya 2Iván Darío Sierra Ariza 3
1. MD. Médico con entrenamiento en Lípidos y Diabetes, División de Lípidos y Diabetes, Facultad de Medicina, Universidad Nacional de Colombia, Bogotá.
2. MD. Médico con entrenamiento en Lípidos y Diabetes, Especialista en Estadística, División de Lípidos y Diabetes, Facultad de Medicina, Universidad Nacional de Colombia, Bogotá.
3. MD, PhD. Especialista en Nutrición y Diabetes, Profesor Titular y Maestro Universitario. Coordinador División de Lípidos y Diabetes, Facultad de Medicina, Universidad Nacional de Colombia, Bogotá.
Correspondencia: john.felicianoa@gmail.com
Resumen
La necesidad de alcanzar un tratamiento óptimo para el tabaquismo, la obesidad y sus comorbilidades, conocidos factores de riesgo cardiovascular, ha fomentado la búsqueda de objetivos terapéuticos novedosos. Es el caso del sistema endocanabinoide, involucrado en diversos fenómenos fisiológicos entre los que se encuentran el refuerzo de ciertos comportamientos y la regulación del apetito.
La sobreactivación de este sistema altera la homeostasis corporal predisponiendo a dependencias o a un aumento en la ingesta alimentaria, lo que puede traducirse en tabaquismo u obesidad. La intervención farmacológica sobre el sistema endocanabinoide puede contribuir al manejo de estos factores de riesgo cardiovascular, teniendo en cuenta que a tales beneficios se suman otros independientes de la suspensión del tabaquismo o la reducción de peso, como el aumento del colesterol de alta densidad, la disminución de triglicéridos y la mejoría del control glucémico en pacientes con diabetes.
Ensayos clínicos controlados aleatorizados adelantados en poblaciones con diferentes características, han evaluado la utilidad de la regulación farmacológica del sistema endocanabinoide; confirmando su eficacia en personas con factores de riesgo cardiovascular establecidos.
Palabras clave: endocanabinoides, tabaquismo, obesidad, factores de riesgo, receptores canabinoides, leptina, rimonabant.
Feliciano JE, Mendivil CO, Sierra ID. Sistema endocanabinoide: modificando los factores de riesgo cardiovascular. Rev Fac Med Univ Nac Colomb. 2006; 54: 269-282.
Summary
The need for an optimal treatment for smoking, obesity and their comorbidities, well-known cardiovascular risk factors; has prompted the search for novel therapeutic targets. This is the case of the endocannabinoid system, involved in several physiological phenomena including the reinforcement of certain behaviors and the regulation of appetite.
Introducción
Las enfermedades cardiovasculares (ECV) constituyen un grave problema de salud pública en el mundo. Se estima que el 30 por ciento del total de las muertes actualmente en todo el mundo se deben a ECV y se prevé que éste número se incrementará a través del tiempo a medida que las poblaciones envejezcan (1). Algunos factores de riesgo cardiovascular pueden modificarse favorablemente, en especial, con terapia farmacológica. Es el caso de la hipertensión arterial (2,3), las dislipidemias (4-6) o la diabetes mellitus tipo 2 (7-9). Sin embargo, aún existen opciones limitadas para tratar farmacológicamente otros factores modificables de riesgo cardiovascular como el tabaquismo o la obesidad, que en conjunto representan las principales causas prevenibles de muerte y discapacidad, y cuyas prevalencias están llegando a proporciones alarmantes: en la población adulta estadounidense el 65.1 por ciento tiene sobrepeso y un 30.4 por ciento presenta obesidad, mientras que el 22.5 por ciento de la población total de todas las edades tiene el hábito de fumar (10).
Colombia no es ajena a estos problemas. Un estudio realizado en población adulta bogotana encontró una prevalencia de tabaquismo de 11.8 por ciento y una prevalencia de 46.9 por ciento de sobrepeso: 37.3 por ciento para preobesidad (índice de masa corporal entre 25 y 29.9 Kg/m 2 ) y 9.6 por ciento para obesidad (índice de masa corporal mayor o igual a 30 Kg/m 2 ) (11).
Ante este panorama, el sistema endocanabinoide, gracias a sus diversas acciones fisiológicas, ha emergido durante la última década como un blanco farmacológico atractivo para el tratamiento no sólo de la obesidad y el tabaquismo, sino también de otros factores de riesgo cardiovascular asociados a estas alteraciones.
Esta revisión se enfoca en la fisiología del sistema endocanabinoide en cuanto a su relación con el tabaquismo y la obesidad, para finalizar con la evidencia que sustenta su modulación en el tratamiento de estas dos entidades y sus factores de riesgo asociados.
Fisiología del sistema endocanabinoide
Además de sus efectos sicoactivos, desde hace varios siglos se conoce la capacidad de la marihuana ( Cannabis sativa ) de estimular notablemente el apetito (12). Esta planta contiene más de 60 canabinoides, los cuales comprenden una familia de compuestos con estructuras de anillos triciclícos caracterizados por un anillo fenol, un anillo central pirano y un anillo ciclohexil monoinsaturado (13), de los cuales se destaca el D 9-tetrahidrocannabinol ( D 9-THC) (Figura 1). De hecho, un derivado sintético del D 9-THC, el dronabinol, se ha aprobado para el tratamiento de pacientes con anorexia asociada al síndrome de inmunodeficiencia adquirida (SIDA) con el fin de estimular el apetito y prevenir la caquexia (14).
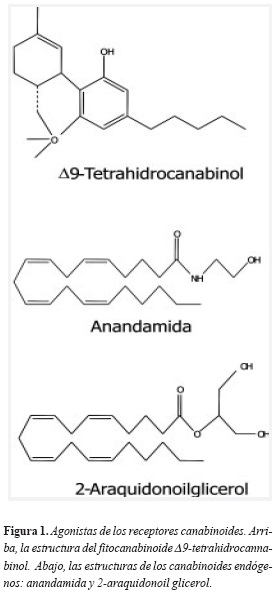
Los canabinoides naturales (fitocanabinoides) y sintéticos actúan sobre dos tipos de receptores bien caracterizados denominados receptores CB1 y CB2, que tienen sus propios ligandos endógenos: los endocanabinoides.
