Publicado
Isolation of the Human Cytomegalovirus from bodily fluids
Aislamiento de citomegalovirus humano a partir de fluidos corporales
DOI:
https://doi.org/10.15446/abc.v24n3.79406Palabras clave:
Body fluids, cell culture, cytomegalovirus, isolation (en)Aislamiento, citomegalovirus, cultivo de célula, líquidos corporales (es)
Descargas
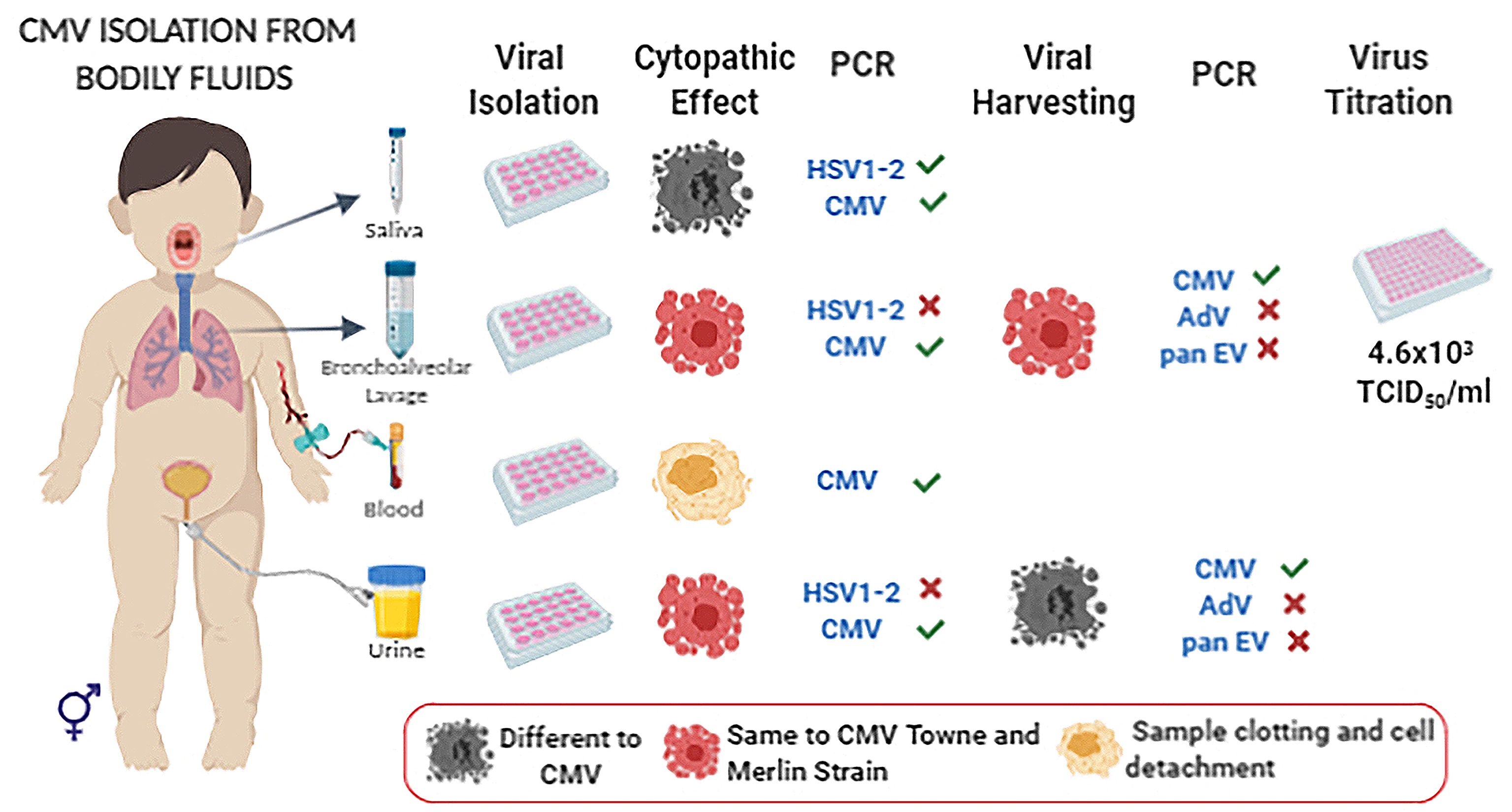
In vitro studies on the pathogenesis of the human cytomegalovirus (HCMV) are conducted regularly using laboratory adapted strains that lose some characteristics during the adaptation process. Since HCMV is excreted from bodily fluids during infection or reactivation, this work aimed to isolate and culture HCMV from the MRC-5 human cells found in the urine, bronchoalveolar lavage, saliva, and plasma samples of pediatric patients with probable or confirmed infection. The samples were inoculated on cell cultures either for 14 days or until a cytopathic effect (CPE) of 80 % was observed. The cell lysates and supernatants were used to perform successive viral passages. Besides HCMV, the herpes simplex virus was detected from all the saliva samples. Inoculation of the HCMV positive sera induced cell clustering and immediate monolayer damage that restricted their use. One sample of bronchoalveolar lavage induced a CPE after inoculation like that of the HCMV reference strains (Towne and Merlin), which was consequently propagated and titrated. A second viral isolate derived from the urine sample of a patient with congenital infection did not demonstrate a CPE, although presence of the virus had been confirmed using PCR. The viral isolates were examined and found to be negative for adenoviruses or enteroviruses. Despite the evident difficulty encountered for the isolation and harvesting of the HCMV, this work shows that it was possible to obtain a low passage viral strain using a modified shell vial method and inoculation protocol with extended follow-up and confirmation.
