MICROMORFOLOGÍA Y HISTOQUÍMICA DE LOS LATICÍFEROS DE ÓRGANOS VEGETATIVOS DE ESPECIES DE ASCLEPIADOIDEAE (APOCYNACEAE)
Micromorphology and Histochemistry of the Laticifers from Vegetative Organs of Asclepiadoideae species (Apocynaceae)
DOI:
https://doi.org/10.15446/abc.v20n1.42375Palabras clave:
Asclepiadeae, látex, metabolitos secundarios, pared celular. (es)Asclepiadeae, cell wall, latex, secondary metabolites. (en)
Descargas
La micromorfología y las principales clases de componentes de las paredes de los laticíferos y del látex de Asclepias curassavica, Fischeria stellata, Gonioanthela axillaris, Matelea denticulata y Oxypetalum banksii, fueron analizadas y comparadas con las de especies de Apocynaceae de Cerrado. Los laticíferos de las especies estudiadas de diferentes tipos de bosques presentan características semejantes. Las paredes son primarias, altamente hidratadas, pecto-celulósicas y fuertemente adheridas a las paredes de las células adyacentes. El látex es blanco lechoso, constituido por polisacáridos, incluyendo mucílago, proteínas, lípidos, incluyendo ácidos grasos, compuestos fenólicos e alcaloides. Fischeria y Gonioanthela difieren de las demás especies por producir lípidos neutros. El látex de las especies investigadas está asociado a la protección de la planta contra herbivoría, pudiendo ser tóxico e inhibir la proliferación de microorganismos, además de coagular, sellando heridas de las plantas y funcionando como barrera física. Estas propiedades del látex proporcionan una mayor ventaja adaptativa a estas plantas en relación a las no latescentes.
Artículo de investigación
MICROMORFOLOGÍA Y HISTOQUÍMICA DE LOS LATICÍFEROS DE ÓRGANOS VEGETATIVOS DE ESPECIES DE ASCLEPIADOIDEAE (APOCYNACEAE)
Micromorphology and Histochemistry of the Laticifers from Vegetative Organs of Asclepiadoideae species (Apocynaceae)
Diego DEMARCO1
1 Departamento de Botânica, Instituto de Biociências, Universidade de São Paulo, 05508-090, São Paulo, Brasil.
For correspondence: diegodemarco@usp.br
Received 28th February 2014, Returned for revision 24th July 2014, Accepted 20th August 2014.
Citation / Citar este artículo como: Demarco D. Micromorfología y histoquímica de los laticíferos de órganos vegetativos de especies de Asclepiadoideae (Apocynaceae). Acta biol. Colomb. 2015;20(1):57-65. doi: https://doi.org/10.15446/abc.v20n1.42375.
RESUMEN
La micromorfología y las principales clases de componentes de las paredes de los laticíferos y del látex de Asclepias curassavica, Fischeria stellata, Gonioanthela axillaris, Matelea denticulata y Oxypetalum banksii, fueron analizadas y comparadas con las de especies de Apocynaceae de Cerrado. Los laticíferos de las especies estudiadas de diferentes tipos de bosques presentan características semejantes. Las paredes son primarias, altamente hidratadas, pecto-celulósicas y fuertemente adheridas a las paredes de las células adyacentes. El látex es blanco lechoso, constituido por polisacáridos, incluyendo mucílago, proteínas, lípidos, incluyendo ácidos grasos, compuestos fenólicos e alcaloides. Fischeria y Gonioanthela difieren de las demás especies por producir lípidos neutros. El látex de las especies investigadas está asociado a la protección de la planta contra herbivoría, pudiendo ser tóxico e inhibir la proliferación de microorganismos, además de coagular, sellando heridas de las plantas y funcionando como barrera física. Estas propiedades del látex proporcionan una mayor ventaja adaptativa a estas plantas en relación a las no latescentes.
Palabras clave: Asclepiadeae, látex, metabolitos secundarios, pared celular.
ABSTRACT
Micromorphology and main substances of the laticifer walls and latex of Asclepias curassavica, Fischeria stellata, Gonioanthela axillaris, Matelea denticulata, and Oxypetalum banksii were analyzed and compared with those of Apocynacean Cerrado species. Laticifers of the studied species from different rainforest vegetations exhibit similar features. The walls are primary, highly hydrated, pectic-cellulosic, and firmly attached to adjacent cell walls. The latex is milky white, constituted by polysaccharides, including mucilage, proteins, lipids, including fatty acids, phenolic compounds, and alkaloids; however, Fischeria and Gonioanthela differ from the other species for also producing neutral lipids. The latex of the investigated species is related to plant defense against herbivory, and may be toxic and inhibit microorganism proliferation. Also, it can coagulate, sealing plants wounds and working as a physical barrier. These latex properties provide greater adaptive advantage to these plants in relation to non-latescent plants.
Keywords: Asclepiadeae, cell wall, latex, secondary metabolites.
INTRODUCCIÓN
Los laticíferos están presentes en todos los representantes de Apocynaceae sensu lato (Solereder, 1908; Metcalfe y Chalk, 1950) y su ocurrencia fue considerada por Metcalfe y Chalk (1950), una de las principales características que demuestran la relación próxima entre las familias, hasta entonces separadas, Apocynaceae y Asclepiadaceae. Actualmente, Asclepiadaceae corresponde a dos subfamilias de Apocynaceae (Endress y Bruyns, 2000).
Las paredes de los laticíferos pueden ser finas o engrosadas, son altamente hidratadas y contienen una gran proporción de sustancias pécticas y hemicelulosas (Fahn, 1979). La composición, el arreglo de las moléculas y el metabolismo de la pared celular están relacionados a las características del crecimiento de una célula vegetal (Carpita y McCann, 2000) y, aunque haya discordancia con relación al modo de crecimiento de los laticíferos en las especies de esta familia (Demarco et al., 2006), poco se sabe sobre sus paredes (Serpe et al., 2001; Serpe et al., 2002; Demarco et al., 2006).
El látex contiene una suspensión o emulsión de pequeñas partículas en un líquido (Fahn, 1979) que generalmente es denominado como el contenido vacuolar (Warnaar, 1982; Giordani y Lafon, 1993); sin embargo, actualmente es considerado el protoplasto del laticífero (Demarco et al., 2006), puesto que todo su protoplasto se libera en la gota de látex que exuda cuando el laticífero se ha roto. En algunos casos, la composición de la fracción particulada del látex puede servir como auxiliar en la distinción de especies morfológicamente semejantes, como ocurre en Hoya y Plumeria (Van Die, 1955; Baas et al., 1981).
Diversas sustancias pueden ser encontradas en el látex, como hidrocarburos poliisoprénicos, triterpenos y esteroides, ácidos grasos y aromáticos, carotenos, fosfolípidos, proteínas, constituyentes inorgánicos, azúcares, granos de almidón, taninos y alcaloides (Van Die, 1955; Fahn, 1979).
