Cognición, respuesta electroencefalográfica y su relación con la variabilidad de la frecuencia cardíaca
Cognition, electroencephalographic response and their relation with heart rate variability
DOI:
https://doi.org/10.15446/revfacmed.v65n1.57140Palabras clave:
Sistema nervioso autónomo, Electroencefalografía, Frecuencia cardíaca, Corteza prefrontal (es)Autonomic Nervous System, Electroencephalography, Heart Rate, Prefrontal Cortex (en)
Introducción. La corteza cerebral frontal tiene una mayor actividad teta durante procesos cognitivos de observación y aprendizaje.
Objetivo. Establecer la relación entre actividad electroencefalográfica orbitofrontal y sistema nervioso autónomo en procesos cognitivos.
Materiales y métodos. 20 hombres y 19 mujeres con edad promedio de 21.2 (±2.32) años fueron evaluados mediante electroencefalografía (EGG) FP1-T3, FP2-T4 y electrocardiografía (EKG, del alemán elektrokardiogramm) para determinar frecuencia cardíaca (HR, del inglés heart rate) y variabilidad de la frecuencia cardíaca (HRV, del inglés heart rate variability). La evaluación tuvo cinco fases: reposo, observación, memoria, concentración y juego. Las señales de EGG y EKG fueron analizadas en el dominio de la frecuencia usando la transformada rápida de Fourier (FFT, del inglés fast Fourier transform). Las diferencias por etapa entre las variables se establecieron con el uso de ANOVA de dos vías.
Resultados. Comparado con el reposo, se observó en todas las fases incremento de la actividad teta del EGG (p<0.01), aumento en la baja frecuencia LF (p<0.01) y la HR (p<0.01) y disminución de la alta frecuencia HF (p<0.01). Además, hubo una correlación inversa entre la actividad teta y la potencia de HF (r=-0.86).
Conclusiones. Los datos mostraron una reducción de la actividad parasimpática y un aumento de la actividad simpática asociado a actividad teta de la corteza orbitofrontal, mediante una conexión con el núcleo central de la amígdala.
Introduction: The frontal cortex has a greater theta activity during cognitive observation and learning processes.
Objective: To establish the relation between orbitofrontal electroencephalographic activity and the autonomic nervous system in cognitive processes.
Materials and methods: 20 men and 19 women with a mean age of 21.2 (±2.32) were evaluated by electroencephalography (EGG) FP1-T3, FP2-T4 and electrocardiography (ECG) to determine heart rate (HR) and heart rate variability (HRV). The evaluation was made in five phases during rest, observation, memory, concentration and playing conditions. EGG and ecg signals were analyzed in the frequency domain using the Fast Fourier Transform (FFT). The differences between the variables found during each phase were established through a two-way ANOVA.
Results: When compared to rest conditions, all phases showed an increase of theta activity of the EGG (p <0.01), as well as of the low frequencies LF (p <0.01) and HR (p <0.01), and a decrease in the high frequency HF (P <0.01). In addition, there was an inverse correlation between theta activity and HF power (r=-0.86).
Conclusions: The data showed a reduction in parasympathetic activity and an increased sympathetic activity associated with theta activity in the orbitofrontal cortex, using a connection with the central nucleus of the amygdala.
investigación original
DOI: https://doi.org/10.14556.revfacmed.v65n1.57140
Cognición, respuesta electroencefalográfica y su relación
con la variabilidad de la frecuencia cardíaca
Cognition, electroencephalographic response and their relation with heart rate variability
Recibido: 24/04/2016. Aceptado: 20/06/2016.
Henry Humberto León-Ariza1,2 • Daniel Alfonso Botero-Rosas1,2 • Alveiro Sánchez-Jiménez3 • Jhon Fredy Ramírez-Villada4
Edward Javier Acero-Mondragón1
1 Universidad de La Sabana - Facultad de Medicina - Área de Morfofisiología - Chía - Colombia.
2 Universidad de La Sabana - Facultad de Medicina, Facultad de Ingeniería - Doctorado en Biociencias - Grupo PROSEIM - Chía - Colombia.
3 Universidad Santo Tomás - Sede Bogotá - Facultad de Cultura Física, Deporte y Recreación - Bogotá D.C. - Colombia.
4 Universidad de Antioquia - Instituto Universitario de Educación Física y Deporte - Medellín - Colombia.
Correspondencia: Daniel Alfonso Botero-Rosas. Área de Morfofisiología, Facultad de Medicina, Universidad de La Sabana. Campus del Puente del Común, km 7, autopista Norte de Bogotá, edificio F, oficina de docentes, Teléfono: +57 1 8615555, ext.: 23329. Chía. Colombia. Correo electrónico: daniel.botero@unisabana.edu.co.
| Resumen |
Introducción. La corteza cerebral frontal tiene una mayor actividad teta durante procesos cognitivos de observación y aprendizaje.
Objetivo. Establecer la relación entre actividad electroencefalográfica orbitofrontal y sistema nervioso autónomo en procesos cognitivos.
Materiales y métodos. 20 hombres y 19 mujeres con edad promedio de 21.2 (±2.32) años fueron evaluados mediante electroencefalografía (EGG) FP1-T3, FP2-T4 y electrocardiografía (EKG, del alemán elektrokardiogramm) para determinar frecuencia cardíaca (HR, del inglés heart rate) y variabilidad de la frecuencia cardíaca (HRV, del inglés heart rate variability). La evaluación tuvo cinco fases: reposo, observación, memoria, concentración y juego. Las señales de EGG y EKG fueron analizadas en el dominio de la frecuencia usando la transformada rápida de Fourier (FFT, del inglés fast Fourier transform). Las diferencias por etapa entre las variables se establecieron con el uso de ANOVA de dos vías.
Resultados. Comparado con el reposo, se observó en todas las fases incremento de la actividad teta del EGG (p<0.01), aumento en la baja frecuencia LF (p<0.01) y la HR (p<0.01) y disminución de la alta frecuencia HF (p<0.01). Además, hubo una correlación inversa entre la actividad teta y la potencia de HF (r=-0.86).
Conclusiones. Los datos mostraron una reducción de la actividad parasimpática y un aumento de la actividad simpática asociado a actividad teta de la corteza orbitofrontal, mediante una conexión con el núcleo central de la amígdala.
Palabras clave: Sistema nervioso autónomo; Electroencefalografía; Frecuencia cardíaca; Corteza prefrontal (DeCS).
León-Ariza HH, Botero-Rosas DA, Sánchez-Jiménez A, Ramírez-Villada JF, Acero-Mondragón EJ. Cognición, respuesta electroencefalográfica y su relación con la variabilidad de la frecuencia cardíaca. Rev. Fac. Med. 2017;65(1): 67-72. Spanish. doi:https://doi.org/10.15446.v65n1.57140.
| Abstract |
Introduction: The frontal cortex has a greater theta activity during cognitive observation and learning processes.
Objective: To establish the relation between orbitofrontal electroencephalographic activity and the autonomic nervous system in cognitive processes.
Materials and methods: 20 men and 19 women with a mean age of 21.2 (±2.32) were evaluated by electroencephalography (EGG) FP1-T3, FP2-T4 and electrocardiography (ECG) to determine heart rate (HR) and heart rate variability (HRV). The evaluation was made in five phases during rest, observation, memory, concentration and playing conditions. EGG and ecg signals were analyzed in the frequency domain using the Fast Fourier Transform (FFT). The differences between the variables found during each phase were established through a two-way ANOVA.
Results: When compared to rest conditions, all phases showed an increase of theta activity of the EGG (p <0.01), as well as of the low frequencies LF (p <0.01) and HR (p <0.01), and a decrease in the high frequency HF (P <0.01). In addition, there was an inverse correlation between theta activity and HF power (r=-0.86).
