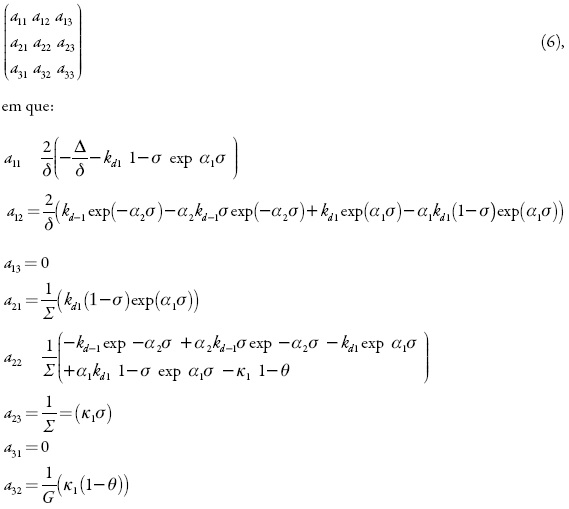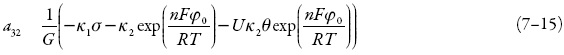O mecanismo do desempenho de poli-2-mercaptobenzimidazol na detecção eletroquímica de colesterol e as suas representações eletroquímica e matemática
The mechanism of electroanalytical function of 2-mercaptobenzimidazole in cholesterol electrochemical detection and its mathematical and chemical evaluations
DOI:
https://doi.org/10.15446/rcciquifa.v45n2.59938Palabras clave:
Colesterol, polímeros condutores, sensores eletroquímicos, poli-2mercaptobenzimidazol, estado estacionário estável (pt)Cholesterol, conducting polymers, electrochemical sensors, poly-2-mercaptobenzimidazole, stable steady-state (en)
The electroanalytical function of poly-2-mercaptobenzimidazole in cholesterol electrochemical detection was evaluated phenomenologically and mathematically. The corresponding mathematical model was developed and analyzed by linear stability theory and bifurcation analysis. The theoretical data were compared with the experimental data and with theoretical data for analogous systems.
https://doi.org/10.15446/rcciquifa.v45n2.59938
O mecanismo do desempenho de poli-2-mercaptobenzimidazol na detecção eletroquímica de colesterol e as suas representações eletroquímica e matemática
The mechanism of electroanalytical function of 2-mercaptobenzimidazole in cholesterol electrochemical detection and its mathematical and chemical evaluations
Volodymyr V. Tkach1,2*, Sílvio C. de Oliveira2, Mohammad Reza M. Hosseini3, Reza Ojani4, Olga V. Yelenich1, Petró I. Yagodynets´1
1 Universidade Nacional de Chernivtsi, 58012, Rua de Kotsyubyns´ky, 2, Ucrânia
2 Universidade Federal de Mato Grosso do Sul, Av. Sen. Felinto. Müller, 1555, C/P. 549, 79074460, Campo Grande, MS, Brasil
3 Universidade Iraniana de Ciência e Tecnologia, 16846 – 13114, Teerã, República Islâmica do Irã
4 Universidade de Mazandarã, 47416-95447, 3o km. Rodovia de Forças Aéreas Iranianas, Babolsar, República Islamica do Irã
* Correio eletrônico: nightwatcher2401@gmail.com
Recebido em: 12 de dezembro de 2015 Aceito em: 02 de maio de 2016
Resumo
O desempenho de poli-2-mercaptobenzimidazol na detecção eletroquímica de colesterol foi avaliado fenomenológica e matematicamente. O modelo matemático correspondente foi desenvolvido e analisado por meio de teoria de estabilidade lineal e análise de bifurcações. Os dados teóricos foram comparados com os experimentais e com os teóricos para os sistemas análogos.
Palavras-chave: Colesterol, polímeros condutores, sensores eletroquímicos, poli-2mercaptobenzimidazol, estado estacionário estável.
Summary
The electroanalytical function of poly-2-mercaptobenzimidazole in cholesterol electrochemical detection was evaluated phenomenologically and mathematically. The corresponding mathematical model was developed and analyzed by linear stability theory and bifurcation analysis. The theoretical data were compared with the experimental data and with theoretical data for analogous systems.
Keywords: Cholesterol, conducting polymers, electrochemical sensors, poly-2-mercaptobenzimidazole, stable steady-state.