Canabinoides endógenos: todos los endocanabinoides derivan de los fosfolípidos que hacen parte de las membranas celulares, por lo tanto son moléculas lipídicas. Los endocanabinoides de interés en relación con el apetito y con las conductas de recompensa (y por tanto con el tabaquismo), son el 2-araquidonoilglicerol (2-AG) y la n-araquidonoiletanolamina, denominada anandamida, nombre proveniente del sánscrito ananda que significa alegría o dicha interna, quizás debido a los conocidos efectos eufóricos del fitocanabinoide D 9-THC(15). La biosíntesis de estas dos moléculas se esquematiza detalladamente en las figura 2 y figura 3.
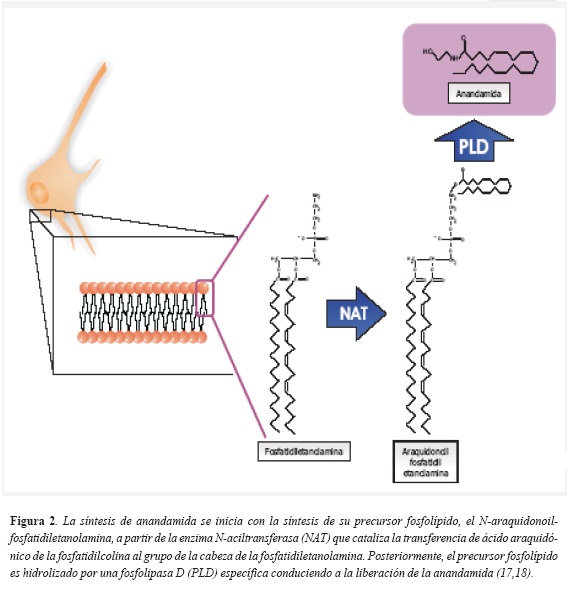
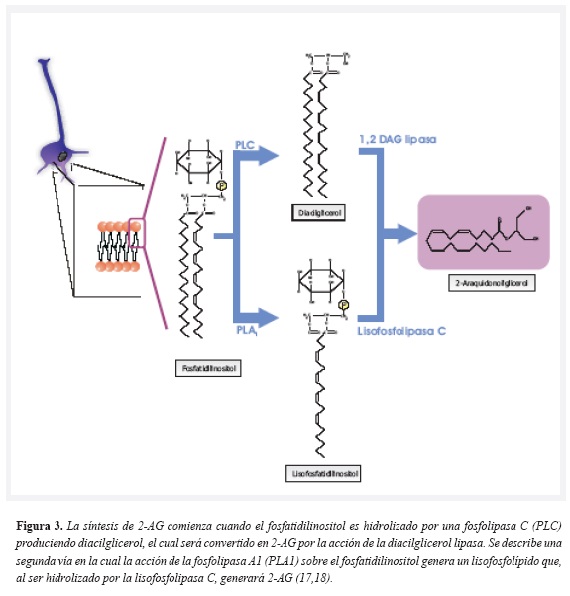
Al sintetizarse primordialmente (pero no exclusivamente) en las células neuronales, los endocanabinoides se han considerado como neurotransmisores no clásicos, puesto que no se almacenan en vesículas sinápticas, sino que son producidos y liberados "a demanda" (16). La liberación de endocanabinoides se produce en respuesta a la despolarización de la membrana y al influjo de Ca ++ dentro de la célula, tal como sucede con los neurotransmisores clásicos, para luego unirse a su receptor y posteriormente ser inactivados a través de mecanismos de recaptación y de degradación enzimática. La enzima fundamental en la degradación de endocanabinoides se denomina hidrolasa de amidas de ácidos grasos (Fatty acid amide hydrolase-FAAH) (17).
Receptores endocanabinoides : son receptores con siete regiones transmembrana, acoplados a proteínas de unión a GTP (proteínas G inhibitorias [Gi]). Los receptores endocanabinoides se encuentran localizados en diferentes regiones. El primer receptor endocanabinoide descubierto, denominado CB1, se distribuye principalmente en el sistema nervioso central (SNC) sobre las terminales nerviosas presinápticas. El segundo receptor endocanabinoide, denominado CB2, se localiza especialmente en las células del sistema inmune y aparentemente no cumple ningún papel dentro de la regulación del apetito (15).
La unión de los endocanabinoides a su receptor CB1 conlleva a la inhibición de adenil ciclasa, disminución de los niveles de AMPc, apertura de canales de K + , hiperpolarización celular y cierre de canales de Ca ++ , impidiendo su entrada a la célula (18). Como consecuencia, el efecto neto de la activación del receptor CB1 por los endocanabinoides es menor generación de potenciales de acción en la neurona presináptica, inducida por un mediador liberado por la neurona postsináptica. Así mismo, la menor disposición de Ca ++ intracelular evitará la liberación de neurotransmisores en la neurona presináptica. De esta manera, la activación del receptor CB1 por los endocanabinoides causa un efecto presináptico inhibitorio, suprimiendo la liberación de neurotransmisores tanto excitatorios (glutamato), como inhibitorios (GABA) (18).
Relajar, comer, olvidar y proteger
Entre las diversas regiones del SNC en las que se expresa el receptor CB1 están los ganglios basales, el hipotálamo, el giro dentado y la médula espinal, proporcionándonos pistas sobre sus diversas funciones (19), entre las cuales se encuentran analgesia, inhibición de la consolidación de la memoria e inhibición de la actividad motora, por lo que se considera que el sistema endocanabinoide es un sistema de recuperación al permitir la relajación, el olvido y la protección.
De importancia es la expresión de los receptores endocanabinoides en el hipotálamo, estructura relacionada con la regulación de la energía y el apetito. Sin embargo, los receptores CB1 también se localizan en órganos periféricos relacionados con el mismo fenómeno como el tracto gastrointestinal, el hígado, el músculo esquelético y el tejido adiposo (20). Sin embargo, la expresión de receptores CB1 en el SNC es más acentuada que en cualquier otro órgano periférico (Figura 4).