Estudios in vitro de la patogénesis del citomegalovirus humano (HCMV) se hace empleando cepas adaptadas de laboratorio que han perdido algunas de sus características durante ese proceso. En vista que el HCMV se excreta en distintos fluidos corporales, dependiendo de la condición clínica del paciente, en este trabajo se propuso aislar y propagar HCMV en fibroblastos MRC-5 usando muestras de orina, lavado broncoalveolar, saliva y plasma de pacientes pediátricos. Estas muestras fueron inoculadas sobre los cultivos celulares por 14 días o hasta alcanzar un efecto citopático en el 80 % de la monocapa. El lisado celular y el sobrenadante del aislamiento se usaron para hacer pasajes virales sucesivos. Además de HCMV, el virus de herpes simple se aisló en todas las muestras de saliva. Con el empleo de los sueros positivos para HCMV se observó la formación de agregados y daño inmediato en la monocapa que impidieron su uso. Una muestra de lavado broncoalveolar indujo ECP desde la inoculación, similar al control positivo para HCMV (cepas Towne y Merlin), por lo que fue propagada y se tituló. Un segundo aislamiento viral obtenido de la orina de un paciente con infección congénita no produjo ECP a pesar de ser confirmado por PCR. En los aislamientos llevados hasta el pasaje 1, se descartó la presencia de enterovirus y adenovirus. A pesar de la evidente dificultad para aislar y propagar el HCMV, fue posible obtener un aislamiento usando un protocolo de Shell vial e inoculación modificado, y con un seguimiento prolongado del proceso.
Referencias
An P, Sáenz Robles MT, Duray AM, Cantalupo PG, Pipas JM. Human polyomavirus BKV infection of endothelial cells results in interferon pathway induction and persistence. PLoS Pathog. 2019;15(1):e1007505. Doi: https://doi.org/10.1371/journal.ppat.1007505
Ban T. Cytomegalovirus Treatment. Curr Treat Options Infect Dis. 2014;6(3):256–270. Doi: https://doi.org/10.1007/s40506-014-0021-5
Bohórquez SP, Díaz J, Rincón CM, Estupiñán M, Chaparro M, Low-Calle AM, et al. Shedding of HSV-1, HSV-2, CMV, and EBV in the saliva of hematopoietic stem cell transplant recipients at Fundación HOMI - Hospital de la Misericordia, Bogotá, D.C. Biomedica. 2016:19;36(0):201-210. Doi: https://doi.org/10.7705/biomedica.v36i0.2985
Britt WJ. Congenital Human Cytomegalovirus infection and the enigma of the maternal immunity. J Virol. 2017;91(15):e02392-16. Doi: https://doi.org/10.1128/JVI.02392-16
Dou Y, Li Y, Ma C, Zhu H, Du J, Liu H, et al. Rapid diagnosis of human adenovirus B, C and E in the respiratory tract using multiplex quantitative polymerase chain reaction. Mol Med Rep. 2018;18(3):2889-2897. Doi: https://doi.org/10.3892/mmr.2018.9253
Elder E, Sinclair J. HCMV latency: what regulates the regulators? Med Microbiol Immunol. 2019;208(3-4): 431-438. Doi: https://doi.org/10.1007/s00430-019-00581-1
Gouarin S, Gault E, Vabret A, Cointe D, Rozenberg F, Grangeot-Keros L, et al. Real-time PCR quantification of human cytomegalovirus DNA in amniotic fluid samples from mothers with primary infection. J Clin Microbiol. 2002;40(5):1767-1772. Doi: https://doi.org/10.1128/jcm.40.5.1767-1772.2002
Gregory WW, Menegus MA. Practical protocol for cytomegalovirus isolation: use of MRC-5 cell monolayers incubated for 2 weeks. J Clin Microbiol. 1983;17(4):605-609.
