CHARACTERIZATION OF THE OXIDATIVE ENZYME POTENTIAL IN WILD WHITE ROT FUNGI FROM THE SUBTROPICAL FOREST OF MISIONES (ARGENTINA)
Caracterización del potencial enzimático oxidativo de cepas nativas de hongos de pudrición blanca de la selva subtropical de Misiones (Argentina)
DOI:
https://doi.org/10.15446/abc.v20n1.38322Palabras clave:
oxidative enzymes, screening, white-rot fungi (en)enzimas oxidativas, hongos de pudrición blanca, screening (es)
Descargas
Artículo de investigación
CHARACTERIZATION OF THE OXIDATIVE ENZYME POTENTIAL IN WILD WHITE ROT FUNGI FROM THE SUBTROPICAL FOREST OF MISIONES (ARGENTINA)
Caracterización del potencial enzimático oxidativo de cepas nativas de hongos de pudrición blanca de la selva subtropical de Misiones (Argentina)
María Isabel FONSECA1, Pedro Darío ZAPATA1, Laura Lidia VILLALBA1, Julia Inés FARIÑA2.
1 Laboratorio de Biotecnología Molecular, Instituto de Biotecnología Misiones, Facultad de Ciencias Exactas Químicas y Naturales, UNaM, Ruta 12 Km 7,5 – Campus Universitario UNaM – 3304. Posadas, Misiones, Argentina.
2 Departamento de Biotecnología Fúngica, PROIMI-CONICET; Cátedra de Química Biológica, Facultad de Cs. Exactas y Naturales, UNCA. San Miguel de Tucumán, Argentina.
For correspondence. biotecmol2010@gmail.com
Received 3rd June 2013, Returned for revision 24th April 2014, accepted 7th May 2014.
Citation/Citar este artículo como: Fonseca MI, Zapata PD, Villalba LL, Fariña JI. Characterization of the oxidative enzyme potential in wild white rot fungi from the subtropical forest of Misiones (Argentina). Acta biol. Colomb. 2015;20(1):47-56. doi: https://doi.org/10.15446/abc.v20n1.38322.
ABSTRACT
This research aimed to evaluate the potential of several native white rot fungi (WRF) isolated from subtropical environments of Misiones (Argentina) to produce different ligninolytic enzymes. Coriolus versicolor f. antarcticus BAFC 266, Pycnoporus sanguineus BAFC 2126 and Phlebia brevispora BAFC 633 showed the highest phenoloxidase activity. Ganoderma applanatum strain E, P. sanguineus BAFC 2126 and P. brevispora BAFC 633 revealed marked laccase and peroxidase activity. C. versicolor f. antarcticus, G. applanatum (strain A) and Trametes villosa, gave high positive reactions with 2,6-dimethoxyphenol oxidation at the lowest tested pH. C. versicolor f. antarcticus, G. applanatum strains D and F, T. elegans BAFC 2127and T. villosa, showed the highest manganese peroxidase activity. C. versicolor f. antarcticus also produced the highest lignin peroxidase activity. Tyrosinase activity was mostly evident in G. applanatum strains (D and F) and Phanerochaete chrysosporium HHB 11741. Kraft liquor decolorization results were variable and depended on the fungus and the liquor concentration. Some fungi with moderate ligninolytic activity showed high decolorization rates (e.g. Pleurotus sajor-caju and Steccherinium sp. BAFC 1171) indicating the significance of additional approach to evaluate a potential biotechnological application.
Keywords: oxidative enzymes, screening, white-rot fungi.
RESUMEN
El objetivo de este trabajo fue evaluar el potencial para producir enzimas ligninolíticas de diversas cepas de hongos de pudrición blanca, nativas de la Provincia de Misiones (Argentina). Coriolus versicolor v. antarcticus BAFC 266, Pycnoporus sanguineus BAFC 2126 y Phlebia brevispora BAFC 633 mostraron un gran potencial para producir fenoloxidasas. En Ganoderma applanatum cepa E, P. sanguineus BAFC 2126 y P. brevispora BAFC 633 se observó una marcada actividad lacasa y peroxidasa. C. versicolor v. antarcticus. G. applanatum (cepa A) y Trametes villosa, mostraron una reacción oxidativa altamente positiva sobre 2,6-dimetoxifenol a pH 4. C. versicolor v. antarcticus, G. applanatum (cepas D y F), T. elegans BAFC 2127 y T. villosa, mostraron una alta actividad manganeso peroxidasa y solo C. versicolor v. antarcticus mostró una alta actividad de lignino peroxidasa. La actividad tirosinasa fue evidente en las cepas D y F de G. applanatum y en Phanerochaete chrysosporium HHB 11741. La decoloración del licor negro Kraft reveló una actividad ligninolítica variable para las diferentes cepas estudiadas dependiendo de la concentración utilizada. Algunos hongos mostraron una moderada actividad ligninolítica con grandes halos de decoloración (p.e. Pleurotus sajor-caju y Steccherinium sp. BAFC 1171) indicando un significativo potencial para el aprovechamiento biotecnológico de estas cepas.
Palabras clave: enzimas oxidativas, hongos de pudrición blanca, screening.
INTRODUCTION
The major component of biomass is lignocellulose and it represents the most abundant renewable organic resource in soil. A large amount of cellulose, hemicellulose and lignin by-products from agriculture and forestry are usually wasted (Sánchez, 2009). Lignocellulosic plant material can be efficiently decomposed by wood-decaying filamentous fungi (Basidiomycota and Ascomycota), which are indispensable for the Earth's carbon cycle, generation of soil humic matter and formation of soil fine structure. These fungi can be frequently found in Nature forming brackets, caps or resupinaceous (corticioid) fruiting bodies on wood, thus facilitating their sexual basidiospores dissemination (Lundell et al., 2010).
The ability to break down lignin, the most recalcitrant component of cell walls, to CO2 has become an essential role in the carbon cycle played by a small group of basidiomycetes commonly called white-rot fungi (WRF). This potential, mainly based on their strong oxidative activity and low substrate specificity of their ligninolytic enzymes, has been also proposed for the degradation of a variety of persistent environmental pollutants (Tekere et al., 2001a; Levin et al., 2004). As a secondary benefit, promising WRF may be used not only to alleviate environmental deterioration derived from lignocellulosic residue generation, but also to promote its bioconversion to different added-value products (Millati et al., 2011).
The enzyme production of phenol-type oxidases differentiates WRF behavior from other wood-rotting basidiomycetes and different wood decomposers (Cañas and Camarero, 2010).
Extracellular enzymatic systems of WRF display differential features depending on the species, strains and culture conditions. Nevertheless, they usually involve enzymes such as lignin peroxidase (LiP, EC 1.11.1.14), able to directly oxidize non-phenolic units, whilst manganese peroxidase (MnP, EC 1.11.1.13) and laccase (Lac, EC 1.10.3.2) preferentially oxidize phenolic compounds, although non-phenolic units may eventually be degraded in the presence of mediators (Saparrat et al., 2002; Lundell et al., 2010).
Biotechnological applications developed in the postgenomic era are associated with the use of isoenzymes (or the genes involved) with different physicochemical properties such as pI, optimal pH, response to inducers and repressors, etc. These particular features make them interesting to be used in "tailor made processes". On the other hand, there is some evidence that same species from different habitats may show different genomic and proteomic features that may be exploited for relevant biotechnological applications (Tekere et al., 2001b; Fonseca et al., 2010).