Conclusions: The data showed a reduction in parasympathetic activity and an increased sympathetic activity associated with theta activity in the orbitofrontal cortex, using a connection with the central nucleus of the amygdala.
Keywords: Autonomic Nervous System; Electroencephalography; Heart Rate; Prefrontal Cortex (MeSH).
León-Ariza HH, Botero-Rosas DA, Sánchez-Jiménez A, Ramírez-Villada JF, Acero-Mondragón EJ. [Cognition, electroencephalographic response and their relation with heart rate variability]. Rev. Fac. Med. 2017;65(1): 67-72. Spanish. doi:https://doi.org/10.15446.v65n1.57140.
Introducción
La electroencefalografía (EGG) es una técnica diagnóstica que permite evaluar la actividad eléctrica de las estructuras cerebrales con el uso de electrodos colocados sobre el cuero cabelludo. La potencia obtenida se clasifica de acuerdo a bandas de frecuencia, que pueden ser: beta (>13 Hz), alfa (8-13 Hz), teta (4-8 Hz) y delta (0.5-4 Hz). En adultos jóvenes en condiciones de reposo, relajación, vigilia y sin actividad mental, la actividad predominante es beta y alfa, mientras que, en ocasiones, la presencia de teta se relaciona a condiciones patológicas sin origen o significado evidente (1).
Diversos estudios electrofisiológicos han demostrado que se incrementa la actividad teta durante procesos de aprendizaje en áreas del cerebro como la corteza frontal (2). Esto se manifiesta en la transición del reposo hacia la observación y coincide con las respuestas de uso de memoria episódica (3), en especial cuando se utiliza memoria de trabajo (4,5).
De otra parte, la variabilidad de la frecuencia cardíaca (HRV, del inglés heart rate variability) ha sido muy usada para evaluar múltiples condiciones tanto fisiológicas como patológicas, por la reacción directa de la actividad del sistema nervioso autónomo a múltiples estímulos externos (6). Estudios previos han demostrado que el estrés mental (evaluado a través de pruebas de concentración) lleva a una disminución de la HRV, lo que sugiere un aumento de la actividad simpática y una reducción de la actividad parasimpática (7).
Diversos autores han relacionado la actividad de la corteza frontal (en especial la corteza frontal derecha) con el proceso de control de la actividad autonómica y, por lo tanto, con la modulación de la HRV (8). Sin embargo, existen pocos trabajos que vinculen la actividad electroencefalográfica propia de los procesos de aprendizaje con cambios autonómicos.
Este trabajo busca valorar la relación entre la actividad neuronal cortical orbitofrontal, determinada a través de EGG, y la respuesta autonómica, determinada a través de HRV, frente a tareas cognitivas observacionales, de concentración, memoria y juego.
Materiales y métodos
Tipo de estudio
El presente es un estudio transversal descriptivo (cross sectional study) con dos grupos (hombres n=20 y mujeres n=19) con promedio de edad de 21.2 (±2.32) años. Los criterios de inclusión fueron: individuos entre 18 y 24 años, físicamente activos durante los últimos 12 meses, con una práctica deportiva de mínimo 4 días por semana o mayor a 250 minutos semanales y sin consumo previo de sustancias que afecten el sistema nervioso (cafeína, alcohol, tabaco). Incluso con los criterios descritos, los participantes fueron excluidos si, al analizar las señales biológicas, estas se encontraban contaminadas con ruido excesivo que impidiera realizar su limpieza, así como alguna condición limitante observada en la valoración clínica previa que pudiera afectar la respuesta autonómica (privación de sueño, estrés físico asociado a dolor o lesiones, entre otras).
El estudio y sus procedimientos fueron ajustados a los principios establecidos en la declaración de Helsinki. Para esto, los protocolos y procedimientos fueron revisados y autorizados por el Comité Bioético de las Instituciones que particparon en la investigación. En consecuencia, todos los individuos fueron informados de las características del estudio, sus objetivos, los procedimientos a aplicar, los riesgos potenciales y el uso de la información, a fin de que aprobaran su participación con su firma y huella. Para garantizar la integridad de los pacientes, fue necesaria una valoración clínica inicial por parte del personal biomédico del estudio, en la cual se evaluaron las condiciones de elegibilidad y el control de potenciales riesgos para la salud.
Muestreo
Considerando los criterios descritos y la complejidad de los procedimientos del laboratorio, se realizó un muestreo no probabilístico y una agrupación de los participantes por sexo. Por tanto, un total de 40 individuos de ambos géneros fueron incluidos en el estudio, quienes se dividieron en dos grupos (hombres n=20; mujeres n=20). Al final del proceso, se estableció una pérdida muestral del 2.5% (una mujer), explicada por la baja calidad de las señales adquiridas.
Descripción de los procedimientos
Con la intención de controlar fuentes de invalidación relacionadas con las condiciones de aplicación de protocolos e instrumentos, se determinó que los participantes fueran evaluados en horas de la mañana e interrogados sobre la calidad del sueño y el uso de cafeína, alcohol o cigarrillo durante las 24 horas previas. Por tanto, aquellos individuos que incumplían alguna de estas condiciones fueron programados de nuevo en otra sesión, de manera que se garantizaran unas condiciones mínimas para la toma y el registro de las variables.
Adquisición de señales
La adquisición de las variables se realizó con PowerLab 8/35 (ADInstruments Europe, Oxford, UK), la señal electroencefalográfica se obtuvo mediante electrodos de oro plateado dispuestos en posición frontopolar izquierda FP1, frontopolar derecha FP2, temporal izquierda T3 y temporal derecha T4 y un electrodo de referencia a nivel auricular A1, para un montaje longitudinal FP1-T3 y FP2-T4, según el sistema internacional 10/20. Para la adquisición, se utilizó una ganancia x106 y un filtro digital pasa baja con frecuencia de corte 35 Hz y frecuencia de muestreo de 400 Hz.
La señal electrocardiográfica se obtuvo por medio de una derivación DII (60°), al ubicar un electrodo negativo en el acromion derecho, un electrodo positivo en la espina ilíaca anterosuperior izquierda y un electrodo neutro en la espina ilíaca anterosuperior derecha. Para la adquisición, se utilizó una ganancia x103 y un filtro digital pasa baja de 30 Hz a una tasa de muestreo de 400 Hz.
Fases del procedimiento
Cada uno de los participantes cumplió con cinco fases seguidas durante el estudio: a) reposo: se recolectaron las señales en reposo con el paciente en posición sedente durante dos minutos; b) observación: se le permitió al evaluado observar una serie de imágenes con su respectivo nombre durante 30 segundos (Figura 1A); c) memoria: el evaluado tuvo un tiempo de dos minutos para evocar el nombre del mayor número de imágenes; d) concentración: el evaluado resolvió el Test de Harris & Harris para concentración durante tres minutos (9), que consiste en una cuadrícula con números aleatorios entre el 0 y el 99 (Figura 1B), y e) juego: los participantes del estudio jugaron “reto mental” durante diez minutos. Este juego reúne pruebas de memoria, habilidad visual, lógica, matemáticas y atención.


Figura 1. A) Figuras utilizadas para la evaluación de la observación y la memoria; B) Test de Harris & Harris utilizado para evaluar la concentración (9).
Fuente: Elaboración propia.
Análisis de las señales
Para el análisis de la señal electroencefalográfica, se utilizó Matlab (MathWorks) y luego de seleccionar un período de tiempo libre de artefactos, se extrajo la serie temporal (ST) y se obtuvo la densidad de potencia espectral (DPE) mediante la transformada rápida de Fourier (FFT, del inglés fast Fourier transform). Después, se calculó la energía en cada banda del EGG (beta, alfa, delta y teta) empleando el método trapezoidal. Y al final, se determinó el peso porcentual de energía que aporta cada ritmo a la energía total del EGG.