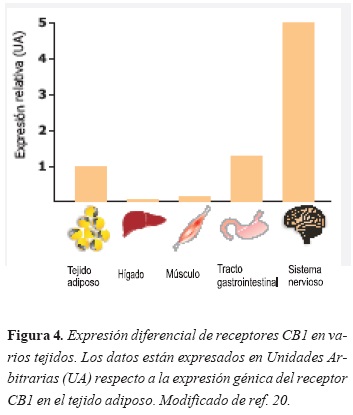
Así mismo, estos receptores se encuentran en el sistema mesolímbico dopaminérgico, un conjunto de estructuras cerebrales implicadas en el reforzamiento de comportamientos, como las adicciones (incluye al tabaquismo) y también en la motivación por la ingesta de alimentos.
Comer: endocanabinoides y su papel en la alimentación
Mecanismos centrales: se ha demostrado que la administración central (incluyendo la vía intrahipotalámica) de anandamida y 2-AG produce hiperfagia en roedores de experimentación y que esta propiedad es bloqueada por un antagonista selectivo de los receptores CB1 (21,22), demostrando que la acción orexigénica de los agonistas canabinoides puede bloquearse y consecuentemente disminuirse la ingesta y el peso corporal.
Los principales neurotransmisores implicados en la regulación hipotalámica del apetito y del gasto energético son la hormona alfa-estimulante de los melanocitos ( a -MSH) y el transcripto relacionado con cocaína y anfetaminas (CART) como señales anorexígenas. El neuropéptido Y (NPY), la orexina y la proteína relacionada con Agouti (AgRP) como su contraparte orexigena (23). En este último grupo podemos incluir a los endocanabinoides por sus propiedades antes descritas, si bien se diferencian de los demás en su acción retrógada, su producción "a demanda" y su naturaleza lipídica, no peptídica.
Las neuronas del núcleo paraventricular del hipotálamo también presentan receptores para hormonas de origen periférico que igualmente están relacionadas con la regulación del peso corporal, como la leptina producida por el tejido adiposo en proporción a su masa y al balance energético, que actua como una señal de adiposidad para indicar que ya existe suficiente tejido adiposo y se debe disminuir la ingesta e incrementar el gasto calórico (23). En modelos animales de obesidad como los ratones ob/ob (carentes de leptina) o db/db (con defectos en su receptor y señalización) se han encontrado niveles incrementados de endocanabinoides intrahipotalámicos. Cuando a estos animales se les administra un antagonista CB1 disminuyen su ingesta y retardan la ganancia de peso (24).
Esto nos indica que al parecer en la obesidad el sistema endocanabinoide está sobreactivado permanentemente en el hipotálamo, causando un aumento en la ingesta de alimentos, y que el uso de antagonistas de los receptores CB1 puede ser útil en el tratamiento de la obesidad.
Mecanismos periféricos: como se ha mencionado, los receptores CB1 también se localizan en órganos periféricos relacionados con la regulación del peso, del apetito y del gasto energético, entre ellos el tejido adiposo en el cual se ha demostrado que la activación in vitro del receptor CB1 incrementa la actividad de la lipoproteinlipasa estimulando la lipogénesis (25), lo cual conducirá grosso modo a que el tejido adiposo se acumule, aumentando así el perímetro de la cintura y produciendo ganancia de peso.
Los receptores CB1 también se encuentran localizados en el hígado. La activación de los receptores CB1 hepáticos en ratones (por aumento de los niveles de anandamida debido a una dieta alta en grasas) incrementa la expresión del factor de transcripción SREBP-1c y de sus genes asociados: acetil-CoA carboxilasa-1 y sintetasa de ácidos grasos. Por tanto, la activación de los receptores CB1 hepáticos incrementa la síntesis de ácidos grasos, los niveles plasmáticos de triglicéridos, el peso corporal y contribuye al desarrollo del hígado graso (26). Adicionalmente estos animales tenían una disminución en los niveles de adiponectina y un aumento en los niveles de leptina e insulina (12,27), hallazgo consistente con otras investigaciones que demuestran que animales obesos por exposición a una dieta alta en grasas también tienen altos niveles de leptina, de insulina y de glucosa, los cuales se reducen, al igual que el peso corporal, luego de la administración de un antagonista de los receptores CB1 (27). La expresión del gen de adiponectina en el tejido adiposo de las ratas obesas fa/fa se incrementó luego de la administración del antagonista de los receptores CB1(28), lo cual es importante si se tiene en cuenta que los efectos de esta hormona son protectores o antiaterogénicos. De igual manera, se ha demostrado que este antagonista de los receptores CB1 incrementa la captación de glucosa en el músculo esquelético aislado de ratones obesos ob/ob , sugiriendo un papel clave sobre el metabolismo de carbohidratos (29).
Proteger: endocanabinoides y su papel en el refuerzo de conductas
Sistema mesolímbico dopaminérgico: los receptores CB1 también se han identificado en las regiones de este sistema cerebral de recompensa relacionado con el refuerzo de comportamientos tales como las adicciones o la motivación para la búsqueda de comida. El tabaquismo, más que un hábito, es considerado una adicción (30) y se ha demostrado que la nicotina, el principal componente del tabaco y responsable de sus propiedades adictivas afecta el sistema endocanabinoide.
Conformado entre otras estructuras, por el área tegmentaria ventral (ATV), el núcleo accumbens (Nacc) y la corteza frontal medial (CFM), el sistema mesolímbico dopaminérgico (SMD) se encuentra bajo influencia del sistema endocanabinoide tal como lo demuestra la inhibición de la conducta motivacional tras la administración de un antagonista endocanabinoide en animales de experimentación (18).
Las neuronas dopaminérgicas del ATV proyectan sus axones hacia la CFM y hacia el Nacc dentro del fascículo prosencefálico medial. Las proyecciones de las neuronas GABA-érgicas en el núcleo accumbens se dirigen de forma retrógrada también por el fascículo prosencefálico medial a las neuronas del ATV (31). Es a través de la liberación de dopamina que en el Nacc se refuerzan los comportamientos y esta acción es modulada tanto por neurotransmisores inhibitorios (GABA) como excitatorios (glutamato) (Figura 5).