Exploration and description of new microorganisms from rich and vast biodiversity of poorly explored natural environments can reveal innovative capacities for potential biotechnological applications such as bioremediation and biomass conversion, areas where WRF play an essential role (Saparrat et al., 2002; Levin et al., 2004; Sánchez, 2009; Giorgio et al., 2012). Moreover, the relevance of screening new isolates in addition to culture collection strains has been already highlighted (Tekere et al., 2001a).
The aim of this study was to disclose qualitative differences in the production of oxidative enzymes in subtropical WRF native from Misiones (Northeast, Argentina) and to categorize most promising strains for future biotechnological applications on biomass conversion.
MATERIAL AND METHODS
Microorganisms
This work includes strains from the United States, such as Phanerochaete and Ceriporiopsis of recognized efficiency on biomass conversion such as biopulping (Blanchette and Burnes 1988; Akhtar et al., 1993; Ferraz et al., 2003; Villalba et al., 2006), in order to compare them with the native strains in the Misiones province.
The fungal strains used in the present work were previously isolated from the subtropical rainforest of Misiones (Argentina) and properly deposited in public culture collections. Ganoderma applanatum (strains A, B, C, D, E, F) and Pleurotus sajor-caju were deposited at the Culture Collection of the Faculty of Forestry, Universidad Nacional de Misiones, Argentina. Coriolus versicolor f. antarcticus BAFC 266, Phlebia brevispora BAFC 633, Pycnoporus sanguineus BAFC 2126, Trametes elegans BAFC 2127, Trametes villosa BAFC 2755 and Steccherinium sp. BAFC 1171 were deposited at the Mycological Culture Collection of the Department of Biological Sciences, Faculty of Exact and Natural Sciences, Universidad de Buenos Aires, Argentina.
On the other hand, fungi used for comparison purposes such as Ceriporiosis subvermispora FP 90031, Phanerochaete chrysosporium ME 446, P. chrysosporium BKMF 1767, Ceriporiosis subvermispora L 6332, P. chrysosporium HHB 11741 and C. subvermispora FP 105752 were provided by the Center for Forest Mycology Research, Forest Products Laboratory, USDA, USA.
Culture conditions
Stock cultures of fungal strains were maintained by periodic sub-culturing on malt extract agar plates containing 12.7 g/l malt extract and 20 g/l agar (MEA) incubated for seven days at 28°C. Sub-cultures were carried out every 30 days followed by storage at 4°C. Inocula consisted in 0.5-mm2agar plugs covered with mycelium from five-seven-day-old colonies previously grown at 28°C on MEA plates. Mycelium plugs were aseptically cut from actively growing zones and then, transferred to the corresponding plates for screening.
Tests were performed in triplicate and repeated when the target reaction was not induced.
Enzyme detection on solid media
Enzymes are assumed as extracellular form if they diffuse widely into the agar which was not yet overgrown (Gramss et al., 1998).
Phenoloxidase activity (PhOX) is noticed by the agar color change of samples grown on solid MEA supplemented with 0.5% w/v tannic acid (Bavendamm 1928, Serrano Silva et al. 2010). After seven days of incubation at 28°C, a dark brown color around the mycelium is considered a positive result (Rayner and Boddy, 1988).
Peroxidase (POX) activity was also revealed by pouring equal parts of 0.4% v/v H2O2 and 1 % w/v pyrogallol on MEA growth plates. A yellowish-brown color in the solid media indicates a positive result. POX is rated positive when staining with H2O2 is clearly browner than those without H2O2. The high rate of applied H2O2 ensured the partial inhibition of most laccase activities, as previously recommended (Gramss et al., 1998).
To reveal laccase activity (Lac), fungi were grown at 28°C on plates with MEA medium supplemented with the textile dye Vilmafix® Blue RR-BB dye (0.2 g/l) (Pajot et al., 2007). After complete decolorization (five days), the plate was covered with 1.2 mM ABTS [2,2'-azino-bis(3ethylbenzothiazoline-6-sulphonic acid)] in acetate buffer pH 4.5 and incubated in the dark for 20 min. The appearance of green color in the solid media indicates a positive result. A solution containing 0.7 mM H2O2 was subsequently added to the previous plate in order to reveal peroxidase activity (POX). The intensification of green color is considered a positive reaction (Murugesan et al., 2007).
Another useful technique for Lac detection was used as follows. Fungi were grown for five days at 28#ºC on MEA plates. Lac activity was evidenced by adding 12.4 g/l guaiacol in 96% v/v ethanol. The appearance of an orange coloration after 4 h is considered a positive result (López et al., 2006).
A third test carried out to detect Lac activity was performed by inoculating fungal strains on MEA plates at pH 4, 5 or 6, containing 0.8 g/l 2,6-dimethoxyphenol (DMP), as an adaptation of previous protocols for Lac evaluation in liquid media (Park et al., 2008; Flores et al., 2009). Dark orange oxidation zones were daily measured during 5 days.
To detect manganese peroxidase activity (MnP) fungi were grown on MEA plates for five days at 28°C. Plates were developed by the addition of 0.03 mM N,N,N',N'tetramethyl-p-phenylenediamine (TMPD), 1 mM H2O2, 0.7 mM MnSO4·7H2O, in citrate-phosphate buffer pH 5.0. Enzyme activity is evidenced by the occurrence of a purple color on the agar plates (Toh et al., 2003).
For lignin peroxidase activity (LiP), WRF were grown on MEA for five days at 28°C and plates were then revealed with 32 mM Azure II in tartrate buffer pH 4.5 plus 0.1 mM H2O2 (Archibald, 1992; Zhao et al., 1996). The discoloration after ten min is considered a positive result.
Tyrosinase (Tyr) activity was assessed according to a modified method by Rayner and Boddy (1988). After fungal growth on MEA for five days at 28°C, Tyr was revealed with 1 % w/v pyrocatechol in phosphate buffer pH 7.4. A brownish pink color on the agar plates after ten min is considered positive.
Black liquor decolorization assays on solid medium
In order to test the fungal ability for decolorization usually associated with ligninolytic activity, fungi were inoculated onto plates containing a culture medium supplemented with two different concentrations of Kraft liquor effluent.
Lignin degradation was assayed on plates containing 10 g/l glucose malt agar supplemented with either 3.3 or 6.6 % v/v black liquor. Black liquor, a lignin-rich effluent of the Kraft cooking process, was obtained at laboratory scale and consisted mainly of remaining substances after dissolving wood components in the cooking chemicals. Degradation was followed by measuring changes from 3rd to 14th culture day (Fonseca et al., 2010).
RESULTS
Enzyme detection on solid media
Many microorganisms producing ligninolytic enzymes have been selected using solid media containing substrates or dye indicators that allow direct visualization of enzyme production (Nishida et al., 1988, De Jong et al., 1992; Barbosa et al., 1996).