El análisis de la HRV se llevó a cabo con ayuda del software Kubios HRV (10). La fase de preprocesamiento de la señal de frecuencia cardíaca (HR, del inglés heart rate) consistió en el retiro de artefactos (variaciones del intervalo rr superiores a 0.45 s con respecto al promedio) y el filtrado de la señal con un filtro pasa alta tipo Smoothness Prior, con un valor lambda de 500 y una frecuencia de corte de 0.035 Hz (11).
A partir del tacograma obtenido libre de ruido, fue analizado el promedio de HR en la ST. Para el análisis en el dominio de la frecuencia a partir de la ST, se requirió interpolar spline cubic, ya que los latidos cardíacos no eran equidistantes. Finalmente, una tasa de 4 Hz con muestras equidistantes fue utilizada, a continuación se aplicó una FFT para obtener la DPE y los parámetros de potencia fueron obtenidos (en valores RMS y porcentual del total de la potencia) en la muy baja frecuencia (VLF: 0-0.04 Hz), baja frecuencia (LF: 0.04-0.15 Hz) y alta frecuencia (HF: 0.15-0.4 Hz).
Análisis estadístico
La homogeneidad en el comportamiento de las variables se evaluó con el estadístico de Levene. Las diferencias entre cada una de las fases del estudio se determinaron mediante el análisis de la varianza (ANOVA) de dos vías, seguido de comparaciones post hoc de diferencias mínimas significativas (DMS). Se usó una t-Student para muestras no pareadas que mostraran las diferencias entre hombres y mujeres. Las conexiones entre las variables se establecieron mediante el coeficiente de correlación de Pearson. La diferencia se consideró estadísticamente significativa con una p<0.05 y altamente significativa con p<0.01.
Resultados
No se encontraron diferencias estadísticamente significativas en la distribución de la actividad electroencefalográfica entre hombres y mujeres, si bien en ambos grupos predominó beta a lo largo de toda la evaluación (Figura 2A). De la misma forma, tampoco se observaron diferencias en las variables asociadas a HRV y la HR. Aunque la HR fue superior siempre en mujeres, solo tuvo una diferencia significativa en la fase de memoria (p<0.01) y juego (p=0.02) (Figura 2B).

Figura 2. Respuestas encontradas entre los datos de hombres y mujeres. A) actividad electroencefalográfica. δ - delta 0.5-4 Hz, θ - teta 4-8 Hz, α = alfa 8-13 Hz y β - beta >13 Hz; B) diferencia entre los datos de frecuencia cardíaca (HR) y HRV.
VJF: muy baja frecuencia 0-0.04 Hz; LF: baja frecuencia 0.04-0.15 Hz; HF: alta frecuencia 0.15-0.4 Hz.
*Diferencia estadísticamente significativa p<0.05.
Fuente: Elaboración propia.
El análisis de la varianza mostró diferencias significativas entre las fases de la evaluación para el comportamiento de actividad teta. Para beta, las diferencias se observaron al comparar cada fase con el reposo. A su vez, la HR y la HRV mostraron diferencias significativas en la mayoría de las variables (Tabla 1).
Tabla 1. Resultados de ANOVA para la evaluación de diferencias entre las variables.
|
ANOVA |
Observación |
Memoria |
Concentración |
Juego |
|
|
EEG-Teta |
0.00 † |
0.00 † |
0.00 † |
NS |
0.00 † |
|
EEG-Beta |
NS |
0.02 * |
0.01 * |
0.03 * |
0.02 * |
|
HR |
0.00 † |
0.00 † |
0.00 † |
0.00 † |
0.01 * |
|
HRV-VLF% |
0.00 † |
NS |
0.00 † |
0.00 † |
0.00 † |
|
HRV-LF% |
0.00 † |
0.00 † |
0.01 * |
NS |
NS |
|
HRV-HF% |
0.00 † |
0.00 † |
0.00 † |
0.00 † |
0.00 † |
|
LF/HF |
0.00 † |
0.00 † |
NS |
NS |
NS |
Prueba de diferencias mínimas y su significativas entre las variables comparada con el reposo. HR: frecuencia cardíaca; HRV: variabilidad de la frecuencia cardíaca; VJF: muy baja frecuencia; LF: baja frecuencia; HF: alta frecuencia ; NS: no significativo.
* p<0.05.
† p<0.01.
Fuente: Elaboración propia.
Comparadas con la señal obtenida en reposo, se observa que las variables electroencefalográficas con cambios estadísticamente significativos fueron el porcentaje de actividad teta, que se incrementó (p<0.01). Además, la actividad beta se redujo, delta no presentó variación y el aumento para alfa solo ocurrió en la fase de concentración de los hombres, donde pasó de 13.2% a 16.3% (p=0.03). (Tabla 1, Figura 1A).
Las correlaciones entre cambios electroencefalográficos y HRV vincularon el aumento de la actividad teta con una disminución del porcentaje de señal HF (r=-0.86) y un aumento de la HR (r=0.71). Del mismo modo, se asoció la reducción de beta a una reducción del porcentaje HF (r=0.96) y a un incremento de la HR (r=-0.75) (Tabla 2).
Tabla 2. Correlaciones entre los datos electroencefalográficos y datos de variabilidad de la frecuencia cardíaca.
|
Teta 4 - 8 Hz |
Beta < 13 Hz |
|||||
|
Hombres |
Mujeres |
Todos |
Hombres |
Mujeres |
Todos |
|
|
HR |
0.50 |
0.73 |
0.71 |
-0.69 |
-0.57 |
-0.75 |
|
VLF% |
0.57 |
0.24 |
0.63 |
-0.59 |
-0.39 |
-0.68 |
|
LF% |
-0.24 |
0.65 |
0.03 |
0.13 |
-0.46 |
-0.06 |
|
HF% |
-0.69 |
-0.71 |
-0.86 |
0.80 |
0.76 |
0.96 |
|
LF/HF |
0.26 |
0.70 |
0.47 |
-0.39 |
-0.45 |
-0.45 |
HR: frecuencia cardíaca; HRV: variabilidad de la frecuencia cardíaca; VLF: muy baja frecuencia; LF: baja frecuencia; HF: alta frecuencia.
Fuente: Elaboración propia.
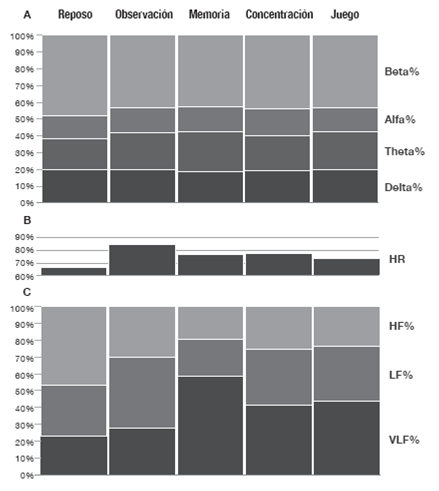
Los cambios en conjunto de la distribución de la actividad en el EGG, la variación de la HR y la distribución de la HRV se pueden observar en la Figura 3.

Figura 3. Respuestas fisiológicas observadas durante cada fase del estudio, promedio para todos los individuos n=39. A) Comportamiento de la actividad electroencefalográfica; B) HR = frecuencia cardíaca promedio; C) Variabilidad de la frecuencia cardíaca expresada en porcentaje: VLF = muy baja frecuencia, LF = baja frecuencia y HF = alta frecuencia.
Fuente: Elaboración propia.