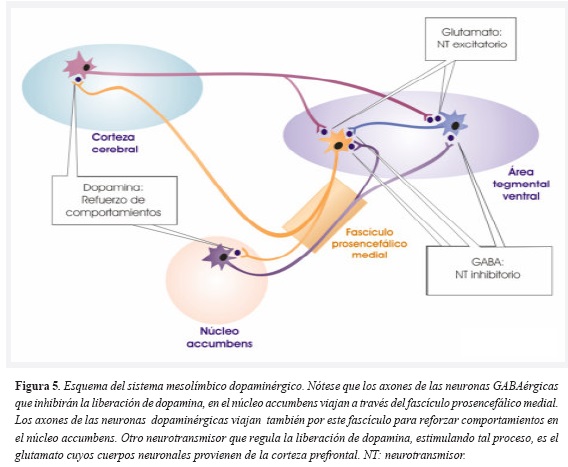
Además de la importancia del SMD para la motivación en la búsqueda de alimentos, es en este sistema donde radica la lógica para tratar el tabaquismo, por medio del antagonismo de los receptores CB1.
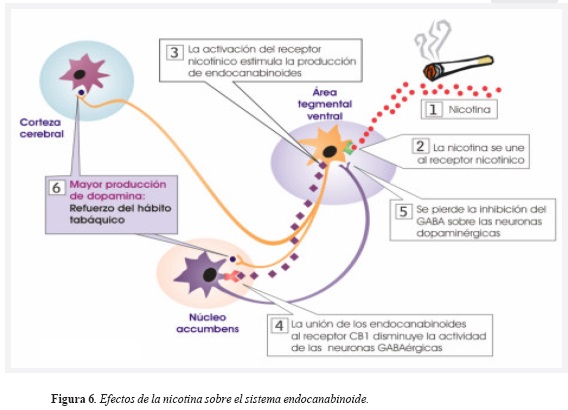
En el sistema nervioso central se hallan ampliamente distribuidos los receptores nicotínicos de acetilcolina (RN-Ach). Cuando la nicotina está disponible en grandes cantidades como consecuencia del tabaquismo, activará a los RN-Ach en las neuronas dopaminérgicas, ocasionando la liberación de este neurotransmisor en el Nacc (32) reforzando, finalmente la conducta de fumar.
La exposición crónica a la nicotina también aumenta los niveles de endocanabinoides en el SMD, de forma que al activar los receptores CB1 en la membrana de la neurona GABAérgica del Nacc impide su efecto inhibitorio sobre la liberación de dopamina en el ATV (33), reforzando aún más el tabaquismo (Figura 6). Así, teóricamente, la administración de un antagonista de los receptores CB1 puede lograr que el efecto inhibitorio del GABA sobre la neurona dopaminérgica sea restaurado permitiendo así que exista un menor reforzamiento al tabaquismo. La evidencia de esta teoría en humanos se encuentra disponible con los resultados del estudio STRATUS-US.
Comer: endocanabinoides y evidencia
El primer antagonista de los receptores CB1 disponible para el tratamiento de adicciones como el tabaquismo y de las alteraciones metabólicas relacionadas con la obesidad es rimonabant. Este compuesto es un potente ligando selectivo de los receptores CB1 y sus acciones se han demostrado desde los estudios preclínicos hasta los estudios fase III cuyos resultados se han publicado recientemente.
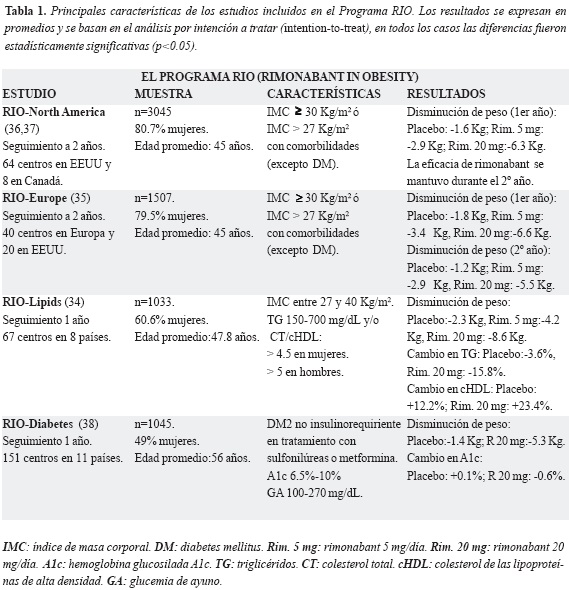
El programa RIO (Rimonabant In Obesity): consistió en cuatro ensayos clínicos controlados (estudios fase III): Rio-Europe, Rio-North America (RIO-NA), Rio-Lipids y Rio-Diabetes; aleatorizados, doble enmascaramiento, con rimonabant (5 ó 20 mg) vs. placebo. Su objetivo primario fue evaluar la eficacia y la seguridad de este antagonista de los receptores CB1 en la pérdida y mantenimiento de peso en más de 6600 personas con sobrepeso u obesidad (34,35). En los estudios RIO-NA y RIO-Europe se incluyeron respectivamente 3045 y 1507 pacientes con sobrepeso u obesidad, con o sin comorbilidades y su seguimiento fue de dos años. Los estudios RIO-Lipids y RIO-Diabetes incluyeron 1036 pacientes con dislipidemia y 1047 pacientes con diabetes mellitus tipo 2 respectivamente, todos con sobrepeso u obesidad, durante un año de seguimiento (34-35).
En el estudio RIO-Lipids, los pacientes con sobrepeso u obesidad, y dislipidemia habían perdido al final del año del seguimiento 8.6 Kg de peso corporal, en promedio, en el grupo de rimonabant 20 mg contra una disminución de 2.3 Kg en el grupo placebo ( p <0.001). Además, un 32.6 por ciento de quienes fueron tratados con rimonabant 20 mg logró una disminución del 10 por ciento o más del peso inicial mientras que sólo un 7.2 por ciento de los pacientes alcanzó esta meta en el grupo placebo ( p <0.001) (34). Los efectos adversos más comunes náuseas, mareo y diarrea fueron transitorios, leves y bien tolerados.