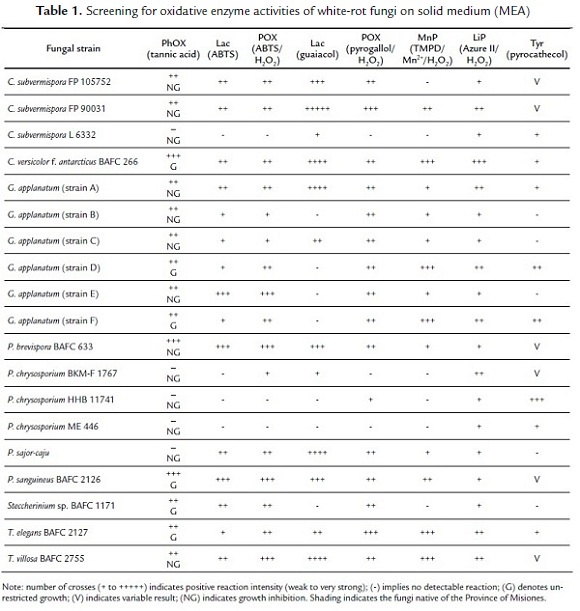
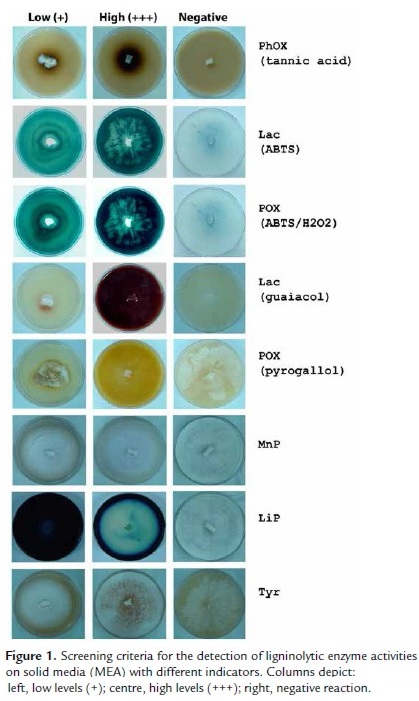
Data corresponding to ligninolytic activities detection on solid media for all tested fungal strains are presented in Table 1 and are representative of three independent experiments. Screening criteria for ligninolytic enzyme activities on solid media (MEA) with different indicators are shown in Fig. 1.
Malt agar medium supplemented with tannic acid is frequently used to detect the presence of extracellular oxidases which are generally characteristic of ligninolytic activity (Bavendamm, 1928; Morozova et al. 2007; Herter et al. 2012). Many of the tested fungi showed activity when grown in tannic acid-amended medium (Table 1). C. versicolor f. antarcticus BAFC 266, P. sanguineus BAFC 2126 and P. brevispora BAFC 633 (with no evident growth) showed the maximal oxidation of tannic acid. On the other hand, G. applanatum (strains D and F), Steccherinium sp. BAFC 1171 and T. elegans BAFC 2127 were able to grow in this medium with moderate PhOX activity. Conversely, C. subvermispora FP 90031 and C. subvermispora FP 105752, G. applanatum strains A, B, C, E, and T. villosa BAFC 2755, showed PhOX release under these conditions but were unable to grow, a fact probably related to the high tannic acid concentration applied (Gramss et al., 1998).
In the present work, textile dye Vilmafix® Blue RR-BB was totally decolorized after five days of cultivation with C. versicolor f. antarcticus BAFC 266, P. brevispora BAFC 633 and C. subvermispora FP 105752. Fungi G. applanatum strain E, P. sanguineus BAFC 2126 and P. brevispora BAFC 633 revealed high Lac activity when confronted to ABTS and the blue-green intensity of haloes was increased after addition of H2O2 due the presence of POX (Table 1). Other fungi like C. subvermispora FP 105752, C. subvermispora FP 90031, C. versicolor f. antarcticus BAFC 266, G. applanatum cepa A, P. sajor-caju and Steccherinium sp. BAFC 1171 showed moderate Lac and POX activity. T. villosa BAFC 2755 showed moderate Lac activity while showed high POX activity (Table 1). Additionally T. elegans 2127 and G. applanatum (strain D and F) showed low activity Lac and revealed POX activity more intense (Table 1). C. subvermispora L6332, P. chrysosporium ME 446, P. chrysosporium HHB 11741 did not show ABTS oxidation (Table 1.). In the results analysis, it was important to consider the degree of discoloration achieved during growth, prior to the addition of ABTS ± H2O2, since it could be interpreted as false positives; therefore discoloration plates were taken as positive controls to determine the Lac and POX real activities during development.
The detection of POX with pyrogallol plus H2O2 appeared a quite sensitive technique and showed activity in most of the tested fungi with the exception of P. chrysosporium ME 466, C. subvermispora L 6332 and P. chrysosporium BKMF 1767, latter two confirmed previous results with ABTS/ H2O2 (Table 1). The wide distribution of POX reaction found virtually in all tested fungi, particularly when assayed by pyrogallol/ H2O2 (Table 1), has been also documented for different ecological groups of fungi (Gramss et al., 1998). The extracellular nature of detected enzymes was also herein observed. The role of POXs could be essential for wood degradation, as already emphasized for the degradation of litter (Gramss et al., 1998).
When the guaiacol modified technique was applied, most of the above results for Lac activity could be confirmed, being C. subvermispora FP 90031 the highest positive (Table 1). Meanwhile, P. brevispora BAFC633, G. applanatum (strains A and C), P. sanguineus BAFC 2126, T. villosa BAFC 2755, T. elegans BAFC 2127, C. versicolor f. antarcticus BAFC 266, P. sajor-caju and C. subvermispora FP 105752 showed qualitatively lower guaiacol oxidation. Negatives as those of G. applanatum (strain B, D, E and F), P. chrysosporium HHB 11741, P. chrysosporium ME 466 and Steccherinium sp. BAFC 1171 might be related to the inability of some laccases to oxidize certain substrates depending on their redox potential (Pajot et al., 2007). The presence of Lac as one of the main ligninolytic activities was not unexpected, in agreement with previous screening reports on native argentinian fungi (Saparrat et al., 2002).
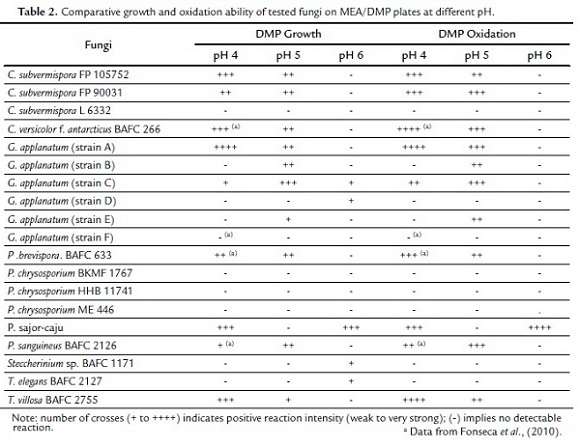
Many authors have used DMP for laccase quantification (Park y Park, 2008; Flores et al., 2009). In this paper, the method was applied to solid medium. In agreement with ABTS and guaiacol data (Table 1), C. versicolor f. antarcticus BAFC 266, P. brevispora BAFC 633, C. subvermispora FP 105752, T. villosa BAFC 2755, and C. subvermispora FP 90031 gave highly positive results for DMP oxidation at 4 and 5 pH values (Table 2). On the other hand, marked Lac activities for G. applanatum strain A, P. sajor-caju and P. sanguineus BAFC 2126 were also coincident with results from ABTS and guaiacol tests (Table 1), but in the case of Steccherinium sp. BAFC 1171, negative results from DMP (Table 2) showed more correlation with guaiacol oxidation (Table 1).