Discusión
El presente estudio reveló que el comportamiento de la HR en todo el grupo fue superior durante el período de observación, memoria, concentración y juego, en comparación con el reposo, la cual es una respuesta que puede vincularse en primer lugar con una inactivación autonómica parasimpática que se manifestó en la reducción de la señal HF de la HRV (6). Estos resultados fueron similares a los reportados en otras situaciones como la transición desde el reposo hacia la actividad física, que tuvieron un aumento de la HR explicado por supresión vagal en especial (12).
Se han encontrado respuestas autonómicas en diversas formas de estrés psicosocial similares a las reportadas en este estudio (13). Se presume que la presión evolutiva ha impulsado adaptaciones funcionales para centrar la atención en la detección de amenazas, basadas en la información que provee la vía visual sin mediación inicial de la conciencia para facilitar respuestas fisiológicas autonómicas (14). Esta situación se parece a la observada en actividades que requieren memoria a corto plazo, en las que la actividad simpática ha aumentado (15).
Experimentos en ratas han revelado un crecimento de ondas teta ante un estímulo nuevo asociado al proceso de aprendizaje (2). Así mismo ocurre durante procesos de atención, en los que se reducen los ritmos corticales beta y gamma precedentes a un incremento de teta (16). Estos hallazgos son semejantes a los encontrados en el presente estudio, donde se evidenció una reducción de ritmo beta y aumento de teta durante la transición reposo-observación.
Los hallazgos proponen que los estímulos visuales que evocan procesos cognitivos de aprendizaje generan estímulos en la corteza orbitofrontal, que se encarga de procesos de asociación e involucra contexto, ubicación espacial, eventos, respuestas emocionales y toma de decisiones (17), entre otros. La participación de esta región es evidenciable por una mayor actividad teta (18). Desde la corteza prefontal se proyectan neuronas inhibitorias (GABAérgicas) hacia el núcleo central de la amígdala (CeA), lo que se asocia al desarrollo de emociones (19).
Del mismo modo, está demostrado que desde el CeA, se proyectan axones glutaminérgicos hacia la médula ventrolateral rostral desde donde se proyectan fibras excitatorias simpáticas preganglionares vía retículo espinal (20). Por otra parte, desde el CeA también se proyectan fibras inhibitorias GABAérgicas hacia el núcleo del tracto solitario, lo que podría conectarse con una reducción de la actividad parasimpática (21).
En general, en los procesos de comprensión de la regulación de la actividad autonómica cardiovascular, se tiene en cuenta el papel de los barorreceptores (22,23) y los quimiorreceptores (24). Sin embargo, en la actualidad, la adecuada comprensión requiere considerar de forma adicional el efecto mecánico del sistema respiratorio (25), el efecto de mecanorreceptores musculares (26), la presencia de citoquinas proinflamatorias por acción directa sobre el hipotálamo (27,28) y el efecto de la actividad cortical descrito hasta ahora en condiciones de sueño (29) y, como lo evidencian los resultados expuestos, también la actividad cortical en vigilia cuando hay transición del estado de reposo hacia demandas cognitivas observacionales de concentración, memoria y juego.
Con base en la evidencia, se ha sugerido el núcleo central de la amígdala como posible estructura fundamental en la relación entre la corteza cerebral y el sistema nervioso autónomo. No obstante, es necesario desarrollar nuevos estudios para comprobar esto.
Conclusiones
Los resultados en este estudio sugieren cambios en la respuesta electroencefalográfica de la corteza orbitofrontal, dados por una mayor actividad teta y asociados a una mayor actividad del sistema nervioso simpático y una menor respuesta del sistema nervioso parasimpático. Esto plantea que, adicional a moduladores clásicos como los barorreceptores y los quimiorreceptores, la corteza cerebral participa de forma activa en la regulación de la actividad autonómica durante la vigilia. Este fenómeno es evidente cuando se pasa del estado de reposo a una demanda cognitiva observacional, de concentración, memoria y juego.
Conflicto de intereses
Ninguno declarada por los autores.
Financiación
Ninguna declarada por los autores.
Agradecimientos
Al Laboratorio de Fisiología de la Universidad de La Sabana, que aportó los equipos para el desarrollo de la investigación, aunque no participó en el diseño, análisis o interpretación de datos, ni en la escritura, revisión o preparación del manuscrito. Además, agradecen a los estudiantes que participaron en la adquisición y tabulación de los datos y al ingeniero Oscar Mosquera por su apoyo en el procesamiento de los datos.
Referencias
1.Schomer DL, Lopez da Silva FH. Niedermeyer’s Electroencephalography: Basic Principles, Clinical Applications, and Related Fields. 6th ed. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins; 2011.
2.Buzsáki G, Moser EI. Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nat Neurosci. 2013;16(2):130-8. http://doi.org/f4j6d2.
3.Suthana N, Haneef Z, Stern J, Mukamel R, Behnke E, Knowlton B, et al. Memory enhancement and deep-brain stimulation of the entorhinal area. N Engl J Med. 2012;366(6):502-10. http://doi.org/b4d3.
4.Gärtner M, Grimm S, Bajbouj M. Frontal midline theta oscillations during mental arithmetic: effects of stress. Front Behav Neurosci. 2015;9:96. http://doi.org/b4d4.
5.Gärtner M, Rohde-Liebenau L, Grimm S, Bajbouj M. Working memory-related frontal theta activity is decreased under acute stress. Psychoneuroendocrinology. 2014;43:105-13. http://doi.org/f5zzk5.
6.Kleiger RE, Stein PK, Bigger JT Jr. Heart rate variability: measurement and clinical utility. Ann Noninvasive Electrocardiol. 2005;10(1):88-101. http://doi.org/dj82kt.
7.Taelman J, Vandeput S, Spaepen A, Van Huffel S. Influence of Mental Stress on Heart Rate and Heart Rate Variability. In: Vander Sloten J, Verdonck P, Nyssen M, Haueisen J, editors. 4th European Conference of the International Federation for Medical and Biological Engineering. 22nd ed. Springer Berlin Heidelberg; 2009. p. 1366-9.
8.Thayer JF, Hansen AL, Saus-Rose E, Johnsen BH. Heart rate variability, prefrontal neural function, and cognitive performance: the neurovisceral integration perspective on self-regulation, adaptation, and health. Ann Behav Med. 2009;37(2):141-53. http://doi.org/czs6k3.
9.Harris DV, Harris BL. The athlete’s guide to sports psychology: Mental skills for physical people. New York: Leisure Press; 1984.
10.Tarvainen MP, Niskanen JP, Lipponen JA, Ranta-Aho PO, Karjalainen PA. Kubios HRV--heart rate variability analysis software. Comput Methods Programs Biomed. 2014;113(1):210-20. http://doi.org/b4d6.
11.Tarvainen MP, Ranta-Aho PO, Karjalainen PA. An advanced detrending method with application to HRV analysis. IEEE Trans Biomed Eng. 2002;49(2):172-5. http://doi.org/cfc247.
12.Beltrame T, Karsten M, Chacon-Mikahil MPT, Madruga VA, Silva E, Borghi-Silva A, et al. Age influence on the heart rate behavior on the rest-exercise transition: an analysis by deltas and linear regression. Rev Bras Med Esporte. 2012;18(5):300-4. http://doi.org/b4d7.
13.Petrowski K, Herold U, Joraschky P, Mück-Weymann M, Siepmann M. The effects of psychosocial stress on heart rate variability in Panic Disorder. German J Psychiatry. 2010;13(2):66-73.
14.Hedger N, Adams WJ, Garner M. Autonomic arousal and attentional orienting to visual threat are predicted by awareness. J Exp Psychol Hum Percept Perform. 2015;41(3):798-806. http://doi.org/f7fngz.
15.Sara SJ. The locus coeruleus and noradrenergic modulation of cognition. Nat Rev Neurosci. 2009;10(3):211-23. http://doi.org/cx34qb.