Griffiths P, Baraniak I, Reeves M. The pathogenesis of human cytomegalovirus. J Pathol. 2015; 235(2):288-297. Doi: https://doi.org/10.1002/path.4437
Hamprecht K, Maschmann J, Jahn G, Poets C.F, Goelz R. Cytomegalovirus transmission to preterm infants durin glactation. J Clin Virol. 2008;41(3):198-205. Doi: https://doi.org/10.1016/j.jcv.2007.12.005
Hematian A, Sadeghifard N, Mohebi R, Taherikalani M, Nasrolahi A, Amraei M, et al. Traditional and Modern Cell Culture in Virus Diagnosis. Osong Public Health Res Perspect. 2016;7(2):77-82. Doi: https://doi.org/10.1016/j.phrp.2015.11.011
Jain S, Williams DJ, Arnold SR, Ampofo K, Bramley AM, Reed C, et al. Community-acquired pneumonia requiring hospitalization among U.S. children. N Engl J Med. 2015;372(9):835-845. Doi: https://doi.org/10.1056/NEJMoa1405870
Kaufman HE, Azcuy AM, Varnell ED, Sloop GD, Thompson HW, Hill JM. HSV-1 DNA in tears and saliva of normal adults. Invest Ophthalmol Vis Sci. 2005;46(1):241-247.
Knowles WA. Discovery and epidemiology of the human polyomaviruses BK virus (BKV) and JC virus (JCV). Adv Exp Med Biol. 2006;577:19-45. Doi: https://doi.org/10.1007/0-387-32957-9_2
Lancini D, Faddy H, Flower R, Hogan C. Cytomegalovirus disease in immunocompetent adults. Med J Aust. 2014;201(10):578-580.
Leland D1, Hansing RL, French ML. Clinical experience with cytomegalovirus isolation using conventional cell cultures and early antigen detection in centrifugation-enhanced shell vial cultures. J Clin Microbiol. 1989;27(6):1159-1162.
Leland DS, Ginocchio CC. Role of cell culture for virus detection in the age of technology. Clin Microbiol Rev. 2007;20(1):49-78. Doi: https://doi.org/10.1128/CMR.00002-06
Montes M, Oñate E, Muguruza A, Tamayo E, Carrera IM, Iturzaeta A, et al. Enterovirus D68 Causing Acute Respiratory Infection: Clinical Characteristics and Differences With Acute Respiratory Infections Associated With Enterovirus Non-D68. Pediatr Infect Dis J. 2019;38(7):687-691. Doi: https://doi.org/10.1097/INF.0000000000002289
Nozawa C, Hattori LY, Galhardi LC, Lopes N, Bomfim WA, Cândido LK, et al. Herpes simplex virus: isolation, cytopathological characterization and antiviral sensitivity. An Bras Dermatol. 2014;89(3):448-452. Doi: https://doi.org/10.1590/abd1806-4841.20142574
Paz-Bailey G, Ramaswamy M, Hawkes SJ, Geretti AM. Herpes simplex virus type 2: epidemiology and management options in developing countries. Postgrad Med J. 2008;84(992):299-306. Doi: https://doi.org/10.1136/sti.2006.020966
Prichard M, Penfold M, Duke G, Spaete R, Kemble G. A review of genetic differences between limited and extensively passaged human cytomegalovirus strains. Rev Med Virol. 2001;11(3):191-200.