Introdução
Colesterol [1] é um álcool policíclico, derivado de ciclopentanperidrofenantreno, considerado um esteróide do grupo de esteróis. É precursor de hormônios sexuais [2] de animais vertebrados (inclusive o homem), bem como de hormônio de mudança de cor dos insetos. Toma parte ativa do metabolismo.
No entanto, haja vista a sua insolubilidade em meio aquoso, o seu excesso pode provocar o bloqueio de corrente sanguínea [3], bem como problemas renais e de fígado. Destarte, a procura de um método eficiente da sua detecção é uma tarefa atual [4–8].
A utilização de elétrodos quimicamente modificados é um dos métodos modernos, utilizados na química eletroanalítica contemporânea [9–12]. Neste caso, um modificador usado (como polímero condutor, complexo, corante, ou outra) é escolhido haja vista a sua compatibilidade com o analito (segundo o princípio de chave e fechadura). No entanto, há dificuldades no seu uso com o colesterol, haja vista os problemas da previsão de mecanismo mais provável do desempenho de um sensor hipotético, bem como o existirem instabilidades eletroquímicas durante a eletrooxidação de compostos orgânicos (inclusive a eletrossíntese de polímeros condutores) [13–21], o que pode prejudicar a interpretação do sinal analítico.
Os dois problemas, bem como o da comparação com os sistemas análogos, empregando outros analitos e outros materiais [22–25] sem ensaios experimentais, resolvem-se pelo desenvolvimento de um modelo matemático, capaz de descrever adequadamente o desempenho do sistema eletroanalítico, o que se fará neste trabalho para colesterol como analito e poli(2-mercaptobenzimidazol) como substância modificadora [12].
O sistema e o seu modelo
No sensor, descrito em [12], foi descrita a eletropolimerização de 2-mercaptobenzimidazol na presença de colesterol, que se acompanhava pelo entrapment das suas moléculas. Não foi, de fato, um processo químico – as moléculas do hormônio ficaram presas entre as moléculas do polímero condutor sem poder sair da sua "matriz". Este processo será controlado pela difusão de mercaptobenzimidazol e do hormônio e dar-se-á, em termos gerais, em conformidade com os modelos matemáticos, expostos em [26–28], levemente modificados (por conta do acréscimo de um adendo que descreve a saída do analito da matriz polimérica).
No caso da ausência de analito, o comportamento durante a eletropolimerização obedecerá a um dos modelos, descritos em [26–28] (dependendo das condições da síntese), sendo o 2-mercaptobenzimidazol o monômero.
Para descrever a eletrodetecção de colesterol, por meio da sua imobilização por dopagem da matriz polimérica, introduzir-se-ão as três variáveis:
c – a concentração de colesterol na camada pré-superficial;
σ – o grau de dopagem da matriz polimérica pelo colesterol;
θ – o grau de revestimento da parte do polímero, quimicamente modificada pelo analito.
É possível provar que, haja vista as suposições, feitas, por exemplo, em [26–28], o conjunto de equações diferenciais que descreve este sistema se expõe como:
Em que Δ é coeficiente de difusão, δ é espessura da camada pré-superficial, d1e d-1, a velocidade de dopagem-desdopagem da matriz pelo analito, Σ é o número máximo de sítios de dopagem, fornecidos pela matriz do polímero condutor revestido, G é a concentração máxima do polímero condutor, r1e r2 são as reações de modificações química e eletroquímica do polímero condutor.
As velocidades das respectivas reações podem calcular-se como:
em que os parâmetros k são constantes de respectivas reações, α1 e α1 descrevem as interações entre o dopante e a matriz, n é o número de elétrons transferidos, F é o número de Faraday, φ0 é o salto do potencial na dupla camada elétrica (DCE), relacionado ao da carga zero, R é a constante universal de gases e T, a temperatura absoluta do vaso.
A principal diferença deste sistema dos relacionados [22–25] é a presença de uma etapa preliminar de dopagem-dedopagem do analito pelo polímero condutor, anterior à sua reação específica. Isto dá impacto significante ao comportamento do sistema.