Los demás estudios del programa RIO tuvieron resultados similares y consistentes con el estudio RIO-Lipids, demostrando adicionalmente que la eficacia y el buen perfil de seguridad con rimonabant 20 mg se mantiene a través de dos años de seguimiento (36). En la tabla 1 se muestra un resumen práctico de los estudios incluidos en el programa RIO.
El estudio RIO-Lipids igualmente nos proporciona información respecto a los efectos de rimonabant en cuanto a sus efectos metabólicos periféricos: en las personas tratadas con rimonabant 20 mg el perímetro abdominal disminuyó 9.1 cm , los triglicéridos disminuyeron en 15.8 por ciento, el cHDL se incrementó un 23.4 por ciento y se redujeron significativamente las partículas LDL pequeñas y densas, respecto al grupo placebo (34), de donde se puede deducir que antagonizando los receptores CB1 se pueden alcanzar cambios favorables no sólo en cuanto a obesidad o tabaquismo, sino que también hay una mejora en el perfil lipídico de estos pacientes.
Conviene resaltar que en el estudio RIO-Diabetes hubo una mejoría en el perfil glucémico en aquellos tratados con rimonabant 20 mg quienes alcanzaron una disminución en la hemoglobina glucosilada (HbA1c) de 0.7 por ciento respecto al grupo placebo ( p <0.001) (38). Aún más, en el estudio RIO-Lipids la incidencia de diabetes tipo 2 en el grupo de rimonabant 20 mg se redujo 10.9 por ciento, frente al grupo placebo (39).
Proteger: endocanabinoides y evidencia
Programa STRATUS (Studies with Rimonabant And Tobacco Use): comprendió tres estudios fase III: STRATUS-US ( United States ), STRATUS-Europe y STRATUS-Worldwide. El estudio STRATUS-US fue un ensayo clínico controlado, aleatorizado, doble enmascaramiento, con Rimonabant (5 ó 20 mg) vs. placebo, seguimiento de 10 semanas y 784 personas fumadoras incluidas. Su objetivo primario fue evaluar la abstinencia prolongada durante las últimas cuatro semanas de tratamiento medida a través de un autoreporte de abstinencia y confirmado mediante niveles de cotinina (el principal metabolito de la nicotina) en plasma y concentraciones de CO en aire expirado. El porcentaje de pacientes con abstinencia prolongada en el análisis por intención de tratamiento fue de 27.6 por ciento para el grupo de rimonabant 20 mg frente a sólo 16.1 por ciento en el grupo placebo ( p =0.004; OR=2.0, IC 95%=1.3-3.0) (39,40). Aquellos pacientes con sobrepeso u obesidad que tomaron rimonabant 20 mg no aumentaron de peso, incluso lo perdieron (menos de un kilo), mientras quienes tomaron placebo y dejaron de fumar ganaron más de un kilo en promedio, evidenciando que el tratamiento del tabaquismo con rimonabant es de utilidad y evita el potencial aumento de peso asociado al abandono del tabaco, con un perfil de seguridad muy similar al encontrado en los estudios del programa RIO. La tabla 2 proporciona las principales características del estudio STRATUS-US.

Perspectivas
Actualmente está en curso el estudio STRADIVARIUS (Strategy to Reduce Atherosclerosis Development Involving Administration of Rimonabant - the intravascular Ultrasound Study) (42), que evaluará la capacidad de este antagonista de los receptores CB1 para reducir la progresión de la aterosclerosis coronaria en pacientes con sobrepeso y con otros factores de riesgo cardiovascular. Sus resultados se esperan para finales del próximo año. Un estudio que actualmente está en proceso de reclutar pacientes es CRESCENDO (Comprehensive Rimonabant Evaluation Study of Cardiovascular End points and Outcomes) (43) cuyo objetivo primario es evaluar la eficacia de rimonabant para reducir el riesgo de muerte por infarto miocárdico o ataque cerebro vascular en pacientes con obesidad abdominal y otros factores de riesgo cardiovascular.
Conclusión
Los canabinoides endógenos y sus receptores hacen parte de los sistemas corporales que ayudan a mantener la homeostasis luego de algún evento que conduce a estrés. Sin embargo, su sobreactivación mediada por el tabaquismo o la obesidad pone fin a este balance, produciendo todas las consecuencias negativas que estas condiciones generan en la salud, consecuencias que en último término aumentan el riesgo cardiovascular. La modulación de este sistema a través del antagonismo de los receptores CB1 puede aportar un evidente beneficio en el tabaquismo o la obesidad, factores modificables de riesgo cardiovascular, pero actualmente poco controlados. Otros beneficios que esta modulación puede aportar son las mejoras en el perfil lipídico y glucémico de los pacientes, modificando favorablemente no sólo uno, sino varios factores de riesgo cardiovascular.
Referencias
1. Strong K, Mathers C, Leeder S, Beaglehole R. Preventing chronic diseases: how many lives we can save?. Lancet. 2005; 366: 1578-82.
2. Psaty BM, Smith NL, Siscovick DS, Koepsell TD, Weiss NS, Heckbert SR, et al. Health outcomes associated with antihipertensive therapies used as first-line agents. A sistematic review and meta-analysis. JAMA. 1997; 277: 739-45
3. Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr, et al. The Seventh Report of the Joint National Comittee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure. JAMA. 2003; 289: 2560-72
4. Jones PH, Davidson MH, Stein EA, Bays HE, McKenney JM, Miller E, et al . Comparison of the efficacy and safety of rosuvastatin versus atorvastatin, simvastatin and pravastatin across doses (STELLAR Trial). Am J Cardiol. 2003; 92: 152-60
5. Kashyap ML, McGovern ME, Berra K, Guyton JR, Kwiterovich PO, Harper WL, et al. Long-term safety and efficacy of a once-daily niacin/lovastatin formulation for patients with dyslipidemia. Am J Cardiol. 2002; 89: 672-78
6. Pearson TA, Denke MA, Mc Bride PE, Battisti WP, Brady WE, Palmisano J. A community-based, randomized trial of ezetimibe added to statin therapy to attain NCEP ATP III goals for LDL cholesterol in hypercholesterolemic patients: the ezetimibe add-on to statin for effectiveness (EASE) trial. Mayo Clin Proc. 2005;80: 587-95.