16.Brown RA, Walling SG, Milway JS, Harley CW. Locus ceruleus activation suppresses feedforward interneurons and reduces beta-gamma electroencephalogram frequencies while it enhances theta frequencies in rat dentate gyrus. J Neurosci. 2005;25(8):1985-91. http://doi.org/ffvrk9.
17.Euston DR, Gruber AJ, McNaughton BL. The role of medial prefrontal cortex in memory and decision making. Neuron. 2012;76(6):1057-70. http://doi.org/f4jhv4.
18.Hsieh LT, Ranganath C. Frontal midline theta oscillations during working memory maintenance and episodic encoding and retrieval. Neuroimage. 2014;85 Pt 2:721-9. http://doi.org/f5mhk8.
19.Barbas H, Saha S, Rempel-Clower N, Ghashghaei T. Serial pathways from primate prefrontal cortex to autonomic areas may influence emotional expression. BMC Neurosci. 2003;4:25. http://doi.org/dncqg7.
20.Chapp AD, Gui L, Huber MJ, Liu J, Larson RA, Zhu J, et al. Sympathoexcitation and pressor responses induced by ethanol in the central nucleus of amygdala involves activation of NMDA receptors in rats. Am J Physiol Heart Circ Physiol. 2014;307(5):H701-9. http://doi.org/f6fsj4.
21.Saha S. Role of the central nucleus of the amygdala in the control of blood pressure: descending pathways to medullary cardiovascular nuclei. Clin Exp Pharmacol Physiol. 2005;32(5-6):450-6. http://doi:.org/dhnkhw.
22.Kumagai H, Oshima N, Matsuura T, Iigaya K, Imai M, Onimaru H, et al. Importance of rostral ventrolateral medulla neurons in determining efferent sympathetic nerve activity and blood pressure. Hypertens Res. 2012;35(2):132-41. http://doi.org/d7x3c2.
23.Albaghdadi M. Baroreflex control of long-term arterial pressure. Rev Bras Hipertens. 2007;14(4):212-25.
24.Moreira TS, Takakura AC, Damasceno RS, Falquetto B, Totola LT, Sobrinho CR, et al. Central chemoreceptors and neural mechanisms of cardiorespiratory control. Braz J Med Biol Res. 2011;44(9):883-9. http://doi.org/dtn9hd.
25.Yasuma F, Hayano J. Respiratory sinus arrhythmia: why does the heartbeat synchronize with respiratory rhythm? Chest. 2004;125(2):683-90. http://doi.org/dtd8gm.
26.Gladwell VF, Fletcher J, Patel N, Elvidge LJ, Lloyd D, Chowdhary S, et al. The influence of small fibre muscle mechanoreceptors on the cardiac vagus in humans. J Physiol. 2005;567(2):713-21. http://doi.org/chjk2f.
27.Sajadieh A, Nielsen OW, Rasmussen V, Hein HO, Abedini S, Hansen JF. Increased heart rate and reduced heart-rate variability are associated with subclinical inflammation in middle-aged and elderly subjects with no apparent heart disease. Eur Heart J. 2004;25(5):363-70. http://doi.org/dd939s.
28.Marsland AL, Gianaros PJ, Prather AA, Jennings JR, Neumann SA, Manuck SB. Stimulated production of proinflammatory cytokines covaries inversely with heart rate variability. Psychosom Med. 2007;69(8):709-16. http://doi.org/ffns2t.
29.Kuo TB, Chen CY, Hsu YC, Yang CC. EEG beta power and heart rate variability describe the association between cortical and autonomic arousals across sleep. Auton Neurosci. 2016;194:32-7. http://doi.org/f776kf.
Recibido: 24 de abril de 2016; Aceptado: 20 de junio de 2016
Resumen
Introducción.
La corteza cerebral frontal tiene una mayor actividad teta durante procesos cognitivos de observación y aprendizaje.
Objetivo.
Establecer la relación entre actividad electroencefalográfica orbitofrontal y sistema nervioso autónomo en procesos cognitivos.
Materiales y métodos.
20 hombres y 19 mujeres con edad promedio de 21.2 (±2.32) años fueron evaluados mediante electroencefalografía (EGG) FP1-T3, FP2-T4 y electrocardiografía (EKG, del alemán elektrokardiogramm) para determinar frecuencia cardíaca (HR, del inglés heart rate) y variabilidad de la frecuencia cardíaca (HRV, del inglés heart rate variability). La evaluación tuvo cinco fases: reposo, observación, memoria, concentración y juego. Las señales de EGG y EKG fueron analizadas en el dominio de la frecuencia usando la transformada rápida de Fourier (FFT, del inglés fast Fourier transform). Las diferencias por etapa entre las variables se establecieron con el uso de ANOVA de dos vías.
Resultados.
Comparado con el reposo, se observó en todas las fases incremento de la actividad teta del EGG (p<0.01), aumento en la baja frecuencia LF (p<0.01) y la HR (p<0.01) y disminución de la alta frecuencia HF (p<0.01). Además, hubo una correlación inversa entre la actividad teta y la potencia de HF (r=-0.86).
Conclusiones.
Los datos mostraron una reducción de la actividad parasimpática y un aumento de la actividad simpática asociado a actividad teta de la corteza orbitofrontal, mediante una conexión con el núcleo central de la amígdala.
Palabras clave:
Sistema nervioso autónomo, Electroencefalografía, Frecuencia cardíaca, Corteza prefrontal (DeCS)..Abstract
Introduction:
The frontal cortex has a greater theta activity during cognitive observation and learning processes.
Objective:
To establish the relation between orbitofrontal electroencephalographic activity and the autonomic nervous system in cognitive processes.
Materials and methods:
20 men and 19 women with a mean age of 21.2 (±2.32) were evaluated by electroencephalography (EGG) FP1-T3, FP2-T4 and electrocardiography (ECG) to determine heart rate (HR) and heart rate variability (HRV). The evaluation was made in five phases during rest, observation, memory, concentration and playing conditions. EGG and ECG signals were analyzed in the frequency domain using the Fast Fourier Transform (FFT). The differences between the variables found during each phase were established through a two-way ANOVA.
Results:
When compared to rest conditions, all phases showed an increase of theta activity of the EGG (p <0.01), as well as of the low frequencies LF (p <0.01) and HR (p <0.01), and a decrease in the high frequency HF (P <0.01). In addition, there was an inverse correlation between theta activity and HF power (r=-0.86).
Conclusions:
The data showed a reduction in parasympathetic activity and an increased sympathetic activity associated with theta activity in the orbitofrontal cortex, using a connection with the central nucleus of the amygdala.
Keywords:
Autonomic Nervous System, Electroencephalography, Heart Rate, Prefrontal Cortex (MeSH)..Introducción
La electroencefalografía (EGG) es una técnica diagnóstica que permite evaluar la actividad eléctrica de las estructuras cerebrales con el uso de electrodos colocados sobre el cuero cabelludo. La potencia obtenida se clasifica de acuerdo a bandas de frecuencia, que pueden ser: beta (>13 Hz), alfa (8-13 Hz), teta (4-8 Hz) y delta (0.5-4 Hz). En adultos jóvenes en condiciones de reposo, relajación, vigilia y sin actividad mental, la actividad predominante es beta y alfa, mientras que, en ocasiones, la presencia de teta se relaciona a condiciones patológicas sin origen o significado evidente 1.
Diversos estudios electrofisiológicos han demostrado que se incrementa la actividad teta durante procesos de aprendizaje en áreas del cerebro como la corteza frontal 2. Esto se manifiesta en la transición del reposo hacia la observación y coincide con las respuestas de uso de memoria episódica 3, en especial cuando se utiliza memoria de trabajo 4,5.