En Apocynaceae, el látex puede contener terpenos, tales como hidrocarburos poliisoprénicos (caucho) y triterpenos, ácidos grasos y aromáticos, fitoesteroles, polisacáridos, cardenolídeos, compuestos fenólicos, alcaloides y enzimas (Van Die, 1955; Yoder y Mahlberg, 1976; Groeneveld y van der Made, 1982; Warnaar, 1982; Giordani y Lafon, 1993; Appezzato-da-Glória y Estelita, 1997; Sacchetti et al., 1999; Giordani et al., 2000) y en ciertas especies, algunas de estas sustancias como el caucho y alcaloides poseen importancia económica y/o medicinal (Metcalfe y Chalk, 1950; Yoder y Mahlberg, 1976). El látex posee diferentes funciones como proteger contra herbivoria, ataque de microorganismos y sellar heridas de las plantas (Fahn, 1979; Farrell et al., 1991; Demarco et al., 2006; Pickard, 2008), confiriendo mayor éxito evolutivo a las plantas latescentes en relación a las no latescentes en los diversos ambientes (Farrell et al., 1991).
Aunque algunas técnicas histoquímicas sean ampliamente difundidas y actualmente haya diversos métodos para la detección de las más variadas sustancias, pocos investigadores han realizado estudios histoquímicos integrales de las estructuras secretoras de las plantas que contemplen diversas clases químicas de sustancias. A pesar de la gran cantidad de estudios químicos sobre los compuestos presentes en especies de Apocynaceae de importancia medicinal, pocos trabajos identificaron el lugar de producción de estas sustancias. En relación al látex de especies de Apocynaceae, la mayor parte de los estudios se restringe a una o pocas clases de sustancias (Wilson et al., 1976; Yoder y Mahlberg, 1976; Baas et al., 1981; Groeneveld y van der Made, 1982; Warnaar, 1982; Giordani y Lafon, 1993; Appezzato-da-Glória y Estelita, 1997; Sacchetti et al., 1999; Giordani et al., 2000) y apenas el estudio efectuado por Van Die (1955) tuvo un enfoque más incluyente, donde fue investigada la fracción particulada del látex de especies de 46 géneros pertenecientes a Rauvolfioideae y Apocynoideae. No hay estudios que hayan investigado si existe variación en la composición general del látex en diferentes ambientes.
Dada la falta de datos sobre la composición de la pared del laticífero y la información restricta sobre el látex de especies de Apocynaceae, en el presente trabajo se propone analizar la micromorfología de los laticíferos e identificar las principales clases químicas de las sustancias que componen las paredes y el látex de cinco especies de Asclepiadoideae de bosques; además de comparar los resultados obtenidos a partir de estas especies con los datos disponibles en la literatura sobre laticíferos de especies de Apocynaceae de cerrado (s.l.).
MATERIALES Y MÉTODOS
El material de estudio fue obtenido en el Parque Estadual da Serra do Mar – Núcleo Picinguaba, en el municipio de Ubatuba, São Paulo, Brasil (23#º21'S y 44#º51'W). Tres individuos de cada especie, pertenecientes a la tribu Asclepiadeae (Asclepiadoideae, Apocynaceae), fueron colectados para este estudio: individuos de Gonioanthela axillaris (Vell.) Fontella & E.A. Schwarz (D Demarco 35; 15/ julio/2005; D Demarco 48; 12/enero/2006; D Demarco 49; 12/enero/2006) y Oxypetalum banksii subsp. banksii Roem. & Schult. (D Demarco 57; 21/noviembre/2007; D Demarco 70; 03/marzo/2008) fueron colectados en bosques de restinga; individuos de Asclepias curassavica L. (D Demarco 52; 07/febrero/2006; D Demarco 66; 12/diciembre/2007; D Demarco 68; 13/diciembre/2007), Fischeria stellata E. Fourn. (D Demarco 58; 21/noviembre/2007; D Demarco 60; 21/ noviembre/2007) y Matelea denticulata (Vahl) Fontella & E.A. Schwarz (D Demarco 37; 16/julio/2005; D Demarco 38; 16/ julio/2005) fueron colectados en el bosque ombrófilo denso de tierras bajas. Material testigo de los individuos utilizados fueron depositados en el Herbario de la Universidade Estadual de Campinas (UEC 181929 a 181940).
Ramas vegetativas fueron cortadas con una lámina caliente para mantener el látex al interior de los laticíferos, luego fueron fijadas en FAA (formaldehído, ácido acético y alcohol etílico 50 %) por 24h (Johansen, 1940), FNT (formalina neutra tamponada; Lillie, 1965) y SFF (sulfato ferroso en formalina; Johansen, 1940) por 48h y almacenadas en alcohol etílico 70 %. Los tallos y hojas fijados en FAA fueron seccionados transversalmente para análisis micromorfológica de los laticíferos. Los fragmentos fueron deshidratados en una serie etílica, desecados por el método de punto crítico, montados y metalizados con oro. Las observaciones y el registro de imágenes fueron efectuadas en microscopio electrónico de barrido (MEB) Jeol JSM 5800 LV a 10 KV con cámara digital acoplada.
Para el análisis histoquímico, los ápices vegetativos fueron aislados, deshidratados en serie butílica (alcohol butílico terciario; Johansen, 1940), incluidos en "paraplast" y seccionados transversal y longitudinalmente con 10 μm de grosor en micrótomo rotativo (Microm HM340E). Algunas secciones fueron coloreadas con la triple coloración de Flemming (Johansen, 1940) y las láminas montadas en resina sintética para análisis de la composición de la pared de los laticíferos. Los materiales fijados en FAA fueron utilizados para evidenciar las sustancias hidrófilas, los fijados en FNT para las lipófilas y los fijados en SFF para confirmar los resultados de los compuestos fenólicos.
Los tratamientos realizados para evidenciar los principales componentes de las paredes de los laticíferos y del látex fueron: rojo de rutenio para pectinas (Johansen, 1940) y mucílagos ácidos (Gregory y Baas, 1989), ácido tánico y cloruro férrico para mucílago (Pizzolato, 1977), reacción PAS (Periodic-Acid-Schiff's reagent; pararosanilina C.I. 42500) para polisacáridos (Jensen, 1962), reactivo de Lugol para almidón (Johansen, 1940), azul de anilina (C.I. 42755) para calosa (Smith y McCully, 1978), negro de almidón B (C.I. 20470) para proteínas (Fisher, 1968), negro de Sudán B (C.I. 26150) y Sudán IV (C.I. 26105) para lípidos (Pearse, 1985), sulfato azul de Nilo (C.I. 51180) para lípidos ácidos y neutros (Cain, 1947), acetato de cobre y ácido rubeánico para ácidos grasos (Ganter y Jollés, 1969; Ganter y Jollés, 1970), cloruro férrico para compuestos fenólicos (Johansen, 1940), floroglucina acidificada para lignina (Johansen, 1940), reactivos de Dragendorff (Svendsen y Verpoorte, 1983) y Wagner (Furr y Mahlberg, 1981) para alcaloides. Las láminas histológicas del material fijado en sulfato ferroso en formalina y las dela prueba con negro de almidón B fueron montadas en resina sintética y las demás en gelatina glicerinada.