Concerning MnP, G. applanatum (strains D and F), T. elegans BAFC 2127, T. villosa BAFC 2755 and C. versicolor f. antarcticus BAFC 266 showed the highest MnP activity. Likewise, moderate activities were found in C. subvermispora FP 90031 and P. sanguineus BAFC 2126 (Table 1).
The highest LiP activity was found in C. versicolor f. antarcticus BAFC 266, followed by G. applanatum (strains A, D and F), T. elegans BAFC 2127, T. villosa BAFC 2755 C. subvermispora FP 90031 and P. chrysosporium BKM-F 1767, as indicated by the marked degradation of Azure II (Table 1). These findings resulted particularly relevant considering a previous screening on the ligninolytic potential of other argentinian fungi where LiP activity could not be detected (Saparrat et al., 2002). However, they showed correlation with positive results from Zhao et al. (1996) and Levin et al. (2004). As these authors already found, the herein applied LiP technique was highly successful to this purpose.
Considering LiP positive results on P. chrysosporium strains (Table 1), it would interesting to note that differences on the detection methodology, as well as the influence of culture medium and/or conditions should not be neglected at the time of evaluating LiP production (Zhao et al., 1996; Tekere et al., 2001a; Saparrat et al., 2002; Levin et al., 2004).
With regard to Tyr activity, it was distinctly detected in P. chrysosporium HHB 11741, and to a lesser extent, in G. applanatum (strains D and F) (Table 1).
Black liquor decolorization assays on solid medium
Approximately half of the fungi tested showed discoloration at both concentrations tested. In many cases, reasonably, the discoloration was lower at the highest concentration of black liquor tested. P. sajor-caju showed the highest decolorization rates at both concentrations used, followed by Steccherinium sp. BAFC 1171, T. elegans BAFC 2127, G. applanatum strain A and P. sanguineus BAFC 2126. Other fungi showed very good discoloration, but mostly at the lowest concentration of black liquor, such as G. applanatum strain D and C. versicolor f. antarcticus BAFC 266. Meanwhile, P. brevispora BAFC 633 was able to completely decolorize the black liquor only at a concentration of 3.3 % (v/v), and 6 of the 17 fungi tested showed no discoloration at any of the concentrations tested, especially for strains of P. chrysosporium and C. subvermispora.
The demonstrated Kraft liquor degradation capability achieved by some tested fungi along 14 days of cultivation thus suggested their potential use for bioremediation of pulping industry effluents (Fonseca et al., 2010). The reduced peak at 280 nm of the spectral scanning performed in broth of P. brevispora BAFC 633 supplemented with black liquor indicated fungal ligninolytic activity (Shimizu et al., 2009). The intensity of the absorbance is related to the lignin concentration, being significantly lower for the fungus treated samples.
The apparent lack of correlation between ligninolytic enzyme profile and Kraft liquor discoloration was not completely unusual. Previous reports on dye decolorization by WRF also described similar results frequently attributed to other degradative mechanisms rather than ligninolytic system (Tekere et al., 2001a; Levin et al., 2004).
The herein described screening tests allowed to reveal a hidden and up-to-date unexplored potential of wild fungi isolated from subtropical environments, which may behave as competent ligninolytic enzyme-producers, such as C. versicolor f. antarcticus BAFC 266, G. applanatum strains A, D and F, P. sanguineus BAFC 2126, P. brevispora BAFC 633, T. elegans BAFC 2127 and T. villosa BAFC 2755. The ligninolytic enzyme machinery of these fungi might be considered for the residual lignin oxidation during biomass conversion, representing a promising and low cost bioremediation and bioconversion alternative (Fonseca et al., 2010; Giorgio et al., 2012).
DISCUSSION
As already described for different ecological groups such as litter decomposing, wood degrading and ectomycorrhizal fungi (Sinsabaugh et al., 2002), PhOX activity seemed to be common in the herein tested fungi.
Unsurprisingly, only some fungi are able to grow in tannic acid-containing medium because tannins are usually toxic to microorganisms, mainly due to enzymatic inhibition, substrate deficiency, action on membranes and lack of metal ions (Ngono Ngane et al., 2006). Nevertheless, some fungi, bacteria and yeasts are quite resistant and capable of growing in the presence of tannins making its use justifiable to the screening purpose under stringent conditions (Bhat et al., 2007). Eventually, as previously proposed, the use of lower (1/10) tannic acid concentrations was proved to still induce browning in the presence of oxidative activities without inhibiting fungal growth (Gramss et al., 1998).
Dye decolorization potential of different fungi has been extensively studied and associated to lignin degrading ability (Kersten y Cullen, 2007). It has been already documented that laccase enzyme of white rot fungi such as T. versicolor 951022 showed affinity and specific activity for ABTS (Moon-Jeong et al., 2005).
The differences observed with the three substrates used for Lac screening could be attributed at different positions of chemicals groups and different affinities of Lacs for each compound (Min et al., 2001).
Preceding results showed the relevance of investigating these ligninolytic activities according to different protocols and involving varied substrates in order to achieve more conclusive results. Additionally, data from DMP oxidation also gave evidence of the importance of pH at the time of screening these activities, being the highest values generally obtained at pH 4-5 (Table 2). These findings were in agreement with those from Tekere et al. (2001a), who also noted a preference for acidic pHs (< 4.5) for dye degradation by ligninolytic fungi.
It should be however noted that, as previously emphasized, if detected ligninolytic activities were constitutive, titers may be subjected to the influence of inducers such as aromatic compounds or Cu2+ (Saparrat et al., 2002; Dhouib et al., 2005). These results were in agreement with others previously reported by Tekere et al. (2001a), Saparrat et al. (2002), Matos et al. (2007), Lin et al. (2003) and Basto et al. (2006) who also described high ligninolytic activities in closely related fungal specimens such as T. villosa, C. versicolor, G. applanatum and P. sanguineus, consequently proposed for different biotechnological applications.
Different authors have earlier reported high MnP activity in other Trametes and Ganoderma species, e.g. in T. versicolor when grown with high carbon and nitrogen sources (Tekere et al., 2001b), and in G. lucidum IBL-05 as the only secreted enzyme (Bibi et al., 2009). The simultaneous presence of MnP and Lac has been already described as a typical feature in basidiomycetes (Tekere et al., 2001a; Saparrat et al., 2002).
The simultaneous secretion of LiP and MnP in the studied fungi may represent a valuable potential as this enzyme association has been previously related to efficient degraders (Levin et al., 2004).
Compared to other work on Tyr secretion, different chromogenic phenolic solutions were used for its detection, and these results were somehow divergent to the wide distribution of Tyr previously found in different ecological groups by Gramss et al. (1998). The extracellular nature of the Tyr activities described in the present work was also different with respect to previous reports on intracellular Tyr (Gramss et al., 1998), and this property was herein additionally confirmed since no ethanol was applied to dissolve the indicator substance.
Considering the oxidative strains commonly used in USA in biopulping experiments, we observed that in general strains of local origin showed higher oxidative potential that Phanerochaete strains and similar to Ceriporiopsis strains. These finding described ligninolytic activities in isolates from the subtropical rainforest of Misiones are not unexpected. Previous reports on similar screening campaigns already drawn the attention to the highly adaptable physiology and heterogeneous enzyme systems of WRF isolated from harsh subtropical environments where a wide host range and tolerance are usually observed (Tekere et al., 2001a).