De otra parte, la variabilidad de la frecuencia cardíaca (HRV, del inglés heart rate variability) ha sido muy usada para evaluar múltiples condiciones tanto fisiológicas como patológicas, por la reacción directa de la actividad del sistema nervioso autónomo a múltiples estímulos externos 6. Estudios previos han demostrado que el estrés mental (evaluado a través de pruebas de concentración) lleva a una disminución de la HRV, lo que sugiere un aumento de la actividad simpática y una reducción de la actividad parasimpática 7.
Diversos autores han relacionado la actividad de la corteza frontal (en especial la corteza frontal derecha) con el proceso de control de la actividad autonómica y, por lo tanto, con la modulación de la HRV 8. Sin embargo, existen pocos trabajos que vinculen la actividad electroencefalográfica propia de los procesos de aprendizaje con cambios autonómicos.
Este trabajo busca valorar la relación entre la actividad neuronal cortical orbitofrontal, determinada a través de EGG, y la respuesta autonómica, determinada a través de HRV, frente a tareas cognitivas observacionales, de concentración, memoria y juego.
Materiales y métodos
Tipo de estudio
El presente es un estudio transversal descriptivo (cross sectional study) con dos grupos (hombres n=20 y mujeres n=19) con promedio de edad de 21.2 (±2.32) años. Los criterios de inclusión fueron: individuos entre 18 y 24 años, físicamente activos durante los últimos 12 meses, con una práctica deportiva de mínimo 4 días por semana o mayor a 250 minutos semanales y sin consumo previo de sustancias que afecten el sistema nervioso (cafeína, alcohol, tabaco). Incluso con los criterios descritos, los participantes fueron excluidos si, al analizar las señales biológicas, estas se encontraban contaminadas con ruido excesivo que impidiera realizar su limpieza, así como alguna condición limitante observada en la valoración clínica previa que pudiera afectar la respuesta autonómica (privación de sueño, estrés físico asociado a dolor o lesiones, entre otras).
El estudio y sus procedimientos fueron ajustados a los principios establecidos en la declaración de Helsinki. Para esto, los protocolos y procedimientos fueron revisados y autorizados por el Comité Bioético de las Instituciones que particparon en la investigación. En consecuencia, todos los individuos fueron informados de las características del estudio, sus objetivos, los procedimientos a aplicar, los riesgos potenciales y el uso de la información, a fin de que aprobaran su participación con su firma y huella. Para garantizar la integridad de los pacientes, fue necesaria una valoración clínica inicial por parte del personal biomédico del estudio, en la cual se evaluaron las condiciones de elegibilidad y el control de potenciales riesgos para la salud.
Muestreo
Considerando los criterios descritos y la complejidad de los procedimientos del laboratorio, se realizó un muestreo no probabilístico y una agrupación de los participantes por sexo. Por tanto, un total de 40 individuos de ambos géneros fueron incluidos en el estudio, quienes se dividieron en dos grupos (hombres n=20; mujeres n=20). Al final del proceso, se estableció una pérdida muestral del 2.5% (una mujer), explicada por la baja calidad de las señales adquiridas.
Descripción de los procedimientos
Con la intención de controlar fuentes de invalidación relacionadas con las condiciones de aplicación de protocolos e instrumentos, se determinó que los participantes fueran evaluados en horas de la mañana e interrogados sobre la calidad del sueño y el uso de cafeína, alcohol o cigarrillo durante las 24 horas previas. Por tanto, aquellos individuos que incumplían alguna de estas condiciones fueron programados de nuevo en otra sesión, de manera que se garantizaran unas condiciones mínimas para la toma y el registro de las variables.
Adquisición de señales
La adquisición de las variables se realizó con PowerLab 8/35 (ADInstruments Europe, Oxford, UK), la señal electroencefalográfica se obtuvo mediante electrodos de oro plateado dispuestos en posición frontopolar izquierda FP1, frontopolar derecha FP2, temporal izquierda T3 y temporal derecha T4 y un electrodo de referencia a nivel auricular A1, para un montaje longitudinal FP1-T3 y FP2-T4, según el sistema internacional 10/20. Para la adquisición, se utilizó una ganancia x106 y un filtro digital pasa baja con frecuencia de corte 35 Hz y frecuencia de muestreo de 400 Hz.
La señal electrocardiográfica se obtuvo por medio de una derivación DII (60°), al ubicar un electrodo negativo en el acromion derecho, un electrodo positivo en la espina ilíaca anterosuperior izquierda y un electrodo neutro en la espina ilíaca anterosuperior derecha. Para la adquisición, se utilizó una ganancia x103 y un filtro digital pasa baja de 30 Hz a una tasa de muestreo de 400 Hz.
Fases del procedimiento
Cada uno de los participantes cumplió con cinco fases seguidas durante el estudio: a) reposo: se recolectaron las señales en reposo con el paciente en posición sedente durante dos minutos; b) observación: se le permitió al evaluado observar una serie de imágenes con su respectivo nombre durante 30 segundos (Figura 1A); c) memoria: el evaluado tuvo un tiempo de dos minutos para evocar el nombre del mayor número de imágenes; d) concentración: el evaluado resolvió el Test de Harris & Harris para concentración durante tres minutos 9, que consiste en una cuadrícula con números aleatorios entre el 0 y el 99 (Figura 1B), y e) juego: los participantes del estudio jugaron "reto mental" durante diez minutos. Este juego reúne pruebas de memoria, habilidad visual, lógica, matemáticas y atención.
Figura 1: A) Figuras utilizadas para la evaluación de la observación y la memoria; B) Test de Harris & Harris utilizado para evaluar la concentración 9.
Análisis de las señales
Para el análisis de la señal electroencefalográfica, se utilizó Matlab (MathWorks) y luego de seleccionar un período de tiempo libre de artefactos, se extrajo la serie temporal (ST) y se obtuvo la densidad de potencia espectral (DPE) mediante la transformada rápida de Fourier (FFT, del inglés fast Fourier transform). Después, se calculó la energía en cada banda del EGG (beta, alfa, delta y teta) empleando el método trapezoidal. Y al final, se determinó el peso porcentual de energía que aporta cada ritmo a la energía total del EGG.
El análisis de la HRV se llevó a cabo con ayuda del software Kubios HRV (10). La fase de preprocesamiento de la señal de frecuencia cardíaca (HR, del inglés heart rate) consistió en el retiro de artefactos (variaciones del intervalo RR superiores a 0.45 s con respecto al promedio) y el filtrado de la señal con un filtro pasa alta tipo Smoothness Prior, con un valor lambda de 500 y una frecuencia de corte de 0.035 Hz 11.
A partir del tacograma obtenido libre de ruido, fue analizado el promedio de HR en la ST. Para el análisis en el dominio de la frecuencia a partir de la ST, se requirió interpolar spline cubic, ya que los latidos cardíacos no eran equidistantes. Finalmente, una tasa de 4 Hz con muestras equidistantes fue utilizada, a continuación se aplicó una FFT para obtener la DPE y los parámetros de potencia fueron obtenidos (en valores RMS y porcentual del total de la potencia) en la muy baja frecuencia (VLF: 0-0.04 Hz), baja frecuencia (LF: 0.04-0.15 Hz) y alta frecuencia (HF: 0.15-0.4 Hz).
Análisis estadístico
La homogeneidad en el comportamiento de las variables se evaluó con el estadístico de Levene. Las diferencias entre cada una de las fases del estudio se determinaron mediante el análisis de la varianza (ANOVA) de dos vías, seguido de comparaciones post hoc de diferencias mínimas significativas (DMS). Se usó una t-Student para muestras no pareadas que mostraran las diferencias entre hombres y mujeres. Las conexiones entre las variables se establecieron mediante el coeficiente de correlación de Pearson. La diferencia se consideró estadísticamente significativa con una p<0.05 y altamente significativa con p<0.01.