Los ápices caulinares fueron mantenidos por 48h en solución compuesta por metanol/cloroformo/agua/HCl (High, 1984) para realización del control de las pruebas para sustancias lipófilas. Después de este período, los materiales fueron fijados en FNT y SFF y recibieron el mismo tratamiento de las demás piezas. Los controles de las pruebas para sustancias hidrófilas fueron realizadas conforme las respectivas técnicas.
Las fotomicrografías fueron obtenidas en microscopio Olympus BX 51 y las escalas de las figuras fueron calculadas a través de la lámina micrométrica fotografiada en las mismas condiciones ópticas de las demás ilustraciones.
RESULTADOS
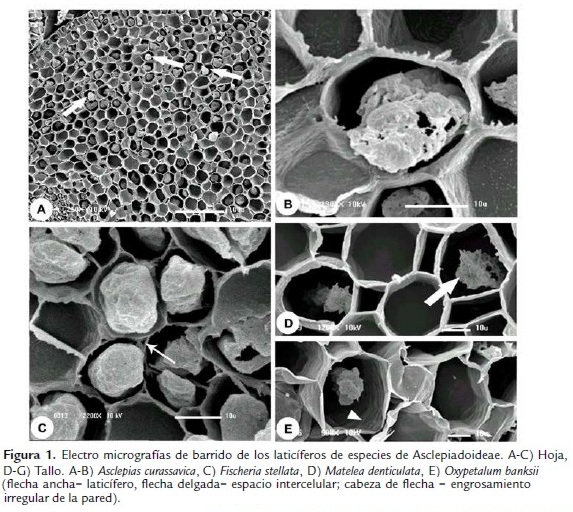
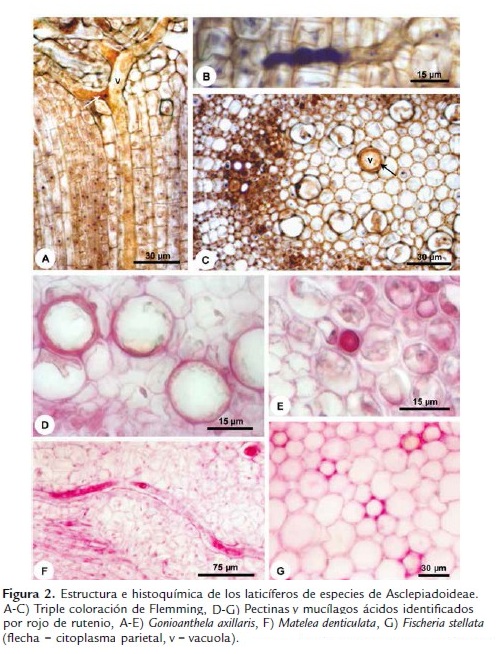
Los laticíferos de los tallos y hojas (Fig. 1) de Asclepias curassavica, Fischeria stellata, Gonioanthela axillaris, Matelea denticulata y Oxypetalum banksii poseen características semejantes en relación a su desarrollo. Las células de los meristemas primarios, entre las que se encuentran los ápices de los laticíferos, poseen paredes que colorean de naranja por la triple coloración de Flemming (Fig. 2A-B). Al ser adicionada a los laticíferos, las células comienzan a acumular secreción en su interior (Fig. 2B) y después de la disolución de las paredes transversales, la composición de las paredes es alterada y estas pasan a colorear de violeta (Fig. 2A,C). Esta afinidad diferenciada de las paredes por el colorante violeta cristal al inicio del desarrollo de los laticíferos persiste hasta que ellos alcanzan el diámetro máximo y cesan el crecimiento por estiramiento. En estas porciones maduras de los laticíferos, las paredes retornan a su mayor afinidad por el naranja G.
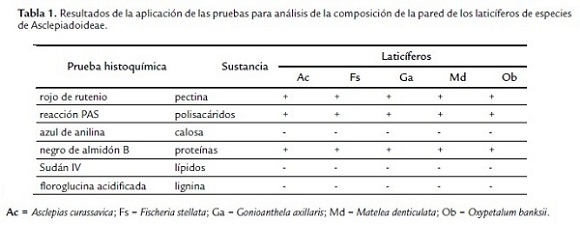
Las paredes de los laticíferos son primarias, altamente hidratadas, más gruesas que las paredes de las células parenquimáticas adyacentes y, aparentemente, con una alta concentración de pectinas (Fig. 2D-F), además de la identificación de polisacáridos totales (Fig. 3A-B) y proteínas (Fig. 3D). No se detectaron lípidos, lignina ni callosa (Tabla 1). En materiales procesados para microscopia electrónica de barrido, las paredes de los laticíferos poseen grosor semejante a las de las células parenquimáticas adyacentes (Fig. 1B-E) y tienen superficie levemente irregular (Fig. 1E, cabeza de flecha). Este engrosamiento irregular puede ser observado en algunas secciones transversales de los laticíferos (Fig. 2D).
La lamela media es especialmente gruesa y rica en pectina en la región de adhesión entre el laticífero y las células adyacentes desde sus porciones más jóvenes (Fig. 2G), con ausencia de espacios intercelulares en estas regiones (Figs. 1B, D-E, 2G y 3A). Espacios intercelulares son observados raramente entre el laticífero y las células adyacentes y cuando están presentes ocurren apenas en órganos adultos (Fig. 1C, flecha delgada).
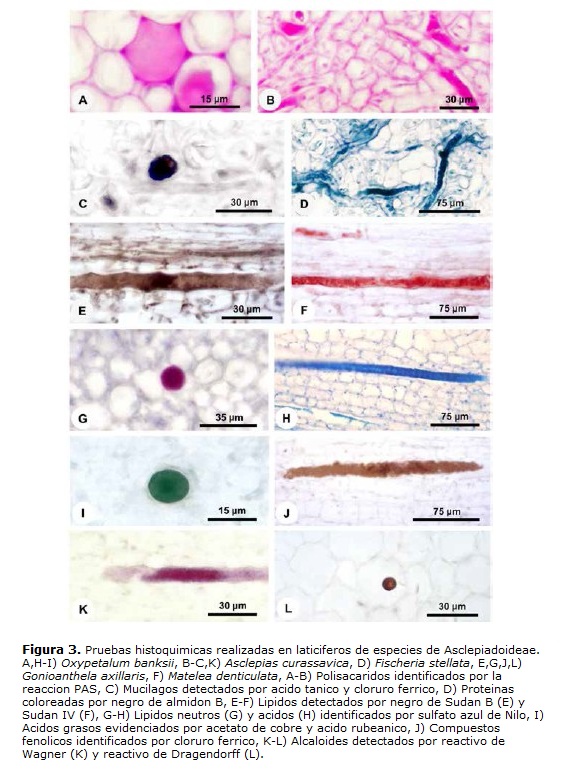
El látex es el propio protoplasto del laticífero y, en materiales bien fijados, llena el lumen celular (Figs. 1B-C, 2A-C, E-F y 3A-J). Este está compuesto por una vacuola única y central que ocupa la mayor parte del lumen celular, mientras que el restante del citoplasma y los núcleos están restringidos a una fina capa parietal, que puede ser distinguida por el color más intenso en la coloración estructural (Fig. 2A,C). In vivo, el látex de las cinco especies analizadas es blanco lechoso. Las diferentes sustancias que lo componen están presentes tanto en la vacuola central como en el citoplasma parietal y las pruebas histoquímicas evidenciaron los diferentes metabolitos en todo el protoplasto (Figs. 2E-F y 3A-K).