The secreted oxidative enzymes may alleviate the environmental impact of industrial processes and chemical manufacturing during biomass conversion, among other applications (Sánchez, 2009). Special interest is based on their undeniable advantages and promising application in specific bioprocesses such as pre-treatment of lignocellulosic materials for bioethanol production, biosensors, bioremediation (Levin et al., 2004) and several processing steps related to the paper industry such as pulping (Villalba et al. 2006), bleaching, deinking, depitching and effluent treatment. Accordingly, next steps will pursue the knowledge of the molecular basis for the production of these biotechnology relevant enzymes, considering their prospective commercial insertion.
At the same time, this work allowed to disclose certain differences in the enzymatic profiles of fungi belonging to the same genus and/or species. This fact highlighted once again, the relevance of evaluating the enzymatic potential of fungal isolates for each particular case, as a desired activity may not be always associated to a given microbial identity. This study also denoted that different methodologies for enzyme assessment would be not excluding but complementary, and may successfully help at the time of selecting fungal strains from nature in order to be applied with specific biotechnological purposes.
CONCLUSIONS
The screening tests showed that our subtropical environments yield effective ligninolytic enzymes producers such as C. versicolor f. antarcticus BAFC 266, G. applanatum strain F, Peniophora sp. BAFC 633, P. sanguineus BAFC 2126 and T. villosa BAFC 2755, that can be used on residual lignin oxidation during biomass conversion.
ACKNOWLEDGEMENTS
M.I. Fonseca is a recipient of a PhD fellowship from CONICET, Argentina. Part of the experimental work was funded by the Secretaría de Ciencia y Tecnología de la Universidad Nacional de Misiones, through grant for innovation project 16Q352 (Disp. SGCyT N#º 001/08).
REFERENCES
Akhtar M, Attridge MC, Myers GC, Blanchette RA. Biomechanical Pulping of Loblolly Pine Chips with Selected White-Rot Fungi. Holzforschung. 1993;47:36–40. Doi: 10.1515/hfsg.1993.47.1.36.
Archibald FS. A new assay for lignin-type peroxidases employing the dye Azure B. Appl Environ Microbiol. 1992;58:3110-3116.
Barbosa AM, Dekker RFH, Hardy GE. Veratryl alcohol as an inducer of laccase by an ascomycete, Botryosphaeria sp., when screened on the polymeric dye Poly R-478. Lett Appl Microbiol. 1996;23:93-96. Doi: 10.1111/j.1472-765X.1996.tb00038.x.
Basto C, Silva CJ, Gübitz G, Cavaco-Paulo A. Stability and decolourization ability of Trametes villosa laccase in liquid ultrasonic fields. Ultrason Sonochem. 2007;14:355-362. Doi: 10.1016/j.ultsonch.2006.07.005.
Bavendamm W. Über das Vorkommen und den Nachweis von Oxydasen bei holzzerstörenden Pilzen. Z. Pfanzenkrankh. Pfanzenschutz. 1928;38:257–276.
Bhat TK, Makkar HPS, Singh B. Preliminary studies on tannin degradation by Aspergillus niger van Tieghem MTCC 2425. Lett Appl Microbiol. 2007;25(1):22-23. Doi: 10.1046/j.1472-765X.1997.00164.x.
Bibi I, Bhatti HN, AsgheR M. Decolourisation of direct dyes with manganese peroxidase from white rot basidiomycete Ganoderma lucidum-IBL-5. Can J Chem Engin. 2009;87(3):435-440. Doi: 10.1002/cjce.20165.
Blanchette RA, Burnes TA. Selection of White-rot Fungi for Biopulping. Biomass. 1988;15(2):93-101. Doi: 10.1016/0144-4565(88)90099-6.
Cañas AI, Camarero S. Laccases and their natural mediators: biotechnological tools for sustainable eco-friendly processes. Biotechnol Adv. 2010;28(6):694-705. Doi: 10.1016/j.biotechadv.2010.05.002.
De Jong EFP, De Vries JA, Field RP, Vander Z, De Bont JAM. Isolation and screening of basidiomycetes with high peroxidative activity. Mycol Res. 1992;96(12):1098-1104. Doi: 10.1016/S0953-7562(09)80121-4.
Dhouib A, Hamza M, Zouari H, Mechichi T, Hmidi R, Labat M. Screening for ligninolytic enzyme production by diverse fungi from Tunisia. World J. Microbiol Biotechnol. 2005;21(8-9):1415-1423. Doi: 10.1007/s11274-005-5774-z.
Ferraz A, Córdova AM, Machuca A. Wood biodegradation and enzyme production by Ceriporiopsis subermispora during solid-state fermentation of Eucalyptus grandis. Enzyme Microb Technol. 2003;32(1):50-65. Doi: 10.1016/S0141-0229(02)00267-3.
Flores C, Vidal C, Trejo-Hernández MR, Galindo E, Serrano-Carreón L. Selection of Trichoderma strains capable of increasing laccase production by Pleurotus ostreatus and Agaricus bisporus in dual cultures. J Appl Microbiol. 2009;106:249-257. Doi: 10.1111/j.1365-2672.2008.03998.x.
Fonseca MI, Shimizu E, Zapata PD, Villalba LL. Copper inducing effect on laccase production of white rot fungi native from Misiones (Argentina). Enzyme Microb Technol. 2010;46:534–539. Doi: 10.1016/j.enzmictec.2009.12.017.
Giorgio EM, Fonseca MI, Tejerina MR, Ramos-Rryb AB, Sanabria N, Zapata PD, Villalba LL. Chips and sawdust substrates application for lignocellulolytic enzymes production by solid state fermentation. Int Res J Microbiol. 2012;3(7):120-127.
Gramss G, Günther T, Fritsche W. Spot tests for oxidative enzymes in ectomycorrhizal, wood-, and litter decaying fungi. Mycol Res. 1998;102(1):67-72. Doi: 10.1017/S095375629700436X.
Herter S, Schmidt M, Thompson ML, Mikolasch A, Schauer F. Investigating the effects of metals on phenol oxidaseproducing nitrogen-fixing Azotobacter chroococcum. J Basic Microbiol. 2013; 53(6):509-517. Doi: 10.1002/jobm.201100443.
Kersten P, Cullen D. Extracellular oxidative systems of the lignin-degrading Basidiomycete Phanerochaete chrysosporium. Fungal Genet Biol. 2007;44(2):77-87. Doi: 10.1016/j.fgb.2006.07.007.
Levin L, Papinutti L, Forchiassin F. Evaluation of Argentinean white rot fungi for their ability to produce ligninmodifying enzymes and decolorize industrial dyes. Biores Technol. 2004;94(2):169-176. Doi: 10.1016/j.biortech.2003.12.002.
Lin JP, Wei L, Xia LM, Cen PL. Production of laccase by Coriolus versicolor and its application in decolorization of dyestuffs: (I). Production of laccase by batch and repeated-batch processes. J Environ Sci. 2003;15(1):1-4.
López MJ, Guisado G, Vargas-García MC, Suárez-Estrella F, Moreno J. Decolorization of industrial dyes by ligninolytic microorganisms isolated from composting environment. Enzyme Microb Technol. 2006;40(1):42-45. Doi: 10.1016/j.enzmictec.2005.10.035.