Resultados e discussão
Para analisar o comportamento do sistema da eletrodetecção de colesterol por meio da sua imobilização na matriz de 2-mercaptobenzimidazol, analisamos o conjunto de equações diferenciais (1), haja vista as relações algébricas (2–5) por meio da teoria de estabilidade linear. A matriz funcional de Jacobi, cujos elementos são calculados para o estado estacionário, vê-se como:
A instabilidade oscilatória neste caso é possível. Outrossim, é mais provável que nos sistemas típicos do desempenho eletroanalítico de polímeros condutores e materiais afins [22–25]. O porquê disso expor-se-á abaixo.
Para a bifurcação de Hopf se realizar, a diagonal principal da matriz de Jacobi deve ter elementos positivos, responsáveis pela positiva conexão de retorno.
Vê-se que, além do adendo−Uκ2θ exp 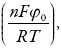 cuja positividade descreve as influências na DCE, causa comum do comportamento oscilatório nestes sistemas, outro elemento capazmente positivo é
cuja positividade descreve as influências na DCE, causa comum do comportamento oscilatório nestes sistemas, outro elemento capazmente positivo é  cuja positividade descreve a atração entre o analito-dopante, que é colesterol, e a matriz polimérica.
cuja positividade descreve a atração entre o analito-dopante, que é colesterol, e a matriz polimérica.
Confirma-se, destarte, a presença de estruturas dissipativas temporais, cuja existência se mantém pela difusão de colesterol (entrada de energia) e pela formação do polímero condutor na sua forma final (saída de energia).
Para analisar a estabilidade de estado estacionário, utilizamos o critério de Routh-Hurwitz, do qual se obtém o requisito geral Det J < 0. Para evitar a análise de expressões grandes, introduzimos as novas variáveis para o determinante da matriz aparecer como:
Abrindo os parênteses, obter-se-á a condição de estabilidade do estado estacionário como:
Obteve-se uma expressão, típica para sistemas, em que a reação é regulada pela difusão, o que acontece, quando a dopagem é mais rápida, em grandes concentrações do analito. No entanto, sendo as concentrações do analito pequenas, a difusão passa a ser mais rápida que a dopagem e a reação reger-se-á pela dopagem.
Vê-se que a negatividade do parâmetro da dopagem-dedopagem X, descrevendo a fraqueza ou ausência de atração entre a matriz e o dopante e a positividade de Υ promovem a estabilidade do estado estacionário, porque deslocam a expressão à esquerda de (17) para valores mais negativos.
No caso da igualdade das influências desestabilizadoras às estabilizadoras, obtém-se a instabilidade monotônica, que aparece em:
Reação lateral efetiva entre o analito e o polímero, paralela à dopagem, acrescentará mais um adendo às equações (1) e (3). Tal reação dá seu impacto na estabilidade do estado estacionário, diminuindo a influência de dopagem.
Autocatálise para este polímero e analito não é possível, mas se houver com outro caso análogo, aparecerá como mais uma causa do comportamento oscilatório [22–25].
Conclusões
A presença da etapa preliminar de dopagem do analito pela matriz do polímero condutor faz o comportamento do sistema mais complicado, haja vista a presença, nele, dos nuances da dopagem.
rápida, em grandes concentrações do analito. No entanto, sendo as con centrações do analito pequenas, a difusão passa a ser mais rápida que a dopagem e a reação reger-se-á pela dopagem.
atração entre o analito-dopante e a matriz polimérica que o "hospeda".
Confirma-se a presença de estruturas dissipativas temporais, cuja existência se mantém pela difusão de colesterol (entrada de energia) e pela formação do polímero condutor na sua forma final (saída de energia).
Conflito de interesses
Os autores declaram que não há conflito de interesses.
Referências
1. Informações da Sociedade Brasileira de Endocrinologia e Metabologia, URL: http://www.endocrino.org.br/colesterol/, acesso aos 8 de setembro de 2015.
2. K. Zhang, R. Kaufmann, Unfolding the toxicity of cholesterol, Nat. Cell. Biol., 5, 769 (2003).
3. Informe da Real Sociedade Britânica de Medicina (RMS), URL: http://www.nhs.uk/conditions/Cholesterol/Pages/Introduction.aspx, acesso aos 8 de setembro de 2015.
4. A. Mondal, N.R. Jana, Fluorescent detection of cholesterol using β-cyclodextrin functionalized graphene, Chem. Comm., 48, 7316 (2012).