El látex está compuesto por diversas sustancias (Tabla 2) y esta heterogeneidad puede ser observada en la triple coloración de Flemming (Fig. 2A-C). El látex de Asclepias, Fischeria, Gonioanthela, Matelea y Oxypetalum posee los mismos metabolitos secundarios, siendo compuestos por polisacáridos (Fig. 3A-B), incluyendo mucílago (Figs. 2E-F y 3C), proteínas (Fig. 3D), lípidos (Fig. 3E-H), incluyendo ácidos grasos (Fig. 3I), compuestos fenólicos (Fig. 3J) y alcaloides (Fig. 3K-L, Tabla 2). La única excepción se refiere a los lípidos. En el tratamiento con sulfato azul de Nilo, lípidos ácidos fueron detectados en el látex de todas las especies (Fig. 3H, Tabla 2), mientras que lípidos neutros están presentes solo en Fischeria y Gonioanthela (Fig. 3G, Tabla 2). Al contrario de las demás sustancias comúnmente observadas en abundancia, los alcaloides fueron localizados siempre en cantidad reducida en las cinco especies (Fig. 3K-L).
No fue observada cualquier señal de herbivoría en los individuos de las especies investigadas y posteriores a la colecta, el látex inmediatamente se desborda por la región seccionada y algunos segundos después, disminuye el flujo y coagula, sellando la herida.
DISCUSIÓN
Los laticíferos de Asclepias curassavica, Fischeria stellata, Gonioanthela axillaris, Matelea denticulata y Oxypetalum banksii tienen paredes finas y eventualmente engrosadas de manera irregular, como ha sido descrito en la literatura para otras especies (Fahn, 1979; Mahlberg, 1993; Demarco et al., 2006).
Las paredes también pueden ser selladas por sustancias incrustantes o adcrustantes (Fineran et al., 1988); sin embargo, los laticíferos de las especies estudiadas no reaccionaron positivamente a las pruebas para calosa, suberina y lignina; sin embargo, así como en Aspidosperma australe y Blepharodon bicuspidatum (Demarco et al., 2006), sus paredes son exclusivamente pecto-celulósicas. Estas presentan características químicas distintas de las paredes de las demás células y varían a lo largo del desarrollo del laticífero. Con la utilización de la triple coloración de Flemming, se observó que las paredes de las porciones jóvenes de los laticíferos colorean de violeta y después estas alcanzan el diámetro máximo y cesan el crecimiento, las paredes pasan a colorearse de naranja. Estas diferencias no ocurren en las demás células de los tejidos adyacentes y demuestran que los laticíferos poseen paredes con composición distinta de las paredes de las demás células. Esta misma variación en las características de la pared también fue observada en Aspidosperma australe (Demarco et al., 2006).
La coloración violeta de las paredes de las porciones jóvenes de los laticíferos se debe al carácter ácido de estas y puede ser dado por una mayor proporción de pectinas, una vez que estas son polisacáridos heterogéneos, ramificados y altamente hidratados, ricos en ácido galacturónico (Carpita y McCann, 2000). En meristemos y células en expansión, donde las concentraciones de iones Ca2+ son mantenidas muy bajas, puede haber significativa de-esterificación de los homogalacturonanos (pectinas) sin que ocurra ligación con iones Ca2+, alterando la densidad de cargas y el pH local (Carpita y McCann, 2000). Eso puede explicar la coloración de las paredes e indicar que estas permanecen más expansibles cuando colorean en violeta, auxiliando en el aumento del diámetro celular y también acentuando su carácter higroscópico, debido a la presencia de gran cantidad de aniones debido al carácter ácido dado por la grande producción y concentración de pectinas, explicando la variación de grosor observada en materiales hidratados y deshidratados, como en microscopia eletrónica de barrido. Las paredes de los laticíferos colorean en naranja en las porciones maduras, pues parte de los aniones de las pectinas está interactuando con los iones Ca2+, haciendo las paredes más rígidas. Aunque los laticíferos de especies de Convolvulaceae sean sellados por una lamela de suberina (Fineran et al., 1988), los de Apocynaceae aparentemente no presentan ningún tipo de sustancia hidrofóbica en las paredes y son permeables al agua. Cuando un órgano es cortado, hay una disminución de la presión de turgor en los laticíferos y el agua de los tejidos adyacentes atraviesa la pared contribuyendo para la exudación del látex (Downton, 1981; Pickard, 2008), generando una exudación más profusa que en Convolvulaceae.
Serpe et al., (2001) y Serpe et al., (2002) investigaron imunocito químicamente las paredes de los laticíferos de Asclepias speciosa con relación a la presencia de sustancias que las diferencien de las paredes de las demás células; calosa, lignina y compuestos fenólicos no fueron encontrados, pero ellos constataron que la porción madura de los laticíferos tiene propiedades citoquímicas diferentes de las paredes de la porción más joven con relación a epitopos de pectina y que los diferentes componentes de la pared exhiben patrón de distribución distinta. Estas mismas características se observaron en el análisis histoquímico de las paredes de los laticíferos en el presente estudio y también fueron observadas durante el desarrollo de los laticíferos en especies de cerrado (Demarco et al., 2006).
En todas las especies analizadas, los laticíferos están fuertemente adheridos a las células adyacentes por regiones ricas en pectina desde sus porciones más jóvenes y raramente son observados espacios intercelulares a su alrededor y, cuando presentes, estos ocurren apenas en órganos adultos, después del término de crecimiento del laticífero. Debido a esta adhesión, se demuestra que es improbable la ocurrencia de crecimiento intrusivo y la actuación de pectinasas que faciliten el crecimiento del laticífero entre las células de los demás tejidos, como generalmente es referido para las especies de Apocynaceae (Wilson et al., 1976). En efecto, el estudio ontogenético de los laticíferos de las mismas cinco especies investigadas en este trabajo (Demarco y Castro, 2008) probó que estos son articulados anastomosados, cuya disolución de las paredes transversales ocurre próxima a sus ápices aún en medio a los meristemos primarios.
El látex está presente desde la porción más joven del laticífero y lo llena, correspondiendo a su propio protoplasto. Mediante la técnica de colecta utilizada en el presente estudio, este permanece bien preservado y las membranas intactas, con la vacuola central llenando casi todo el lumen celular y el restante del citoplasma con los núcleos ocupando posición periférica. No obstante, haya degeneración parcial del protoplasto de los laticíferos durante su desarrollo, este permanece vivo en los laticíferos maduros. Según algunos autores, el protoplasto puede permanecer intacto o degenerar en la madurez (Fahn, 1979); sin embargo, su desarreglo aparentemente se debe a un artefacto durante el corte y fijación del material debido a una desestabilización de su presión de turgor, alterando todo su contenido. En materiales fijados con los debidos cuidados, esto no ocurre.