Razonable RR, Hayden RT. Clinical utility of viral load in management of Cytomegalovirus infection after solid organ transplantation. Clin Microbiol Rev. 2013;26(4):703-727. Doi: https://doi.org/10.1128/CMR.00015-13
Requião-Moura LR, deMatos AC, Pacheco-Silva A. Cytomegalovirus infection in renal transplantation: clinical aspects, management and the perspectives. Einstein (Sao Paulo). 2015;13(1):142-148. Doi: https://doi.org/10.1590/S1679-45082015RW3175
Stanton RJ, Baluchova K, Dargan DJ, Cunningham C, Sheehy O, Seirafian S, et al. Reconstruction of the complete human cytomegalovirus genome in a BAC reveals RL13 to be a potent inhibitor of replication. J Clin Invest. 2010; 120(9):3191-3208. Doi: https://doi.org/10.1172/JCI42955
Sun Y, Kum R, Hoon S, Pei P. Detection and genotyping of human herpes simplex viruses in cutaneous lesions of erythema multiforme by nested PCR. J Med Virol. 2003;71(3):423-428. Doi: https://doi.org/10.1002/jmv.10502
Twite N, Andrei G, Kummert C, Donner C, Perez-Morga D, De Vos R, et al. Sequestration of human cytomegalovirus by human renal and mammary epithelial cells. Virology. 2014;460(461):55-65. Doi: https://doi.org/10.1016/j.virol.2014.04.032
Vanarsdall A, Ryckman BJ, Chase M, Johnson D. Human cytomegalovirus glycoproteins gB and gH/gL mediate epithelial cell-cell fusion when expressed either in cis or in trans. J Virol. 2008;82(23):11837-11850. Doi: https://doi.org/10.1128/JVI.01623-08
Vestergaard HT, Thomsen MK, Nielsen L, Panum I. Diagnostics of congenital cytomegalovirus in Denmark. Ugeskr Laeger. 2018;180(50):pii: V03180221.
Wilkinson GW, Davison AJ, Tomasec P, Fielding CA, Aicheler R, Murrell I, et al. Human cytomegalovirus: taking the strain. Med Microbiol Immunol. 2015;204(3):273-284. Doi: https://doi.org/10.1007/s00430-015-0411-4
Zhang S, Wang J, Yan Q, He S, Zhou W, Ge S, Xia N. A one-step, triplex, real-time RT-PCR assay for the simultaneous detection of enterovirus 71, coxsackie A16 and pan-enterovirus in a single tube. PLoS One. 2014;9(7):e102724. Doi: https://doi.org/10.1371/journal.pone.0102724
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. Sara Delgadillo-Barrera, Lilia J. Bernal-Cepeda, Sigrid Camacho-Ortega, Sonia P. Bohórquez-Avila, Jaime E. Castellanos. (2025). Cyclooxygenase upregulation in cytomegalovirus-infected gingival fibroblasts: Implications for periodontal disease. Journal of Oral and Maxillofacial Surgery, Medicine, and Pathology, 37(1), p.188. https://doi.org/10.1016/j.ajoms.2024.07.004.
2. Abdulaziz K. Assaifan, Rawan A. Almansour, Jana A. Alessa, Amir Altinawi, Soliman Alhudaithy. (2025). Impact of Interdigital Electrode Count on Non-Faradaic Electrochemical Biosensor Performance for Detecting Pathogens Associated with Newborn Disabilities. Journal of The Electrochemical Society, 172(5), p.057515. https://doi.org/10.1149/1945-7111/addb85.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2019 Acta Biológica Colombiana

Esta obra está bajo una licencia internacional Creative Commons Atribución-NoComercial-CompartirIgual 4.0.
1. La aceptación de manuscritos por parte de la revista implicará, además de su edición electrónica de acceso abierto bajo licencia Attribution-NonCommercial-ShareAlike 4.0 (CC BY NC SA), la inclusión y difusión del texto completo a través del repositorio institucional de la Universidad Nacional de Colombia y en todas aquellas bases de datos especializadas que el editor considere adecuadas para su indización con miras a incrementar la visibilidad de la revista.
2. Acta Biológica Colombiana permite a los autores archivar, descargar y compartir, la versión final publicada, así como las versiones pre-print y post-print incluyendo un encabezado con la referencia bibliográfica del articulo publicado.
3. Los autores/as podrán adoptar otros acuerdos de licencia no exclusiva de distribución de la versión de la obra publicada (p. ej.: depositarla en un archivo telemático institucional o publicarla en un volumen monográfico) siempre que se indique la publicación inicial en esta revista.
4. Se permite y recomienda a los autores/as difundir su obra a través de Internet (p. ej.: en archivos institucionales, en su página web o en redes sociales cientificas como Academia, Researchgate; Mendelay) lo cual puede producir intercambios interesantes y aumentar las citas de la obra publicada. (Véase El efecto del acceso abierto).



