7. Fritsche A, Schweitzer MA, Haring H. Glimepiride combined with morning insulin glargine, bedtime neutral protamine hagedorn insulin, or bedtime insulinglargine in patients with type 2 diabetes: A randomrized, controlled trial. Ann Intern Med. 2003; 138:952-59
8. Van Gaal LF, De Leewu IH. Rationale and options for combination therapy in the treatment of Type 2 diabetes . Diabetologia. 2003;46 Suppl 1:M44-50.
9. Tong PC, Chow CC, Jorgensen LN , Cockram CS. The contribution of metformin to glycaemic control in patients with type 2 diabetes mellitus receiving combination therapy with insulin. Diabetes Res Clin Pract. 2002; 57: 93-8.
10. American Heart Association. Heart Disease and Stroke Statistics-2005 Update. Dallas Texas : American Heart Association. 2005.
11. Mendivil CO, Sierra ID, Pérez CE. Valoración del riesgo cardiovascular global y prevalencia de dislipidemias según los criterios del NCEP-ATP III en una población adulta de Bogotá, Colombia. Clin Invest Arterioscl. 2004; 16: 99-107.
12. Litchman AH, Cravatt BF. Food for thougt: endocannabinoid modulation of lipogenesis. J Clin Invest. 2005; 115: 1130-33.
13. Howlett AC, Breivogel CS, Childers SR, Deadwyler SA, Hampson RE, Porrino LJ. Cannabinoid physiology and pharmacology: 30 years of progress. Neuropharmacology. 2004; 47 Suppl 1: 345-58.
14. Beal JE, Olson R, Lefkowitz L, Laubenstein L, Bellman P, Yangco B, et al. Long-term efficacy and safety of dronabinol for acquired immunodeficiency syndrome-associated anorexia. J Pain Symptom Manage. 1997; 14: 7-14.
15. Black SC. Cannabinoid receptor antagonists and obesity. Curr Opin Investig Drugs. 2004; 5:389-94.
16. Pagotto U, Vicennati V, Pasquali R. The endocannabinoid system and thge treatment of obesity. Ann Med. 2005; 37: 270-5
17. Di Marzo V, Melck D, Bisogno T, De Petrocellis L. Endocannabinoids: endogenous cannabinoid receptor ligands with neuromodulatory action. Trends Neurosci. 1998; 21: 521-8
18. Rodríguez de Fonseca F, Del Arco I, Bérmudez-Silva FJ, Bilbao A, Cippitelli A, Navarro M. The endocannabinoid system: phisiology and pharmacology. Alcohol Alcohol. 2005; 40: 2-14.
19. Cota D, Woods SC. The role of the endocannabinoid system in the regulation of energy homeostasis. Curr Opin Endocrinol Diabetes. 2005; 12: 338-351.
20. Engeli S, Bohnke J, FeldpauschM, Gorzelniak K, Janke J, Bátkai S, et al. Activationof the peripheral endocannabinoid system in human obesity. Diabetes. 2005; 54: 2838-43.
21. Jamshidi N, Taylor DA. Anandamide administration into the ventromedial hypothalamus stimulates appetite in rats. Br J Pharmacol. 2001; 134: 1151-1154.
22. Kirkham TC, Williams CM, Fezza F, Di Marzo V. Endocannabinoid levels in rat limbic forebrain and hypothalamus in relation to fasting, feeding and satiation: stimulation of eating by 2-arachidonoyl glycerol. Br J Pharmacol. 2002; 136: 550-7.
23. Mendivil CO, Sierra ID. Avances en obesidad. Rev Fac Med Univ Nac Colomb 2004; 52(4): 270-286.
24. Di Marzo V, Goparaju SK, Wang L, Liu J, Batkai S, Jarai Z, et al. Leptin-regulated endocannabinoids are involved in maintaining food intake. Nature. 2001; 410:822-825
25. Cota D, Marsciano G, Tschop M, Glubler Y, Flachskamm C, Schubert M, et al. The endogenous cannabinoid system affects energy balance via central orexigenic drive and peripheral lipogenesis . J Clin Invest . 2003;112:423-31.
26. Osei-Hyiaman D, DePetrillo M, Pacher P, Liu J, Radaeva S, Bátkai S, et al. Endocannabinoid activation at hepatic CB1 receptors stimulates fatty acid synthesis and contributes to diet-induced obesity. J Clin Invest. 2005; 115: 1298-1305.
27. Poirier B, Bidouard JP, Cadrouvele C, Marniquet X, Staels B, O´Connor SE, et al. The anti-obesity effect of rimonabant is associated with an improved serum lipid profile. Diabetes Obes Metab . 2005;7:65-72.
28. Bensaid M, Gary Bobo M, Esclangnon A, Maffrand JP, Le Fur G, Oury-Donat F, Soubrie P. The cannabinoid CB1 receptor antagonist SR141716 increases Acrp30 mRNA expression in adipose tissue of obese fa/fa rats and in cultured adipocyte cells. Mol Pharmacol. 63: 908-914.
29. Liu YL, Connoley IP, Wilson CA, Stock MJ. Effects of the cannabinoid CB1 receptor antagonist SR141716 on oxygen consumption and soleus muscle glucoseuptake in Lep(ob)/Lep(ob) mice. Int J Obes Relat Metab Disord. 2005; 29:183-187.
30. Vargas DR. Alcoholismo, tabaquismo y sustancias psicoactivas. Rev Salud Pública. 2001; 3(1)
31. Messing RO. Biología de la adicción. En: Braunwald E, Fauci AS, Kasper DL, Hauser SL, Longo DL, Jameson JL. Harrison, Principios de Medicina Interna 15a edición. Vol II. México: McGraw-Hill, Interamericana; 2002.
32. Cohen C, Kodas E, Griebel G. CB1 receptor antagonists for the treatment of nicotine addiction . Pharmacol Biochem Behav. 2005; 81: 387-95.