El látex de las cinco especies es blanco lechoso, pero látex rojizo, amarillo y verdoso ya fueron registrados para otras especies de la familia (Solereder, 1908; Appezzato-da-Glória y Estelita, 1997) y su coloración puede variar hasta en una misma especie cuando comparada en diferentes órganos. En Mandevilla illustris e M. velutina (hoy M. pohliana), el látex de la parte aérea es blanco lechoso y el de la raíz es amarillo (Appezzato-da-Glória y Estelita, 1997). Este posee diversas clases químicas de sustancias que ocurren en todo el protoplasto y es compuesto por polisacáridos, incluyendo mucílago, proteínas, lípidos, incluyendo ácidos grasos, compuestos fenólicos y alcaloides. El látex de Fischeria stellata y Gonioanthela axillaris se diferencia de las demás especies por presencia también de lípidos neutros. Los alcaloides fueron detectados en pequeña cantidad en el presente estudio y según Yoder y Mahlberg (1976), hay diferencias cualitativas y cuantitativas en relación a los alcaloides dependiendo de la edad y del órgano analizado. Aunque el látex puede tener varios tipos de metabolitos secundarios, el generalmente es descrito teniendo predominantemente lípidos, especialmente terpenos (Van Die, 1955; Warnaar, 1982).
Aunque los componentes del látex puedan ser utilizados para diferenciar especies, como en el caso de Hoya (Baas et al., 1981) y Plumeria (Van Die, 1955), el látex de las especies investigadas posee composición muy semejante con relación a las clases de metabolitos, lo que aparentemente está relacionado a la proximidad filogenética entre estos cinco géneros que pertenecen a la misma tribu – Asclepiadeae. Las secreciones pueden variar dependiendo del ambiente en que la planta se encuentra; sin embargo, no hay diferencias significativas entre la constitución del látex en las diferentes formaciones vegetales. Fischeria stellata fue colectada en bosque ombrófilo denso de tierras bajas y Gonioanthela axillaris en bosque de restinga y ambas poseen látex de composición semejante (incluyendo los lípidos neutros), así como lo observado entre Asclepias curassavica y Matelea denticulata (tierras bajas) y Oxypetalum banksii (restinga). En especies de Apocynaceae ocurrentes en el cerrado también fueron detectados lípidos, polisacáridos y proteínas en Mandevilla illustris y M. velutina (hoy M. pohliana; Appezzato-da-Glória y Estelita, 1997). Esta semejanza encontrada indica que el mecanismo de defensa seleccionado evolutivamente es semejante en las especies de la familia.
El látex, en general, se asocia con la protección de la planta contra herbivoria, debido a la presencia de compuestos tóxicos o disuasivos alimenticios y compuestos que inhiben el crecimiento de microorganismos, además de sellar heridas a través de su coagulación (Farrell et al., 1991; Demarco et al., 2006). Algunos compuestos detectados en el látex de las especies estudiadas poseen estas propiedades, como los lípidos, compuestos fenólicos y alcaloides que pueden ser tóxicos e inhibir la proliferación de microorganismos, además de los lípidos tener la capacidad de coagular, sellando heridas y funcionando como una barrera física a la entrada de patógenos. La ausencia de depredación en todos los individuos de las especies investigadas y rápida coagulación del látex después de la colecta del material corroboran estas probables funciones. Otras diversas funciones ya fueron atribuidas al látex de Asclepias curassavica, como antifúngica, bactericida, actividades enzimáticas de glicosidasas involucradas en la degradación de la pared celular de hongos, proteasas, Rnasas, peroxidasas y ácido fosfatasas (Giordani et al., 2000), además de ser tóxico para larvas de insectos herbívoros generalistas (Dussourd y Hoyle, 2000). El látex también representa una defensa física, pues las mandíbulas del insecto pueden quedar adheridas entre sí o a la planta (Dussourd, 1990).
Sin embargo, algunos herbívoros desarrollaron estrategias co-evolutivamente para conseguir alimentarse de plantas latescentes. Muchos insectos mandibulados cortan la nervadura principal o el pecíolo para que el látex escurra y después se alimentan de la parte distal al corte, donde la cantidad de látex es mínima. En general, estos insectos rompen apenas una nervadura, pero en plantas con sistema ramificado de laticíferos, ellos hacen un surco através de la hoja, damnificando diversas nervaduras y todas las regiones del sistema y así interrumpen el flujo de látex (Dussourd, 1990). Otros insectos son capaces de secuestrar sustancias tóxicas de la planta hospedera, mientras están en el estado larval, para usarlas en su propia defensa contra los enemigos naturales (Trigo, 2000). Por otra parte, algunos parásitos están perfectamente adaptados a vivir en el látex. Especies de Phytomonas (Trypanosomatidae) son parásitos intralaticíferos o floemáticos de transmisión obligatoria por picadas de hemípteros fitófagos y ya fueron registrados en especies de Asclepias. Las plantas infectadas y no infectadas tienen la misma apariencia, no siendo patogénicas al hospedero (Solarte et al., 1995).
Segund Farrell et al., (1991), los linajes de plantas latescentes tiene mayor diversidad que sus grupos hermanos en diversos hábitats. De acuerdo a estos autores, los sistemas de canales o laticíferos promueven diversificación directamente – correlación genética, o indirectamente – aumentando el "fitness" individual. De esta forma, la innovación adaptativa, especialmente de defensa, confiere éxito evolutivo.
AGRADECIMIENTOS
El autor agradece a la Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP) por la beca de doctorado (proc. n#º 04/09729-4) concedida y por la financiación en el ámbito del Projeto Temático Gradiente Funcional (proc. n#º 03/12595-7), que hace parte del Programa BIOTA/ FAPESP–El Instituto Virtual da Biodiversidade (www.biota. org.br). Autorización COTEC/IF 41.065/2005y autorización IBAMA/CGEN 093/2005. Este trabajo se llevó a cabo en el Programa de Pós-Graduação em Biologia Vegetal, Instituto de Biologia, Universidade Estadual de Campinas.
BIBLIOGRAFÍA
Appezzato-da-Glória B, Estelita MEM. Laticifers systems in Mandevilla illustris and M. velutina Apocynaceae. Acta Soc Bot Pol. 1997;66:301-306. Doi: https://doi.org/10.5586/asbp.1997.035.
Baas WJ, Warnaar F, Niemann GJ. Investigations on Hoya species. VI. Latex composition and leaf phenolics and their taxonomic significance. Acta Bot Neerl. 1981;30:257-263.
Cain AJ. The use of Nile Blue in the examination of lipids. Quart J Microsc Sci. 1947;88:383-392.
Carpita N, McCann M. The cell wall. In: Buchanan BB, Gruissem W, Jones R, editors. Biochemistry and molecular biology of plants. Rockville: American Society of Plant Physiologists; 2000. p.52-108.
Demarco D, Castro M de M. Laticíferos articulados anastomosados em espécies de Asclepiadeae (Asclepiadoideae, Apocynaceae) e suas implicações ecológicas. Rev Bras Bot. 2008;31:699-711. Doi: https://doi.org/10.1590/S0100-84042008000400015.