5. T.T.N. Dinh, J.R. Blanton Jr., J.C. Brooks et al., A simplified method for cholesterol determination in meat and meat products, J. Food Comp. Anal., 21, 306 (2008).
6. P. Sriwasadi, M.H. Kroll, P.H. Lolekha, Advantages and disadvantages of serum cholesterol determination by the kinetic versus the endpoint method, Am. J. Clin. Path., 127, 906 (2007).
7. L.C. Bauer, D. de A. Santana, M. dos S. Macedo et al., Method validation for simultaneous determination of cholesterol and cholesterol oxides in milk by RPHPLC-DAD, J. Braz. Chem. Soc., 25, 161 (2014).
8. C.M. Bandeira, J.M. Ferreira, G.C. Nogueira et al., Application of cholesterol determination method to indirectly detect meat and bone meals in ruminant feeds, Quím. Nova, 36, 1222 (2013).
9. R. Ojani, V. Rahimi, J. Raoof, A new voltammetric sensor for hydrazine based on michael addition reaction using 1-amino-2-naphtol-4-sulfonic acid, J. Chin. Chem. Soc., 62, 90 (2015).
10. S.E. Baghbamidi, H. Beitollahi, S. Tajik, Graphene oxide nano-sheets/ferrocene derivative modified carbon paste electrode as an electrochemical sensor for determination of hydrazine, Anal. Bioanal. Electrochem., 6, 634 (2014).
11. J.B. Raoof, R.Ojani, F. Jamali, S.R. Hosseini, Electrochemical detection of hydrazine using a copper oxide nanoparticle modified glassy carbon electrode, Casp. J. Chem., 1, 73 (2012).
12. M.R. Milani Hosseini, Sh. Hemmati, A. Ghaffarinejad, A. Motaharian, Fabrication of molecularly imprinted polymer coated carbon nanotubes modified gold electrode for determination of cholesterol, Anal. Bioanal. Electrochem., 7, 129 (2015).
13. L. Hudson, M.R. Bassett, Oscillatory electrodissolution of metals, Rev. Chem. Eng., 7, 108 (1991).
14. I. Das, N.R. Agrawal, S.A. Ansari, S.K. Gupta, Pattern formation and oscillatory polymerization of thiophene, Ind. J. Chem., 47, 1798 (2008).
15. S.U. Rahman, M.S. Ba-Shammakh, Thermal effects on the process of electropolymerization of pyrrole on mild steel, Synth. Met., 140, 207 (2004).
16. A.S. Liu, M.A.S. Oliveira, Electrodeposition of polypyrrole films on aluminium from tartrate aqueous solution, J. Braz. Chem Soc., 18, 143 (2007).
17. D. Sazou, The dynamic behavior of the electrochemical polymerization of indole in acetonitrile - water mixtures, Synth. Met., 130, 45 (2002).
18. I. Das, N. Goel, N.R. Agrawal, S.K. Gupta, Growth patterns of dendrimers and electric potential oscillations during electropolymerization of pyrrole using mono- and mixed surfactants, J. Phys. Chem., 114, 12888 (2010).
19. M. Bazzaoui, E.A. Bazzaoui, L. Martins, J.I. Martins, Electropolymerization of pyrrole on zinc-lead-silver alloys' electrodes in neutral and acid organic media, Synth. Met., 130, 73 (2002).
20. I. Das, N. Goel, S.K. Gupta, N.R. Agrawal, Electropolymerization of pyrrole: Dendrimers, nano-sized patterns and oscillations in potential in presence of aromatic and aliphatic surfactants, J. Electroanal. Chem., 670, 1 (2012).
21. K. Aoki, I. Mukoyama, J. Chen, Polymerization and dissolution of polythiophene films, Russ. J. Electrochem., 40, 319 (2004).
22. V. Tkach, V. Nechyporuk, P. Yagodynets, El estudio de estabilidad en el proceso del desempeño de los sensores y biosensores, basados en los polímeros conductores en medio fuertemente ácido, Rev. Colomb. Cienc. Quím. Farm., 42, 30 (2013).
23. V. Tkach, V. Nechyporuk, P. Yagodynets', The mathematical stability study of the work of isomerization sensors, based on conduction polymers, Anal. Bioanal. Electrochem., 6, 273 (2014).