33. Harris DS, Anthenelli RM. Expanding treatment of tobacco dependence. Curr Psychiatry Rep . 2005;7:344-51.
34. Després JP, Golay A, Sjostrom L. Effects of rimonabant on metabolic risk factors in overweight patients with dyslipidemia. N Engl J Med. 2005; 353: 121-34.
35. Van Gaal LF, Rissanen AM, Scheen AJ, Ziegler, Rossner S. Effects of the cannabinoid-1 receptor blocker rimonabant on weight reduction and cardiovascular risk factors in overweight patients: 1-year experience from the RIO-Europe. Lancet. 2005; 365: 1389-1397.
36. Pi-Sunyer X. Late-breaking clinical trials abstracts. Circulation. 2005; 111: 1727.
37. Pi-Sunyer X, Aronne LJ, Heshmati HM, Devin J, Rosenstock J. Effects of rimonabant, a cannabinoid-1 receptor blocker, on weight and cardiometabolic risk factors in overweight or obese patients. JAMA. 2006; 295: 761-775
38. American Diabetes Association. New drug treats multiple problems of people with type 2 diabetes. Disponible en: http://www.diabetes.org/uedocuments/Rimona bant.pdf. Consultado noviembre 27 de 2005.
39. Segovia J, Bermejo J, Alfonso F. Resúmenes de los ensayos clínicos presentados en la 53 Sesión Científica Anual del American College of Cardiology (New Orleans, EE.UU., 7-10 de marzo de 2004). Rev Esp Cardiol. 2004; 57: 417 - 432.
40. Cleland JG, Ghosh J, Freemantle N, Kaye GC, Nasir M, Clark AL, et al. Clinical trials update and cumulative meta-analyses from the American College of Cardiology: WATCH, SCD-HeFT, DINAMIT, CASINO, INSPIRE, STRATUS-US, RIO-Lipids and cardiac resynchronisation therapy in heart failure. Eur J Heart Fail . 2004; 6:501-8.
41. Cluzel M. Acomplia TM a new approach to cardiovascular risk management. Disponible en: http://www.sanofi-aventis.com/images/101_24494.pdf. Consultado noviembre 27 de 2005.
42. Van Gaal LF, Rissanen A, Scheen A, Ziegler O, Rossner S. Effect of rimonabant on weight reduction and cardiovascular risk. Lancet. 2005; 366: 369-370.
43. U.S National Institutes of Health [Internet]. Disponible en http://www.clinicaltrials.gov/ct/show/NCT00263042?order=3. Consultado enero 20 de 2006.
Referencias
Strong K, Mathers C, Leeder S, Beaglehole R. Preventing chronic diseases: how many lives we can save?. Lancet. 2005; 366: 1578-82.
Psaty BM, Smith NL, Siscovick DS, Koepsell TD, Weiss NS, Heckbert SR, et al. Health outcomes associated with antihipertensive therapies used as first-line agents. A sistematic review and meta-analysis. JAMA. 1997; 277: 739-45
Chobanian AV, Bakris GL, Black HR, Cushman WC, Green LA, Izzo JL Jr, et al. The Seventh Report of the Joint National Comittee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure. JAMA. 2003; 289: 2560-72
Jones PH, Davidson MH, Stein EA, Bays HE, McKenney JM, Miller E, et al . Comparison of the efficacy and safety of rosuvastatin versus atorvastatin, simvastatin and pravastatin across doses (STELLAR Trial). Am J Cardiol. 2003; 92: 152-60
Kashyap ML, McGovern ME, Berra K, Guyton JR, Kwiterovich PO, Harper WL, et al. Long-term safety and efficacy of a once-daily niacin/lovastatin formulation for patients with dyslipidemia. Am J Cardiol. 2002; 89: 672-78
Pearson TA, Denke MA, Mc Bride PE, Battisti WP, Brady WE, Palmisano J. A community-based, randomized trial of ezetimibe added to statin therapy to attain NCEP ATP III goals for LDL cholesterol in hypercholesterolemic patients: the ezetimibe add-on to statin for effectiveness (EASE) trial. Mayo Clin Proc. 2005;80: 587-95.
Fritsche A, Schweitzer MA, Haring H. Glimepiride combined with morning insulin glargine, bedtime neutral protamine hagedorn insulin, or bedtime insulinglargine in patients with type 2 diabetes: A randomrized, controlled trial. Ann Intern Med. 2003; 138:952-59
Van Gaal LF, De Leewu IH. Rationale and options for combination therapy in the treatment of Type 2 diabetes . Diabetologia. 2003;46 Suppl 1:M44-50.
Tong PC, Chow CC, Jorgensen LN , Cockram CS. The contribution of metformin to glycaemic control in patients with type 2 diabetes mellitus receiving combination therapy with insulin. Diabetes Res Clin Pract. 2002; 57: 93-8.
American Heart Association. Heart Disease and Stroke Statistics-2005 Update. Dallas Texas : American Heart Association. 2005.
Mendivil CO, Sierra ID, Pérez CE. Valoración del riesgo cardiovascular global y prevalencia de dislipidemias según los criterios del NCEP-ATP III en una población adulta de Bogotá, Colombia. Clin Invest Arterioscl. 2004; 16: 99-107.
Litchman AH, Cravatt BF. Food for thougt: endocannabinoid modulation of lipogenesis. J Clin Invest. 2005; 115: 1130-33.
Howlett AC, Breivogel CS, Childers SR, Deadwyler SA, Hampson RE, Porrino LJ. Cannabinoid physiology and pharmacology: 30 years of progress. Neuropharmacology. 2004; 47 Suppl 1: 345-58.
Beal JE, Olson R, Lefkowitz L, Laubenstein L, Bellman P, Yangco B, et al. Long-term efficacy and safety of dronabinol for acquired immunodeficiency syndrome-associated anorexia. J Pain Symptom Manage. 1997; 14: 7-14.
Black SC. Cannabinoid receptor antagonists and obesity. Curr Opin Investig Drugs. 2004; 5:389-94.