Demarco D, Kinoshita LS, Castro M de M. Laticíferos articulados anastomosados – novos registros para Apocynaceae. Rev Bras Bot. 2006;29:133-144. Doi: https://doi.org/10.1590/S0100-84042006000100012
Downton WJS. Water relations of laticifers in Nerium oleander. Aust J Plant Physiol. 1981;8:329-334. Doi: 10.1071/PP9810329.
Dussourd DE. The vein drain; or, how insects outsmart plants. Nat Hist. 1990;90:44-49.
Dussourd DE, Hoyle AM. Poisoned plusiines: toxicity of milkweed latex and cardenolides to some generalist caterpillars. Chemoecology. 2000;10:11-16. Doi: 10.1007/PL00001810.
Endress ME, Bruyns PV. A revised classification of Apocynaceae s.l. Bot Rev. 2000;66:1-56. Doi: 10.1007/BF02857781.
Fahn A. Secretory tissues in plants. London: Academic Press; 1979. 302 p.
Farrell BD, Dussourd DE, Mitter C. Escalation of plant defense: do latex/resin canals spur plant diversification? Am Nat. 1991;138:881-900.
Fineran BA, Condon JM, Ingerfeld M. An impregnated suberized wall layer in laticifers of the Convolvulaceae and its resemblance to that in walls of oil cells. Protoplasma. 1988;147:42-54. Doi: 10.1007/BF01403876.
Fisher DB. Protein staining of ribboned epon sections for light microscopy. Histochemie. 1968;16:92-96. Doi: 10.1007/BF00306214.
Furr M, Mahlberg PG. Histochemical analyses of laticifers and glandular trichomes in Cannabis sativa. J Nat Prod. 1981;44:153-159. Doi: 10.1021/np50014a002.
Ganter P, Jollés G. Histochimie normale et pathologique. vol. 1. Paris: Gauthier–Villars; 1969. 923 p.
Ganter P, Jollés G. Histochimie normale et pathologique. vol. 2. Paris: Gauthier–Villars; 1970. 978 p.
Giordani R, Lafon L. A b-D-fucosidase from Asclepias curassavica latex. Phytochemistry. 1993;33:1327-1331. Doi: 10.1016/0031-9422(93)85084-5.
Giordani R, Tolla D, Regli P, Buc J. Role of terpenes from Asclepias curassavica latex for antifungal activity. J Mycol Med. 2000;10:34-38.
Gregory M, Baas P. A survey of mucilage cells in vegetative organs of the dicotyledons. Isr J Bot. 1989;38:125-174.
Groeneveld HW, van der Made LA. Cardenolide and triterpene synthesis in the laticifers of Asclepias curassavica L. Acta Bot Neerl. 1982;31:5-10.
High OB. Lipid histochemistry. New York: Oxford University Press; 1984. 68 p.
Jensen WA. Botanical histochemistry: principles and practice. San Francisco: W. H. Freeman and Co.; 1962. 408 p.
Johansen DA. Plant microtechnique. New York: McGraw-Hill; 1940. 523 p.
Lillie RD. Histopathologic technic and practical histochemistry. 3 ed. New York: McGraw-Hill; 1965. 715 p.
Metcalfe CR, Chalk L. Anatomy of the dicotyledons: leaves, stem and wood in relation to taxonomy with notes on economic uses. vol. 2. Oxford: Clarendon Press; 1950. 775 p.
Pearse AGE. Histochemistry: theoretical and applied. vol. 2. 4 ed. Edinburgh: C. Livingstone; 1985. 614 p.
Pickard WF. Laticifers and secretory ducts: two other tube systems in plants. New Phytol. 2008;177:877-888. Doi: 10.1111/j.1469-8137.2007.02323.x.
Pizzolato TD. Staining of Tilia mucilages with Mayer's tannic acid- ferric chloride. Bul Torrey Bot Club. 1977;104:277-279.
Sacchetti G, Ballero M, Serafini M, Romagnoli C, Bruni A, Poli F. Laticifer tissue distribution and alkaloid location in Vinca sardoa (Stearn) Pign. (Apocynaceae), an endemic plant of Sardinia (Italy). Phyton-Ann Rei Bot. 1999;39:265-275.
Serpe MD, Muir AJ, Keidel AM. Localization of cell wall polysaccharides in nonarticulated laticifers of Asclepias speciosa Torr. Protoplasma. 2001;216:215-226. Doi: 10.1007/BF02673873.
Serpe MD, Muir AJ, Driouich A. Immunolocalization of b-D-glucans, pectins, and arabinogalactan-proteins during intrusive growth and elongation of nonarticulated laticifers in Asclepias speciosa Torr. Planta. 2002;215:357-370. Doi: 10.1007/s00425-002-0756-y.
Smith MM, McCully ME. A critical evaluation of the specificity of aniline blue induce fluorescence. Protoplasma 1978;95:229-254. Doi: 10.1007/BF01294453.
Solarte RY, Moreno EA, Scorza JV. Flageliasis de plantas: comentarios sobre una revisión bibliográfica. Rev Ecol Lat Am. 1995;3:57-68.
Solereder H. Systematic anatomy of the dicotyledons. Oxford: vol. 1 y 2 Clarendon Press; 1908. 1182 p.
Svendsen AB, Verpoorte R. Chromatography of alkaloids. New York: Elsevier Scientific Publishing Company; 1983. 534 p.
Trigo JR. The chemistry of antipredator defense by secondary compounds in neotropical Lepidoptera: facts, perspectives and caveats. J Brazil Chem Soc. 2000;6:551-561. Doi: https://doi.org/10.1590/S0103-50532000000600002.
Van Die J. A comparative study of the particle fractions from Apocynaceae latices. Ann Bogor. 1955;2:1-124.
Warnaar F. Investigation on Hoya species. V. Determination of the amount of latex present in Hoya australis R. Br. ex Traill. and Hoya bella Hook. and its relation with shoot development. Z Pflanzenphysiol. 1982;105:307-314. Doi: 10.1016/S0044-328X(82)80026-3.
Wilson KJ, Nessler CL, Mahlberg PG. Pectinase in Asclepias latex and its possible role in laticifer growth and development. Am J Bot. 1976;63:1140-1144.
Yoder LR, Mahlberg PG. Reactions of alkaloid and histochemical indicators in laticifers and specialized parenchyma cells of Catharanthus roseus (Apocynaceae). Am J Bot. 1976;63:1167-1173.
Referencias
Appezzato-da-Glória B, Estelita MEM. Laticifers systems in Mandevilla illustris and M. velutina Apocynaceae. Acta Soc Bot Pol. 1997;66:301-306. Doi: https://doi.org/10.5586/asbp.1997.035.
Baas WJ, Warnaar F, Niemann GJ. Investigations on Hoya species. VI. Latex composition and leaf phenolics and their taxonomic significance. Acta Bot Neerl. 1981;30:257-263.
Cain AJ. The use of Nile Blue in the examination of lipids. Quart J Microsc Sci. 1947;88:383-392.