24. V. Tkach, B. Kumara Swamy, R. Ojani et al., O comportamento de paracetamol durante a sua oxidação eletrocatalítica sobre poli(azul da anilina) e a sua descrição matemática, Rev. Colomb. Cienc. Quím. Farm., 44, 148 (2015).
25. V. Tkach, B. Kumara Swamy, R. Ojani et al., El mecanismo de la oxidación de omeprazol sobre el electrodo de carbono vitroso, modificado por polializarina, y su descripción matemática, Orbital Elec. J. Chem., 7, 1 (2015).
26. V. Tkach, R. Ojani, V. Nechyporuk et al., Cathodic and anodic potentiostatic polypyrrole electrodeposition in strongly acid media. Theoretical and experimental comparison, Bulg. Chem. Comm., 48, 126 (2016).
27. V. Tkach, V. Nechyporuk, P. Yagodynets´, The mathematical description for the electropolymerization of furan, pyrrole and thiophene derivatives in alkaline media, Med. J. Chem., 3, 1122 (2015).
28. V. Tkach, S.C. de Oliveira, O.I. Aksimentyeva et al., The mathematical modeling for the electropolymerization of acid derivatives of heterocyclic compounds over active metals, Mor. J. Chem., 3, 550 (2015).
Como citar este artigo
V.V. Tkach, S.C. de Oliveira, M.R. Hosseini, R. Ojani, O. Yelenich, P.I. Yagodynets´, O mecanismo do desempenho de poli-2-mercaptobenzimidazol na detecção eletroquímica de colesterol e as suas representações eletroquímica e matemática, Rev. Colomb. Cienc. Quím. Farm., 45(2), 234-242 (2016).
Referencias
(1) Informações da Sociedade Brasileira de Endocrinologia e Metabologia, URL:
http://www.endocrino.org.br/colesterol/ , acesso aos 8 de setembro de 2015.
(2) K. Zhang, R. Kaufmann, Unfolding the toxicity of cholesterol, 5, Nat. Cell. Biol.,
, 769 (2003).
(3) Informe da Real Sociedade Britânica de Medicina (RMS), URL:
href="http://www.nhs.uk/conditions/Cholesterol/Pages/Introduction.aspx”target=_"blank">
http://www.nhs.uk/conditions/Cholesterol/Pages/Introduction.aspx , acesso aos 8 de setembro
de 2015.
(4) A. Mondal, N.R. Jana, Fluorescent detection of cholesterol using β-cyclodextrin
functionalized graphene, Chem. Comm., 48, 7316 (2012).
(5) T.T.N. Dinh, J.R. Blanton Jr., J.C. Brooks et al., A simplified method for cholesterol determination
In meat and meat products, J. Food Comp. Anal., 21, 306 (2008).
(6) P. Sriwasadi, M.H. Kroll, P.H. Lolekha, Advantages and disadvantages of serum cholesterol determination by the kinetic versus the endpoint method, Am. J. Clin. Path., 127, 906 (2007).
(7) L.C. Bauer, D. de A. Santana, M. dos S. Macedo et al., Method validation for simultaneous determination of cholesterol and cholesterol oxides in milk by RPHPLC-DAD, J. Braz. Chem. Soc., 25, 161 (2014).
(8) C.M. Bandeira, J.M. Ferreira, G.C. Nogueira et al., Application of cholesterol determination method to indirectly detect meat and bone meals in ruminant feeds, Quím. Nova, 36, 1222 (2013).
(9) R. Ojani, V. Rahimi, J. Raoof, A new voltammetric sensor for hydrazine based on michael addition reaction using 1-amino-2-naphtol-4-sulfonic acid, J. Chin. Chem. Soc., 62, 90 (2015).
(10) S.E. Baghbamidi, H. Beitollahi, S. Tajik, Graphene oxide nano-sheets/ferrocene derivative modified carbon paste electrode as an electrochemical sensor for determination of hydrazine, Anal. Bioanal. Electrochem., 6, 634 (2014).
(11) J.B. Raoof, R.Ojani, F. Jamali, S.R. Hosseini, Electrochemical detection of hydrazine using
A copper oxide nanoparticle modified glassy carbon electrode, Casp. J. Chem., 1, 73 (2012).
(12) M.R. Milani Hosseini, Sh. Hemmati, A. Ghaffarinejad, A. Motaharian, Fabrication of molecularly imprinted polymer coated carbon nanotubes modified gold electrode for determination of cholesterol, Anal. Bioanal. Electrochem., 7, 129 (2015).