Resultados
No se encontraron diferencias estadísticamente significativas en la distribución de la actividad electroencefalográfica entre hombres y mujeres, si bien en ambos grupos predominó beta a lo largo de toda la evaluación (Figura 2A). De la misma forma, tampoco se observaron diferencias en las variables asociadas a HRV y la HR. Aunque la HR fue superior siempre en mujeres, solo tuvo una diferencia significativa en la fase de memoria (p<0.01) y juego (p=0.02) (Figura 2B).
Figura 2: Respuestas encontradas entre los datos de hombres y mujeres. A) actividad electroencefalográfica. δ - delta 0.5-4 Hz, θ - teta 4-8 Hz, α = alfa 8-13 Hz y β - beta >13 Hz; B) diferencia entre los datos de frecuencia cardíaca (HR) y HRV. VJF: muy baja frecuencia 0-0.04 Hz; LF: baja frecuencia 0.04-0.15 Hz; HF: alta frecuencia 0.15-0.4 Hz. *Diferencia estadísticamente significativa p<0.05.
El análisis de la varianza mostró diferencias significativas entre las fases de la evaluación para el comportamiento de actividad teta. Para beta, las diferencias se observaron al comparar cada fase con el reposo. A su vez, la HR y la HRV mostraron diferencias significativas en la mayoría de las variables (Tabla 1).
Prueba de diferencias mínimas y su significativas entre las variables comparada con el reposo. HR: frecuencia cardíaca; HRV: variabilidad de la frecuencia cardíaca; VJF: muy baja frecuencia; LF: baja frecuencia; HF: alta frecuencia ; NS: no significativo. * p<0.05. † p<0.01. Fuente: Elaboración propia.Tabla 1: Resultados de ANOVA para la evaluación de diferencias entre las variables.

Comparadas con la señal obtenida en reposo, se observa que las variables electroencefalográficas con cambios estadísticamente significativos fueron el porcentaje de actividad teta, que se incrementó (p<0.01). Además, la actividad beta se redujo, delta no presentó variación y el aumento para alfa solo ocurrió en la fase de concentración de los hombres, donde pasó de 13.2% a 16.3% (p=0.03). (Tabla 1,Figura 1A).
Las correlaciones entre cambios electroencefalográficos y HRV vincularon el aumento de la actividad teta con una disminución del porcentaje de señal HF (r=-0.86) y un aumento de la HR (r=0.71). Del mismo modo, se asoció la reducción de beta a una reducción del porcentaje HF (r=0.96) y a un incremento de la HR (r=-0.75) (Tabla 2).
HR: frecuencia cardíaca; HRV: variabilidad de la frecuencia cardíaca; VLF: muy baja frecuencia; LF: baja frecuencia; HF: alta frecuencia. Fuente: Elaboración propia.Tabla 2: Correlaciones entre los datos electroencefalográficos y datos de variabilidad de la frecuencia cardíaca.

Los cambios en conjunto de la distribución de la actividad en el EGG, la variación de la HR y la distribución de la HRV se pueden observar en la Figura 3.
Figura 3: Respuestas fisiológicas observadas durante cada fase del estudio, promedio para todos los individuos n=39. A) Comportamiento de la actividad electroencefalográfica; B) HR = frecuencia cardíaca promedio; C) Variabilidad de la frecuencia cardíaca expresada en porcentaje: VLF = muy baja frecuencia, LF = baja frecuencia y HF = alta frecuencia.
Discusión
El presente estudio reveló que el comportamiento de la HR en todo el grupo fue superior durante el período de observación, memoria, concentración y juego, en comparación con el reposo, la cual es una respuesta que puede vincularse en primer lugar con una inactivación autonómica parasimpática que se manifestó en la reducción de la señal HF de la HRV 6. Estos resultados fueron similares a los reportados en otras situaciones como la transición desde el reposo hacia la actividad física, que tuvieron un aumento de la HR explicado por supresión vagal en especial 12.
Se han encontrado respuestas autonómicas en diversas formas de estrés psicosocial similares a las reportadas en este estudio 13. Se presume que la presión evolutiva ha impulsado adaptaciones funcionales para centrar la atención en la detección de amenazas, basadas en la información que provee la vía visual sin mediación inicial de la conciencia para facilitar respuestas fisiológicas autonómicas 14. Esta situación se parece a la observada en actividades que requieren memoria a corto plazo, en las que la actividad simpática ha aumentado 15.
Experimentos en ratas han revelado un crecimento de ondas teta ante un estímulo nuevo asociado al proceso de aprendizaje 2. Así mismo ocurre durante procesos de atención, en los que se reducen los ritmos corticales beta y gamma precedentes a un incremento de teta 16. Estos hallazgos son semejantes a los encontrados en el presente estudio, donde se evidenció una reducción de ritmo beta y aumento de teta durante la transición reposo-observación.
Los hallazgos proponen que los estímulos visuales que evocan procesos cognitivos de aprendizaje generan estímulos en la corteza orbitofrontal, que se encarga de procesos de asociación e involucra contexto, ubicación espacial, eventos, respuestas emocionales y toma de decisiones 17, entre otros. La participación de esta región es evidenciable por una mayor actividad teta 18. Desde la corteza prefontal se proyectan neuronas inhibitorias (GABAérgicas) hacia el núcleo central de la amígdala (CeA), lo que se asocia al desarrollo de emociones 19.
Del mismo modo, está demostrado que desde el CeA, se proyectan axones glutaminérgicos hacia la médula ventrolateral rostral desde donde se proyectan fibras excitatorias simpáticas preganglionares vía retículo espinal 20. Por otra parte, desde el CeA también se proyectan fibras inhibitorias GABAérgicas hacia el núcleo del tracto solitario, lo que podría conectarse con una reducción de la actividad parasimpática 21.
En general, en los procesos de comprensión de la regulación de la actividad autonómica cardiovascular, se tiene en cuenta el papel de los barorreceptores 22,23 y los quimiorreceptores 24. Sin embargo, en la actualidad, la adecuada comprensión requiere considerar de forma adicional el efecto mecánico del sistema respiratorio 25, el efecto de mecanorreceptores musculares 26, la presencia de citoquinas proinflamatorias por acción directa sobre el hipotálamo 27,28 y el efecto de la actividad cortical descrito hasta ahora en condiciones de sueño 29 y, como lo evidencian los resultados expuestos, también la actividad cortical en vigilia cuando hay transición del estado de reposo hacia demandas cognitivas observacionales de concentración, memoria y juego.
Con base en la evidencia, se ha sugerido el núcleo central de la amígdala como posible estructura fundamental en la relación entre la corteza cerebral y el sistema nervioso autónomo. No obstante, es necesario desarrollar nuevos estudios para comprobar esto.
Conclusiones
Los resultados en este estudio sugieren cambios en la respuesta electroencefalográfica de la corteza orbitofrontal, dados por una mayor actividad teta y asociados a una mayor actividad del sistema nervioso simpático y una menor respuesta del sistema nervioso parasimpático. Esto plantea que, adicional a moduladores clásicos como los barorreceptores y los quimiorreceptores, la corteza cerebral participa de forma activa en la regulación de la actividad autonómica durante la vigilia. Este fenómeno es evidente cuando se pasa del estado de reposo a una demanda cognitiva observacional, de concentración, memoria y juego.
Conflicto de intereses
Ninguno declarada por los autores.
Financiación
Ninguna declarada por los autores.
Agradecimientos
Al Laboratorio de Fisiología de la Universidad de La Sabana, que aportó los equipos para el desarrollo de la investigación, aunque no participó en el diseño, análisis o interpretación de datos, ni en la escritura, revisión o preparación del manuscrito. Además, agradecen a los estudiantes que participaron en la adquisición y tabulación de los datos y al ingeniero Oscar Mosquera por su apoyo en el procesamiento de los datos.