Carpita N, McCann M. The cell wall. In: Buchanan BB, Gruissem W, Jones R, editors. Biochemistry and molecular biology of plants. Rockville: American Society of Plant Physiologists; 2000. p.52-108.
Demarco D, Castro M de M. Laticíferos articulados anastomosados em espécies de Asclepiadeae (Asclepiadoideae, Apocynaceae) e suas implicações ecológicas. Rev Bras Bot. 2008;31:699-711. Doi: https://doi.org/10.1590/S0100-84042008000400015.
Demarco D, Kinoshita LS, Castro M de M. Laticíferos articulados anastomosados – novos registros para Apocynaceae. Rev Bras Bot. 2006;29:133-144. Doi: https://doi.org/10.1590/S0100-84042006000100012
Downton WJS. Water relations of laticifers in Nerium oleander. Aust J Plant Physiol. 1981;8:329-334. Doi: 10.1071/PP9810329.
Dussourd DE. The vein drain; or, how insects outsmart plants. Nat Hist. 1990;90:44-49.
Dussourd DE, Hoyle AM. Poisoned plusiines: toxicity of milkweed latex and cardenolides to some generalist caterpillars. Chemoecology. 2000;10:11-16. Doi: 10.1007/PL00001810.
Endress ME, Bruyns PV. A revised classification of Apocynaceae s.l. Bot Rev. 2000;66:1-56. Doi: 10.1007/BF02857781.
Fahn A. Secretory tissues in plants. London: Academic Press; 1979. 302 p.
Farrell BD, Dussourd DE, Mitter C. Escalation of plant defense: do latex/resin canals spur plant diversification? Am Nat. 1991;138:881-900.
Fineran BA, Condon JM, Ingerfeld M. An impregnated suberized wall layer in laticifers of the Convolvulaceae and its resemblance to that in walls of oil cells. Protoplasma. 1988;147:42-54. Doi: 10.1007/BF01403876.
Fisher DB. Protein staining of ribboned epon sections for light microscopy. Histochemie. 1968;16:92-96. Doi: 10.1007/BF00306214.
Furr M, Mahlberg PG. Histochemical analyses of laticifers and glandular trichomes in Cannabis sativa. J Nat Prod. 1981;44:153-159. Doi: 10.1021/np50014a002.
Ganter P, Jollés G. Histochimie normale et pathologique. vol. 1. Paris: Gauthier–Villars; 1969. 923 p.
Ganter P, Jollés G. Histochimie normale et pathologique. vol. 2. Paris: Gauthier–Villars; 1970. 978 p.
Giordani R, Lafon L. A b-D-fucosidase from Asclepias curassavica latex. Phytochemistry. 1993;33:1327-1331. Doi: 10.1016/0031-9422(93)85084-5.
Giordani R, Tolla D, Regli P, Buc J. Role of terpenes from Asclepias curassavica latex for antifungal activity. J Mycol Med. 2000;10:34-38.
Gregory M, Baas P. A survey of mucilage cells in vegetative organs of the dicotyledons. Isr J Bot. 1989;38:125-174.
Groeneveld HW, van der Made LA. Cardenolide and triterpene synthesis in the laticifers of Asclepias curassavica L. Acta Bot Neerl. 1982;31:5-10.
High OB. Lipid histochemistry. New York: Oxford University Press; 1984. 68 p.
Jensen WA. Botanical histochemistry: principles and practice. San Francisco: W. H. Freeman and Co.; 1962. 408 p.
Johansen DA. Plant microtechnique. New York: McGraw-Hill; 1940. 523 p.
Lillie RD. Histopathologic technic and practical histochemistry. 3 ed. New York: McGraw-Hill; 1965. 715 p.
Metcalfe CR, Chalk L. Anatomy of the dicotyledons: leaves, stem and wood in relation to taxonomy with notes on economic uses. vol. 2. Oxford: Clarendon Press; 1950. 775 p.
Pearse AGE. Histochemistry: theoretical and applied. vol. 2. 4 ed. Edinburgh: C. Livingstone; 1985. 614 p.
Pickard WF. Laticifers and secretory ducts: two other tube systems in plants. New Phytol. 2008;177:877-888. Doi: 10.1111/j.1469-8137.2007.02323.x.
Pizzolato TD. Staining of Tilia mucilages with Mayer's tannic acid- ferric chloride. Bul Torrey Bot Club. 1977;104:277-279.
Sacchetti G, Ballero M, Serafini M, Romagnoli C, Bruni A, Poli F. Laticifer tissue distribution and alkaloid location in Vinca sardoa (Stearn) Pign. (Apocynaceae), an endemic plant of Sardinia (Italy). Phyton-Ann Rei Bot. 1999;39:265-275.
Serpe MD, Muir AJ, Keidel AM. Localization of cell wall polysaccharides in nonarticulated laticifers of Asclepias speciosa Torr. Protoplasma. 2001;216:215-226. Doi: 10.1007/BF02673873.
Serpe MD, Muir AJ, Driouich A. Immunolocalization of b-D-glucans, pectins, and arabinogalactan-proteins during intrusive growth and elongation of nonarticulated laticifers in Asclepias speciosa Torr. Planta. 2002;215:357-370. Doi: 10.1007/s00425-002-0756-y.
Smith MM, McCully ME. A critical evaluation of the specificity of aniline blue induce fluorescence. Protoplasma 1978;95:229-254. Doi: 10.1007/BF01294453.
Solarte RY, Moreno EA, Scorza JV. Flageliasis de plantas: comentarios sobre una revisión bibliográfica. Rev Ecol Lat Am. 1995;3:57-68.
Solereder H. Systematic anatomy of the dicotyledons. Oxford: vol. 1 y 2 Clarendon Press; 1908. 1182 p.
Svendsen AB, Verpoorte R. Chromatography of alkaloids. New York: Elsevier Scientific Publishing Company; 1983. 534 p.
Trigo JR. The chemistry of antipredator defense by secondary compounds in neotropical Lepidoptera: facts, perspectives and caveats. J Brazil Chem Soc. 2000;6:551-561. Doi: https://doi.org/10.1590/S0103-50532000000600002.
Van Die J. A comparative study of the particle fractions from Apocynaceae latices. Ann Bogor. 1955;2:1-124.
Warnaar F. Investigation on Hoya species. V. Determination of the amount of latex present in Hoya australis R. Br. ex Traill. and Hoya bella Hook. and its relation with shoot development. Z Pflanzenphysiol. 1982;105:307-314. Doi: 10.1016/S0044-328X(82)80026-3.
Wilson KJ, Nessler CL, Mahlberg PG. Pectinase in Asclepias latex and its possible role in laticifer growth and development. Am J Bot. 1976;63:1140-1144.
Yoder LR, Mahlberg PG. Reactions of alkaloid and histochemical indicators in laticifers and specialized parenchyma cells of Catharanthus roseus (Apocynaceae). Am J Bot. 1976;63:1167-1173.
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. Ellenhise Ribeiro Costa, Diego Demarco. (2024). Development and Holocrine Secretion of Resin Ducts in Kielmeyera appariciana (Calophyllaceae). Plants, 13(13), p.1757. https://doi.org/10.3390/plants13131757.