Lundell TK, Mäkelä MR, Hildén K. Lignin-modifying enzymes in filamentous basidiomycetes-ecological, functional and phylogenetic review. J Basic Microbiol. 2010; 50(1):5-20. Doi: 10.1002/jobm.200900338.
Matos AJ, Bezerra RM, Dias AA. Screening of fungal isolates and properties of Ganoderma applanatum intended for olive mill wastewater decolourization and dephenolization. Lett Appl Microbiol. 2007;45(3):270-275. Doi: 10.1111/j.1472-765X.2007.02181.x.
Millati R, Syamsiah S, Niklasson C, Nur Cahyanto M, Lundquist K, Taherzadeh M. Biological pretreatment of lignocelluloses with white-rot fungi and its applications: a review. BioRes. 2011;6(4):5224-5259.
Min KL, Kim YH, Kim YW, Jung HS, Hah YC. Characterization of a novel laccase produced by the woo-drotting fungus Phellinus ribis. Arch Biochem Biophys. 2001;392(2):279–286. Doi: 10.1006/abbi.2001.2459
Moon-Jeong H, Hyoung-Tae C, Hong-Gyu S. Purification and characterization of laccase from the white rot fungus Trametes versicolor. J Microbiol. 2005;43(6):555-560.
Morozova OV, Shumakovich GP, Gorbacheva MA, Shleev SV, Yaropolov AI. Blue laccases. Biochem (Moscow). 2007;72(10):1136-1150. Doi: 10.1134/S0006297907100112.
Murugesan K, Nam IH, Kim YM, Chang YS. Decolorization of reactive dyes by a thermostable laccase produced by Ganoderma lucidum in solid culture. Enzyme Microb Technol. 2007;40(7):1662-1672. Doi: 10.1016/j.enzmictec.2006.08.028.
Ngono Ngane A, Ebelle Etame R, Ndifor F, Biyiti L, Amvam Zollo PH, Bouchet P. Antifungal activity of Chromolaena odorata (L.) King & Robinson (Asteraceae) of Cameroon. Chemother. 2006;52(2):103-106. Doi:10.1159/000092373.
Nishida TK, Kashino Y, Mimura A, Takahara Y. Lignin biodegradation by wood rotting fungi I. Screening of lignin degradating fungi. Mokuzai Gakkaishi. 1988;34:530-536.
Pajot HF, Figueroa LIC, Fariña JI. Dye-decolorizing activity in isolated yeasts from the ecoregion of Las Yungas (Tucumán, Argentina). Enzyme Microb Technol. 2007;40(6):1503-1511. Doi: 10.1016/j.enzmictec.2006.10.038.
Park KM, Park SS. Purification and characterization of laccase from basidiomycete Fomitella fraxinea. J Microbiol Biotechnol. 2008;18(6):670-675.
Rayner ADM, Boddy L. Fungal decomposition of wood: its biology and ecology. Editorial John Wiley & Sons Ltd., Chichester, Sussex, UK.; 1988. 587p.
Sánchez C. Lignocellulosic residues: biodegradation and bioconversion by fungi. Biotechnol Adv. 2009;27(2):185-194. Doi: 10.1016/j.biotechadv.2008.11.001.
Saparrat MCN, Martínez MJ, Cabello MN, Arambarri AM. Screening for ligninolytic enzymes in autochthonous fungal strains from Argentina isolated from different substrata. Rev Iberoam Micol. 2002;19:181-185.
Serrano Silva I, Ragagnin De Menezes C, Franciscon E, Da Costa Dos Santos E, Durrant LR. Degradation of lignosulfonic and tannic acids by ligninolytic soil fungi cultivated under icroaerobic conditions. Braz. Arch. Biol. Technol. 2010;53(3):693-699. Doi: 10.1590/S1516-89132010000300026.
Shimizu E, Velez-Rueda JO, Zapata PD, Villalba LL. Relación entre degradación de colorantes y oxidación de lignina residual causados por Ganoderma applanatum y Pycnoporus sanguineus en el licor negro kraft. Rev Cienc Tecnol. 2009; 12:46–51.
Sinsabaugh RL, Carreiro MM, Repert DA. Allocation of extracellular enzymatic activity in relation to litter composition, N deposition, and mass loss. Biogeochem. 2002;60:1-24. Doi: 10.1023/A:1016541114786.
Tekere M, Mswaka AY, Zvauya R, Read JS. Growth, dye degradation and ligninolytic activity studies on Zimbabwean white rot fungi. Enzyme Microb Technol. 2001a;28(4-5):420-426. Doi: 10.1016/S0141-0229(00)00343-4.
Tekere M, Zvauya R, Read JS. Ligninolytic enzyme production in selected sub-tropical white rot fungi under different culture conditions. J Basic Microbiol. 2001b;41(2):115-129.
Toh Y, Jia J, Yen L, Obbard JP, Ting Y. Decolorization of azo dyes by white-rot fungi (WRF) isolated in Singapore. Enzyme Microb Technol. 2003;33:569-575. Doi: 10.1016/S0141-0229(03)00177-7.
Villalba LL, Scott GM, Schoeder LR. Modification of loblolly pine chips with Ceriporiopsis subvermispora. Part 1: Effect of fungal treatment. J Wood Chem Technol. 2006;26(4):339-348. Doi: 10.1080/02773810601105177.
Zhao J, De Koker TH, Janse BJH. Comparative studies of lignin peroxidases and manganese-dependent peroxidases produced by selected white-rot fungi in solid media. FEMS Microb Lett. 1996;145:393-399. Doi: 10.1016/S0378-1097(96)00438-7.
Referencias
Akhtar M, Attridge MC, Myers GC, Blanchette RA. Biomechanical Pulping of Loblolly Pine Chips with Selected White-Rot Fungi. Holzforschung. 1993;47:36–40.
Archibald FS. A new assay for lignin-type peroxidases employing the dye Azure B. Appl Environ Microbiol. 1992;58:3110-3116.Doi: 0099-2240/92/093110-07$02.00/0
Barbosa AM, Dekker RFH, Hardy GE. Veratryl alcohol as an inducer of laccase by an ascomycete, Botryosphaeria sp., when screened on the polymeric dye Poly R-478. Lett Appl Microbiol. 1996;23:93-96. Doi: 10.1111/j.1472-765X.1996.tb00038.x
Basto C, Silva CJ, Gübitz G, Cavaco-Paulo A. Stability and decolourization ability of Trametes villosa laccase in liquid ultrasonic fields. Ultrason Sonochem. 2007;14:355-362. Doi:0.1016/j.ultsonch.2006.07.005
Bavendamm W. Über das Vorkommen und den Nachweis von Oxydasen bei holzzerstörenden Pilzen. Z. Pfanzenkrankh. Pfanzenschutz. 1928;38:257–276.
Bhat TK, Makkar HPS, Singh B. Preliminary studies on tannin degradation by Aspergillus niger van Tieghem MTCC 2425. Lett Appl Microbiol. 2007;25:22-23. Doi: 10.1046/j.1472-765X.1997.00164.x
Bibi I, Bhatti HN, AsgheR M. Decolourisation of direct dyes with manganese peroxidase from white rot basidiomycete Ganoderma lucidum-IBL-5. Can J Chem Engin. 2009;87:435-440. Doi: 10.1002/cjce.20165
Blanchette RA, Burnes TA. Selection of White-rot Fungi for Biopulping. Biomass. 1988;15:93-101. Doi: 10.1016/0144-4565(88)90099-6
Cañas AI, Camarero S. Laccases and their natural mediators: biotechnological tools for sustainable eco-friendly processes. Biotechnol Adv. 2010;28:694-705. Doi: 10.1016/j.biotechadv.2010.05.002.