Referencias
Referencias
Schomer DL, Lopez da Silva FH. Niedermeyer’s Electroencephalography: Basic Principles, Clinical Applications, and Related Fields. 6th ed. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins; 2011.
Buzsáki G, Moser EI. Memory, navigation and theta rhythm in the hippocampal-entorhinal system. Nat Neurosci. 2013;16(2):130-8. http://doi.org/f4j6d2.
Suthana N, Haneef Z, Stern J, Mukamel R, Behnke E, Knowlton B, et al. Memory enhancement and deep-brain stimulation of the entorhinal area. N Engl J Med. 2012;366(6):502-10. http://doi.org/b4d3.
Gärtner M, Grimm S, Bajbouj M. Frontal midline theta oscillations during mental arithmetic: effects of stress. Front Behav Neurosci. 2015;9:96. http://doi.org/b4d4.
Gärtner M, Rohde-Liebenau L, Grimm S, Bajbouj M. Working memory-related frontal theta activity is decreased under acute stress. Psychoneuroendocrinology. 2014;43:105-13. http://doi.org/f5zzk5.
Kleiger RE, Stein PK, Bigger JT Jr. Heart rate variability: measurement and clinical utility. Ann Noninvasive Electrocardiol. 2005;10(1):88-101. http://doi.org/dj82kt.
Taelman J, Vandeput S, Spaepen A, Van Huffel S. Influence of Mental Stress on Heart Rate and Heart Rate Variability. In: Vander Sloten J, Verdonck P, Nyssen M, Haueisen J, editors. 4th European Conference of the International Federation for Medical and Biological Engineering. 22nd ed. Springer Berlin Heidelberg; 2009. p. 1366-9.
Thayer JF, Hansen AL, Saus-Rose E, Johnsen BH. Heart rate variability, prefrontal neural function, and cognitive performance: the neurovisceral integration perspective on self-regulation, adaptation, and health. Ann Behav Med. 2009;37(2):141-53. http://doi.org/czs6k3.
Harris DV, Harris BL. The athlete’s guide to sports psychology: Mental skills for physical people. New York: Leisure Press; 1984.
Tarvainen MP, Niskanen JP, Lipponen JA, Ranta-Aho PO, Karjalainen PA. Kubios HRV--heart rate variability analysis software. Comput Methods Programs Biomed. 2014;113(1):210-20. http://doi.org/b4d6.
Tarvainen MP, Ranta-Aho PO, Karjalainen PA. An advanced detrending method with application to HRV analysis. IEEE Trans Biomed Eng. 2002;49(2):172-5. http://doi.org/cfc247.
Beltrame T, Karsten M, Chacon-Mikahil MPT, Madruga VA, Silva E, Borghi-Silva A, et al. Age influence on the heart rate behavior on the rest-exercise transition: an analysis by deltas and linear regression. Rev Bras Med Esporte. 2012;18(5):300-4. http://doi.org/b4d7.
Petrowski K, Herold U, Joraschky P, Mück-Weymann M, Siepmann M. The effects of psychosocial stress on heart rate variability in Panic Disorder. German J Psychiatry. 2010;13(2):66-73.
Hedger N, Adams WJ, Garner M. Autonomic arousal and attentional orienting to visual threat are predicted by awareness. J Exp Psychol Hum Percept Perform. 2015;41(3):798-806. http://doi.org/f7fngz.
Sara SJ. The locus coeruleus and noradrenergic modulation of cognition. Nat Rev Neurosci. 2009;10(3):211-23. http://doi.org/cx34qb.
Brown RA, Walling SG, Milway JS, Harley CW. Locus ceruleus activation suppresses feedforward interneurons and reduces beta-gamma electroencephalogram frequencies while it enhances theta frequencies in rat dentate gyrus. J Neurosci. 2005;25(8):1985-91. http://doi.org/ffvrk9.
Euston DR, Gruber AJ, McNaughton BL. The role of medial prefrontal cortex in memory and decision making. Neuron. 2012;76(6):1057-70. http://doi.org/f4jhv4.
Hsieh LT, Ranganath C. Frontal midline theta oscillations during working memory maintenance and episodic encoding and retrieval. Neuroimage. 2014;85 Pt 2:721-9. http://doi.org/f5mhk8.
Barbas H, Saha S, Rempel-Clower N, Ghashghaei T. Serial pathways from primate prefrontal cortex to autonomic areas may influence emotional expression. BMC Neurosci. 2003;4:25. http://doi.org/dncqg7.
Chapp AD, Gui L, Huber MJ, Liu J, Larson RA, Zhu J, et al. Sympathoexcitation and pressor responses induced by ethanol in the central nucleus of amygdala involves activation of NMDA receptors in rats. Am J Physiol Heart Circ Physiol. 2014;307(5):H701-9. http://doi.org/f6fsj4.
Saha S. Role of the central nucleus of the amygdala in the control of blood pressure: descending pathways to medullary cardiovascular nuclei. Clin Exp Pharmacol Physiol. 2005;32(5-6):450-6. http://doi:.org/dhnkhw.
Kumagai H, Oshima N, Matsuura T, Iigaya K, Imai M, Onimaru H, et al. Importance of rostral ventrolateral medulla neurons in determining efferent sympathetic nerve activity and blood pressure. Hypertens Res. 2012;35(2):132-41. http://doi.org/d7x3c2.
Albaghdadi M. Baroreflex control of long-term arterial pressure. Rev Bras Hipertens. 2007;14(4):212-25.
Moreira TS, Takakura AC, Damasceno RS, Falquetto B, Totola LT, Sobrinho CR, et al. Central chemoreceptors and neural mechanisms of cardiorespiratory control. Braz J Med Biol Res. 2011;44(9):883-9. http://doi.org/dtn9hd.
Yasuma F, Hayano J. Respiratory sinus arrhythmia: why does the heartbeat synchronize with respiratory rhythm? Chest. 2004;125(2):683-90. http://doi.org/dtd8gm.
Gladwell VF, Fletcher J, Patel N, Elvidge LJ, Lloyd D, Chowdhary S, et al. The influence of small fibre muscle mechanoreceptors on the cardiac vagus in humans. J Physiol. 2005;567(2):713-21. http://doi.org/chjk2f.
Sajadieh A, Nielsen OW, Rasmussen V, Hein HO, Abedini S, Hansen JF. Increased heart rate and reduced heart-rate variability are associated with subclinical inflammation in middle-aged and elderly subjects with no apparent heart disease. Eur Heart J. 2004;25(5):363-70. http://doi.org/dd939s.
Marsland AL, Gianaros PJ, Prather AA, Jennings JR, Neumann SA, Manuck SB. Stimulated production of proinflammatory cytokines covaries inversely with heart rate variability. Psychosom Med. 2007;69(8):709-16. http://doi.org/ffns2t.
Kuo TB, Chen CY, Hsu YC, Yang CC. EEG beta power and heart rate variability describe the association between cortical and autonomic arousals across sleep. Auton Neurosci. 2016;194:32-7. http://doi.org/f776kf.
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. Daniel Alfonso Botero Rosas, Oscar Leonardo Mosquera Dussán, Carlos German Trujillo Rojas, Daniel Guzmán Pérez, Jhonnatan Eduardo Zamudio Palacios, José Antonio García Torres, Angie Paola Terán Ortega. (2020). Decision Making, Stress Assessed by Physiological Response and Virtual Reality Stimuli. Revista Colombiana de Psicología, 29(2), p.89. https://doi.org/10.15446/rcp.v29n2.74280.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2017 Revista de la Facultad de Medicina

Esta obra está bajo una licencia Creative Commons Reconocimiento 3.0 Unported.
-