2. Diego Demarco. (2017). Histochemistry of Single Molecules. Methods in Molecular Biology. 1560, p.313. https://doi.org/10.1007/978-1-4939-6788-9_24.
3. Erika Prado, Diego Demarco. (2018). Ecosystem Services and Global Ecology. https://doi.org/10.5772/intechopen.75705.
4. Danesha Naidoo, Yougasphree Naidoo, Gonasageran Naidoo, Farzad Kianersi, Yaser Hassan Dewir. (2023). Histochemical Analysis and Ultrastructure of Trichomes and Laticifers of Croton gratissimus Burch. var. gratissimus (Euphorbiaceae). Plants, 12(4), p.772. https://doi.org/10.3390/plants12040772.
5. Marcelo R. Pace, Israel L. Cunha Neto, Leyde N. N. Santos‐Silva, Gladys F. A. Melo‐de‐Pinna, Pedro Acevedo‐Rodríguez, Rafael F. Almeida, André M. Amorim, Veronica Angyalossy. (2019). First report of laticifers in lianas of Malpighiaceae and their phylogenetic implications. American Journal of Botany, 106(9), p.1156. https://doi.org/10.1002/ajb2.1350.
6. Diego Demarco. (2017). Floral glands in asclepiads: structure, diversity and evolution. Acta Botanica Brasilica, 31(3), p.477. https://doi.org/10.1590/0102-33062016abb0432.
7. Wallace T. Cruz, Eduardo H.S. Bezerra, Márcio V. Ramos, Bruno A.M. Rocha, Maria C. Medina, Diego Demarco, Cristina Paiva S. Carvalho, Jefferson S. Oliveira, Jeanlex S. Sousa, Pedro F.N. Souza, Valder N. Freire, Francisca M.S. da Silva, Cleverson D.T. Freitas. (2020). Crystal structure and specific location of a germin-like protein with proteolytic activity from Thevetia peruviana. Plant Science, 298, p.110590. https://doi.org/10.1016/j.plantsci.2020.110590.
8. Josiana Cristina Ribeiro, Marcelo José Pena Ferreira, Diego Demarco. (2017). Colleters in Asclepiadoideae (Apocynaceae): Protection of Meristems against Desiccation and New Functions Assigned. International Journal of Plant Sciences, 178(6), p.465. https://doi.org/10.1086/692295.
9. E.D. Tölke, A.P.S. Lacchia, E.A. Lima, D. Demarco, L. Ascensão, S.M. Carmello-Guerreiro. (2021). Secretory ducts in Anacardiaceae revisited: Updated concepts and new findings based on histochemical evidence. South African Journal of Botany, 138, p.394. https://doi.org/10.1016/j.sajb.2021.01.012.
10. Diego Demarco. (2023). Histochemistry of Single Molecules. Methods in Molecular Biology. 2566, p.291. https://doi.org/10.1007/978-1-0716-2675-7_24.
11. Carolina Krebs Kleingesinds, Bruno Nobuya Katayama Gobara, Dioceni Mancilha, Maria Aurineide Rodrigues, Diego Demarco, Helenice Mercier. (2018). Impact of tank formation on distribution and cellular organization of trichomes within Guzmania monostachia rosette. Flora, 243, p.11. https://doi.org/10.1016/j.flora.2018.03.013.
12. M. Haritha, D. Leena Lavanya, T. Muthukumar. (2025). Morphological, Anatomical, and Histochemical Study of Cordia diffusa K.C. Jacob—A Steno Endemic Plant. Microscopy Research and Technique, 88(5), p.1273. https://doi.org/10.1002/jemt.24791.
13. Seher Güven. (2025). A Comprehensive Micromorphological and Anatomical Study on the Poisonous Plant Cionura erecta (L.) Griseb. (Apocynaceae: Asclepiadoideae). Kahramanmaraş Sütçü İmam Üniversitesi Tarım ve Doğa Dergisi, 28(5), p.1159. https://doi.org/10.18016/ksutarimdoga.vi.1691710.
14. Thália do Socorro Serra Gama, Vinicius Santos Rubiano, Diego Demarco. (2017). Laticifer development and its growth mode in Allamanda blanchetii A. DC. (Apocynaceae). The Journal of the Torrey Botanical Society, 144(3), p.303. https://doi.org/10.3159/TORREY-D-16-00050.
15. Clarissa Naidoo, Yougasphree Naidoo, Yaser Hassan Dewir. (2020). The Secretory Apparatus of Tabernaemontana ventricosa Hochst. ex A.DC. (Apocynaceae): Laticifer Identification, Characterization and Distribution. Plants, 9(6), p.686. https://doi.org/10.3390/plants9060686.
16. Márcio Viana Ramos, Diego Demarco, Isabel Cristina da Costa Souza, Cleverson Diniz Teixeira de Freitas. (2019). Laticifers, Latex, and Their Role in Plant Defense. Trends in Plant Science, 24(6), p.553. https://doi.org/10.1016/j.tplants.2019.03.006.
17. Diego Demarco. (2017). Staminal wing and a novel secretory structure of asclepiads. Botany, 95(7), p.763. https://doi.org/10.1139/cjb-2016-0239.
18. Maria Camila Medina, Mariane S. Sousa-Baena, Marie-Anne Van Sluys, Diego Demarco. (2022). Laticifer growth pattern is guided by cytoskeleton organization. Frontiers in Plant Science, 13 https://doi.org/10.3389/fpls.2022.971235.
19. Imen Lahmar, Mouhiba Ben Nasri-Ayachi, Karima Belghith, Riaz Ullah. (2022). Laticifer Identification, Rubber Characterization, Phenolic Content, and Antioxidant Activity of Pergularia tomentosa Latex Extract. BioMed Research International, 2022(1) https://doi.org/10.1155/2022/7158905.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2015 Acta Biológica Colombiana

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
1. La aceptación de manuscritos por parte de la revista implicará, además de su edición electrónica de acceso abierto bajo licencia Attribution-NonCommercial-ShareAlike 4.0 (CC BY NC SA), la inclusión y difusión del texto completo a través del repositorio institucional de la Universidad Nacional de Colombia y en todas aquellas bases de datos especializadas que el editor considere adecuadas para su indización con miras a incrementar la visibilidad de la revista.
2. Acta Biológica Colombiana permite a los autores archivar, descargar y compartir, la versión final publicada, así como las versiones pre-print y post-print incluyendo un encabezado con la referencia bibliográfica del articulo publicado.
3. Los autores/as podrán adoptar otros acuerdos de licencia no exclusiva de distribución de la versión de la obra publicada (p. ej.: depositarla en un archivo telemático institucional o publicarla en un volumen monográfico) siempre que se indique la publicación inicial en esta revista.
4. Se permite y recomienda a los autores/as difundir su obra a través de Internet (p. ej.: en archivos institucionales, en su página web o en redes sociales cientificas como Academia, Researchgate; Mendelay) lo cual puede producir intercambios interesantes y aumentar las citas de la obra publicada. (Véase El efecto del acceso abierto).