De Jong EFP, De Vries JA, Field RP, Vander Z, De Bont JAM. Isolation and screening of basidiomycetes with high peroxidative activity. Mycol Res. 1992;96:1098-1104. Doi: 10.1016/S0953-7562(09)80121-4
Dhouib A, Hamza M, Zouari H, Mechichi T, Hmidi R, Labat M. Screening for ligninolytic enzyme production by diverse fungi from Tunisia. World J. Microbiol Biotechnol. 2005;21:1415-1423. Doi:10.1007/s11274-005-5774-z
Ferraz A, Córdova AM, Machuca A. Wood biodegradation and enzyme production by Ceriporiopsis subermispora during solid-state fermentation of Eucalyptus grandis. Enzyme Microb Technol. 2003;32(1):50-65. Doi: 10.1016/S0141-0229(02)00267-3
Flores C, Vidal C, Trejo-Hernández MR, Galindo E, Serrano-Carreón L. Selection of Trichoderma strains capable of increasing laccase production by Pleurotus ostreatus and Agaricus bisporus in dual cultures. J Appl Microbiol. 2009;106:249-257. Doi: 10.1111/j.1365-2672.2008.03998.x.
Fonseca MI, Shimizu E, Zapata PD, Villalba LL. Copper inducing effect on laccase production of white rot fungi native from Misiones (Argentina). Enzyme Microb Technol. 2010;46:534–539. Doi: 10.1016/j.enzmictec.2009.12.017
Giorgio EM, Fonseca MI, Tejerina MR, Ramos-HRYB AB, Sanabria N, Zapata PD, Villalba LL. Chips and sawdust substrates application for lignocellulolytic enzymes production by solid state fermentation. Int Res J Microbiol. 2012;3:120-127.
Gramss G, Günther T, Fritsche W. Spot tests for oxidative enzymes in ectomycorrhizal, wood-, and litter decaying fungi. Mycol Res. 1998;102(1):67-72. Doi: 10.1017/S095375629700436X
Herter S, Schmidt M, Thompson ML, Mikolasch A, Schauer F. Investigating the effects of metals on phenol oxidase-producing nitrogen-fixing Azotobacter chroococcum. J Basic Microbiol. 2013; 53(6):509-517. Doi: 10.1002/jobm.201100443.
Kersten P, Cullen D. Extracellular oxidative systems of the lignin-degrading Basidiomycete Phanerochaete chrysosporium. Fungal Genet Biol. 2007;44(2):77-87. Doi: 10.1016/j.fgb.2006.07.007
Levin L, Papinutti L, Forchiassin F. Evaluation of Argentinean white rot fungi for their ability to produce lignin-modifying enzymes and decolorize industrial dyes. Biores Technol. 2004;94(2):169-176. Doi: 10.1016/j.biortech.2003.12.002
Lin JP, Wei L, Xia LM, Cen PL. Production of laccase by Coriolus versicolor and its application in decolorization of dyestuffs: (I). Production of laccase by batch and repeated-batch processes. J Environ Sci. 2003;15(1):1-4. Doi: 1001-0742(2013)01-0005-04
López MJ, GUISADO G, VARGAS-GARCÍA MC, SUÁREZ-ESTRELLA F, MORENO J. Decolorization of industrial dyes by ligninolytic microorganisms isolated from composting environment. Enzyme Microb Technol. 2006;40(1):42-45. Doi: 10.1016/j.enzmictec.2005.10.035
Lundell TK, Mäkelä MR, Hildén K. Lignin-modifying enzymes in filamentous basidiomycetes-ecological, functional and phylogenetic review. J Basic Microbiol. 2010; 50(1):5-20. Doi: 10.1002/jobm.200900338.
Matos AJ, Bezerra RM, Dias AA. Screening of fungal isolates and properties of Ganoderma applanatum intended for olive mill wastewater decolourization and dephenolization. Lett Appl Microbiol. 2007;45(3):270-275. Doi: 10.1111/j.1472-765X.2007.02181.x
Millati R, Syamsiah S, Niklasson C, Nur Cahyanto M, Lundquist K, Taherzadeh M. Biological pretreatment of lignocelluloses with white-rot fungi and its applications: a review. BioRes. 2011;6(4):5224-5259.
Min KL, Kim YH, Kim YW, Jung HS, Hah YC. Characterization of a novel laccase produced by the woo-drotting fungus Phellinus ribis. Arch Biochem Biophys. 2001;392(2):279–286. Doi: 10.1006/abbi.2001.2459
Moon-Jeong H, Hyoung-Tae C, Hong-Gyu S. Purification and characterization of laccase from the white rot fungus Trametes versicolor. J Microbiol. 2005;43:555-560.
Morozova OV, Shumakovich GP, Gorbacheva MA, Shleev SV, Yaropolov AI. Blue laccases. Biochem (Moscow). 2007; 72(10):1136–1150. Doi: 10.1134/S0006297907100112
Murugesan K, Nam IH, Kim YM, Chang YS. Decolorization of reactive dyes by a thermostable laccase produced by Ganoderma lucidum in solid culture. Enzyme Microb Technol. 2007;40(7):1662-1672. Doi: 10.1016/j.enzmictec.2006.08.028
Ngono Ngane A, Ebelle Etame R, Ndifor F, Biyiti L, Amvam Zollo PH, Bouchet P. Antifungal activity of Chromolaena odorata (L.) King & Robinson (Asteraceae) of Cameroon. Chemother. 2006;52(2):103-106. Doi:10.1159/000092373
Nishida TK, Kashino Y, Mimura A, Takahara Y. Lignin biodegradation by wood rotting fungi I. Screening of lignin degradating fungi. Mokuzai Gakkaishi. 1988;34:530-536.
Pajot HF, Figueroa LIC, FAriña JI. Dye-decolorizing activity in isolated yeasts from the ecoregion of Las Yungas (Tucumán, Argentina). Enzyme Microb Technol. 2007;40:1503-1511. Doi: 10.1016/j.enzmictec.2006.10.038
Park KM, Park SS. Purification and characterization of laccase from basidiomycete Fomitella fraxinea. J Microbiol Biotechnol. 2008;18(4):670-675.
Rayner ADM, Boddy L. Fungal decomposition of wood: its biology and ecology. Editorial John Wiley & Sons Ltd., Chichester, Sussex, UK.; 1988.
Sánchez C. Lignocellulosic residues: biodegradation and bioconversion by fungi. Biotechnol Adv. 2009;27(2):185-194.
Saparrat MCN, Martínez MJ, Cabello MN, Arambarri AM. Screening for ligninolytic enzymes in autochthonous fungal strains from Argentina isolated from different substrata. Rev Iberoam Micol. 2002;19:181-185.