(13) L. Hudson, M.R. Bassett, Oscillatory electrodissolution of metals, Rev. Chem. Eng., 7, 108 (1991).
(14) I. Das, N.R. Agrawal, S.A. Ansari, S.K. Gupta, Pattern formation and oscillatory polymerization of thiophene, Ind. J. Chem., 47, 1798 (2008).
(15) S.U. Rahman, M.S. Ba-Shammakh, Thermal effects on the process of electropolymerization of pyrrole on mild steel, Synth. Met., 140, 207 (2004).
(16) A.S. Liu, M.A.S. Oliveira, Electrodeposition of polypyrrole films on aluminium from tartrate aqueous solution, J. Braz. Chem Soc., 18, 143 (2007).
(17) D. Sazou, The dynamic behavior of the electrochemical polymerization of indole in acetonitrile water mixtures, Synth. Met., 130, 45 (2002).
(18) I. Das, N. Goel, N.R. Agrawal, S.K. Gupta, Growth patterns of dendrimers and electric potential oscillations during electropolymerization of pyrrole using mono- and mixed surfactants, J. Phys. Chem., 114, 12888 (2010).
(19) M. Bazzaoui, E.A. Bazzaoui, L. Martins, J.I. Martins, Electropolymerization of pyrrole on zinc-lead-silver alloys’ electrodes in neutral and acid organic media, Synth. Met., 130, 73 (2002).
(20) I. Das, N. Goel, S.K. Gupta, N.R. Agrawal, Electropolymerization of pyrrole: Dendrimers, nano-sized patterns and oscillations in potential in presence of aromatic and aliphatic surfactants, J. Electroanal. Chem., 670, 1 (2012).
(21) K. Aoki, I. Mukoyama, J. Chen, Polymerization and dissolution of polythiophene films, Russ. J. Electrochem., 40, 319 (2004).
(22) V. Tkach, V. Nechyporuk, P. Yagodynets, El estudio de estabilidad en el proceso del desempeño de los sensores y biosensores, basados en los polímeros conductores en medio fuertemente ácido, Rev. Colomb. Cienc. Quím. Farm., 42, 30 (2013).
(23) V. Tkach, V. Nechyporuk, P. Yagodynets´, The mathematical stability study of the work of isomerization sensors, based on conduction polymers, Anal. Bioanal. Electrochem., 6, 273 (2014).
(24) V. Tkach, B. Kumara Swamy, R. Ojani et al., O comportamento de paracetamol durante a sua oxidação eletrocatalítica sobre poli(azul da anilina) e a sua descrição matemática, Rev. Colomb. Cienc. Quím. Farm., 44, 148 (2015).
(25) V. Tkach, B. Kumara Swamy, R. Ojani et al., El mecanismo de la oxidación de omeprazol sobre el electrodo de carbono vitroso, modificado por polializarina, y su descripción matemática, Orbital Elec. J. Chem., 7, 1 (2015).
(26) V. Tkach, R. Ojani, V. Nechyporuk et al., Cathodic and anodic potentiostatic polypyrrole electrodeposition in strongly acid media. Theoretical and experimental comparison, Bulg. Chem. Comm., 48, 126 (2016).
(27) V. Tkach, V. Nechyporuk, P. Yagodynets´, The mathematical description for the electropolymerization of furan, pyrrole and thiophene derivatives in alkaline media, Med. J. Chem., 3, 1122 (2015).
(28) V. Tkach, S.C. de Oliveira, O.I. Aksimentyeva et al., The mathematical modeling for the electropolymerization of acid derivatives of heterocyclic compounds over active metals, Mor. J. Chem., 3, 550 (2015).
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
Licencia
Derechos de autor 2016 Revista Colombiana de Ciencias Químico-Farmacéuticas

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
El Departamento de Farmacia de la Facultad de Ciencias de la Universidad Nacional de Colombia autoriza la fotocopia de artículos y textos para fines de uso académico o interno de las instituciones citando la fuente. Las ideas emitidas por los autores son responsabilidad expresa de estos y no de la revista.
Todo el contenido de esta revista, excepto dónde está identificado, está bajo una Licencia Creative Commons de Atribución 4.0 aprobada en Colombia. Consulte la normativa en: http://co.creativecommons.org/?page_id=13