Pagotto U, Vicennati V, Pasquali R. The endocannabinoid system and thge treatment of obesity. Ann Med. 2005; 37: 270-5
Di Marzo V, Melck D, Bisogno T, De Petrocellis L. Endocannabinoids: endogenous cannabinoid receptor ligands with neuromodulatory action. Trends Neurosci. 1998; 21: 521-8
Rodríguez de Fonseca F, Del Arco I, Bérmudez-Silva FJ, Bilbao A, Cippitelli A, Navarro M. The endocannabinoid system: phisiology and pharmacology. Alcohol Alcohol. 2005; 40: 2-14.
Cota D, Woods SC. The role of the endocannabinoid system in the regulation of energy homeostasis. Curr Opin Endocrinol Diabetes. 2005; 12: 338-351.
Engeli S, Bohnke J, FeldpauschM, Gorzelniak K, Janke J, Bátkai S, et al. Activationof the peripheral endocannabinoid system in human obesity. Diabetes. 2005; 54: 2838-43.
Jamshidi N, Taylor DA. Anandamide administration into the ventromedial hypothalamus stimulates appetite in rats. Br J Pharmacol. 2001; 134: 1151-1154.
Kirkham TC, Williams CM, Fezza F, Di Marzo V. Endocannabinoid levels in rat limbic forebrain and hypothalamus in relation to fasting, feeding and satiation: stimulation of eating by 2-arachidonoyl glycerol. Br J Pharmacol. 2002; 136: 550-7.
Mendivil CO, Sierra ID. Avances en obesidad. Rev Fac Med Univ Nac Colomb 2004; 52(4): 270-286.
Di Marzo V, Goparaju SK, Wang L, Liu J, Batkai S, Jarai Z, et al. Leptin-regulated endocannabinoids are involved in maintaining food intake. Nature. 2001; 410:822-825
Cota D, Marsciano G, Tschop M, Glubler Y, Flachskamm C, Schubert M, et al. The endogenous cannabinoid system affects energy balance via central orexigenic drive and peripheral lipogenesis . J Clin Invest . 2003;112:423-31.
Osei-Hyiaman D, DePetrillo M, Pacher P, Liu J, Radaeva S, Bátkai S, et al. Endocannabinoid activation at hepatic CB1 receptors stimulates fatty acid synthesis and contributes to diet-induced obesity. J Clin Invest. 2005; 115: 1298-1305.
Poirier B, Bidouard JP, Cadrouvele C, Marniquet X, Staels B, O´Connor SE, et al. The anti-obesity effect of rimonabant is associated with an improved serum lipid profile. Diabetes Obes Metab . 2005;7:65-72.
Bensaid M, Gary Bobo M, Esclangnon A, Maffrand JP, Le Fur G, Oury-Donat F, Soubrie P. The cannabinoid CB1 receptor antagonist SR141716 increases Acrp30 mRNA expression in adipose tissue of obese fa/fa rats and in cultured adipocyte cells. Mol Pharmacol. 63: 908-914.
Liu YL, Connoley IP, Wilson CA, Stock MJ. Effects of the cannabinoid CB1 receptor antagonist SR141716 on oxygen consumption and soleus muscle glucoseuptake in Lep(ob)/Lep(ob) mice. Int J Obes Relat Metab Disord. 2005; 29:183-187.
Vargas DR. Alcoholismo, tabaquismo y sustancias psicoactivas. Rev Salud Pública. 2001; 3(1)
Messing RO. Biología de la adicción. En: Braunwald E, Fauci AS, Kasper DL, Hauser SL, Longo DL, Jameson JL. Harrison, Principios de Medicina Interna 15a edición. Vol II. México: McGraw-Hill, Interamericana; 2002.
Cohen C, Kodas E, Griebel G. CB1 receptor antagonists for the treatment of nicotine addiction . Pharmacol Biochem Behav. 2005; 81: 387-95.
Harris DS, Anthenelli RM. Expanding treatment of tobacco dependence. Curr Psychiatry Rep . 2005;7:344-51.
Després JP, Golay A, Sjostrom L. Effects of rimonabant on metabolic risk factors in overweight patients with dyslipidemia. N Engl J Med. 2005; 353: 121-34.
Van Gaal LF, Rissanen AM, Scheen AJ, Ziegler, Rossner S. Effects of the cannabinoid-1 receptor blocker rimonabant on weight reduction and cardiovascular risk factors in overweight patients: 1-year experience from the RIO-Europe. Lancet. 2005; 365: 1389-1397.
Pi-Sunyer X. Late-breaking clinical trials abstracts. Circulation. 2005; 111: 1727.
Pi-Sunyer X, Aronne LJ, Heshmati HM, Devin J, Rosenstock J. Effects of rimonabant, a cannabinoid-1 receptor blocker, on weight and cardiometabolic risk factors in overweight or obese patients. JAMA. 2006; 295: 761-775
American Diabetes Association. New drug treats multiple problems of people with type 2 diabetes. Disponible en: http://www.diabetes.org/uedocuments/Rimona bant.pdf. Consultado noviembre 27 de 2005.
Segovia J, Bermejo J, Alfonso F. Resúmenes de los ensayos clínicos presentados en la 53 Sesión Científica Anual del American College of Cardiology (New Orleans, EE.UU., 7-10 de marzo de 2004). Rev Esp Cardiol. 2004; 57: 417 - 432.
Cleland JG, Ghosh J, Freemantle N, Kaye GC, Nasir M, Clark AL, et al. Clinical trials update and cumulative meta-analyses from the American College of Cardiology: WATCH, SCD-HeFT, DINAMIT, CASINO, INSPIRE, STRATUS-US, RIO-Lipids and cardiac resynchronisation therapy in heart failure. Eur J Heart Fail . 2004; 6:501-8.
Cluzel M. Acomplia TM a new approach to cardiovascular risk management. Disponible en: http://www.sanofi-aventis.com/images/101_24494.pdf. Consultado noviembre 27 de 2005.
Van Gaal LF, Rissanen A, Scheen A, Ziegler O, Rossner S. Effect of rimonabant on weight reduction and cardiovascular risk. Lancet. 2005; 366: 369-370.
U.S National Institutes of Health [Internet]. Disponible en http://www.clinicaltrials.gov/ct/show/NCT00263042?order=3. Consultado enero 20 de 2006.
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2006 Revista de la Facultad de Medicina

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
-





