Serrano Silva I, Ragagnin De Menezes C, Franciscon E, Da Costa Dos Santos E, Durrant LR. Degradation of lignosulfonic and tannic acids by ligninolytic soil fungi cultivated under icroaerobic conditions. Braz. Arch. Biol. Technol. 2010;53(3):693-699. Doi: 10.1590/S1516-89132010000300026
Shimizu E, Velez-Rueda JO, Zapata PD, Villalba LL. Relación entre degradación de colorantes y oxidación de lignina residual causados por Ganoderma applanatum y Pycnoporus sanguineus en el licor negro kraft. Rev Cienc Tecnol. 2009; 12:46–51.
Sinsabaugh RL, Carreiro MM, Repert DA. Allocation of extracellular enzymatic activity in relation to litter composition, N deposition, and mass loss. Biogeochem. 2002;60:1-24. Doi: 10.1023/A:1016541114786
Tekere M, Mswaka AY, Zvauya R, Read JS. Growth, dye degradation and ligninolytic activity studies on Zimbabwean white rot fungi. Enzyme Microb Technol. 2001a;28(4-5):420-426. Doi: 10.1016/S0141-0229(00)00343-4
Tekere M, Zvauya R, Read JS. Ligninolytic enzyme production in selected sub-tropical white rot fungi under different culture conditions. J Basic Microbiol. 2001b.;41(2):115-129. Doi: 10.1002/1521-4028(200105)41:2<115::AID-JOBM115>3.0.CO;2-S
Toh Y, Jia J, Yen L, Obbard JP, Ting Y. Decolorization of azo dyes by white-rot fungi (WRF) isolated in Singapore. Enzyme Microb Technol. 2003;33:569-575. Doi: 10.1016/S0141-0229(03)00177-7
Villalba LL, Scott GM, Schoeder LR. Modification of loblolly pine chips with Ceriporiopsis subvermispora. Part 1: Effect of fungal treatment. J Wood Chem Technol. 2006;26(4):339-348. Doi 10.1080/02773810601105177
Zhao J, De Koker TH, Janse BJH. Comparative studies of lignin peroxidases and manganese-dependent peroxidases produced by selected white-rot fungi in solid media. FEMS Microb Lett. 1996;145:393-399. Doi: 10.1016/S0378-1097(96)00438-7
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. Lucas Martín Madrassi, Adriana Elizabet Alvarenga, María Celina Vedoya. (2024). Antagonistic activity of biocontrol agent Trichoderma spp. against Fusarium sp., the causal agent of Ananas comosus fruitlet rot. Bionatura Journal, 1(2), p.1. https://doi.org/10.70099/BJ/2024.02.01.11.
2. Mustansir Abbas, Uroosa Ejaz, Muhammad Sohail, Abdullah K. Alanazi. (2024). Application of Trametes pubescens for dye removal and biological pretreatment of sugarcane bagasse. Biofuels, Bioproducts and Biorefining, 18(2), p.453. https://doi.org/10.1002/bbb.2591.
3. Lucas Martín Madrassi, Adriana Elizabet Alvarenga, María Celina Vedoya. (2024). Antagonistic activity of biocontrol agent Trichoderma spp. against Fusarium sp., the causal agent of Ananas comosus fruitlet rot. Bionatura Journal, 1(2), p.1. https://doi.org/10.70099/BJ/2024.02.01.11.
4. Małgorzata P. Oksińska, Elżbieta G. Magnucka, Krzysztof Lejcuś, Anna Jakubiak-Marcinkowska, Sylwia Ronka, Andrzej W. Trochimczuk, Stanisław J. Pietr. (2019). Colonization and biodegradation of the cross-linked potassium polyacrylate component of water absorbing geocomposite by soil microorganisms. Applied Soil Ecology, 133, p.114. https://doi.org/10.1016/j.apsoil.2018.09.014.
5. Garima Singh, S. K. Dwivedi. (2025). Mechanistic, adsorption kinetics and confirmatory study of Congo red dye removal by native fungus Aspergillus niger. Biomass Conversion and Biorefinery, 15(15), p.21971. https://doi.org/10.1007/s13399-022-03369-1.
6. Guilherme Afonso Kessler de Andrade, Maria Victória Magalhães de Vargas, Sara Navarrete Bohi Goulart, Bruna Mota Bernardes, Jadson D.P. Bezerra, Rafael Plá Matielo Lemos, Filipe de Carvalho Victoria, Margéli Pereira de Albuquerque. (2023). Screening of endophytic fungi from Antarctic mosses: Potential production for L-asparaginase free of glutaminase and urease activity. Journal of Biotechnology, 377, p.1. https://doi.org/10.1016/j.jbiotec.2023.10.001.
7. M.P. Serbent, M.D. Rodríguez, C. Saux, I. Magario. (2025). Immobilization of fungal laccase on peanut shell carriers. Biocatalysis and Agricultural Biotechnology, 66, p.103581. https://doi.org/10.1016/j.bcab.2025.103581.
8. Aparecido Almeida Conceição, Joice Raisa Barbosa Cunha, Vandinelma Oliveira Vieira, Rubén Darío Romero Pelaéz, Simone Mendonça, João Ricardo Moreira Almeida, Eustáquio Souza Dias, Euziclei Gonzaga de Almeida, Félix Gonçalves de Siqueira. (2018). Biology of Macrofungi. Fungal Biology. , p.361. https://doi.org/10.1007/978-3-030-02622-6_18.
9. N S Sepwin, A Fijai, Y Mulyaningsih. (2019). Screening of Ligninolytic activity of some Basidiomycota from domestic Oil Palm Plantation in Bogor. Journal of Physics: Conference Series, 1402(3), p.033033. https://doi.org/10.1088/1742-6596/1402/3/033033.
10. Nadezhda V. Psurtseva, Anna A. Kiyashko, Svetlana V. Senik, Natalya V. Shakhova, Nina V. Belova. (2023). The Conservation and Study of Macromycetes in the Komarov Botanical Institute Basidiomycetes Culture Collection—Their Taxonomical Diversity and Biotechnological Prospects. Journal of Fungi, 9(12), p.1196. https://doi.org/10.3390/jof9121196.
11. Justinas Babinskas, Inga Matijošytė. (2025). Laccase Functional Analysis: Substrates, Activity Assays, Challenges, and Prospects. ChemBioChem, 26(7) https://doi.org/10.1002/cbic.202400939.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2015 Acta Biológica Colombiana

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
1. La aceptación de manuscritos por parte de la revista implicará, además de su edición electrónica de acceso abierto bajo licencia Attribution-NonCommercial-ShareAlike 4.0 (CC BY NC SA), la inclusión y difusión del texto completo a través del repositorio institucional de la Universidad Nacional de Colombia y en todas aquellas bases de datos especializadas que el editor considere adecuadas para su indización con miras a incrementar la visibilidad de la revista.
2. Acta Biológica Colombiana permite a los autores archivar, descargar y compartir, la versión final publicada, así como las versiones pre-print y post-print incluyendo un encabezado con la referencia bibliográfica del articulo publicado.
3. Los autores/as podrán adoptar otros acuerdos de licencia no exclusiva de distribución de la versión de la obra publicada (p. ej.: depositarla en un archivo telemático institucional o publicarla en un volumen monográfico) siempre que se indique la publicación inicial en esta revista.
4. Se permite y recomienda a los autores/as difundir su obra a través de Internet (p. ej.: en archivos institucionales, en su página web o en redes sociales cientificas como Academia, Researchgate; Mendelay) lo cual puede producir intercambios interesantes y aumentar las citas de la obra publicada. (Véase El efecto del acceso abierto).