RP-HPLC-DAD determination of free amino acids in cocoa samples during fermentation and roasting
Determinación de aminoácidos libres en muestras de cacao durante la fermentación y el tostado por cromatografía líquida con detección DAD
Determinação de aminoácidos livres em amostras de cacau durante a fermentação e torrefação por cromatografia líquida com detecção DAD
DOI:
https://doi.org/10.15446/rev.colomb.quim.v1n49.77811Palabras clave:
Liquid chromatography, column switching, derivatization, microwave radiation, cocoa. (en)cromatografía líquida, sistemas de columnas acopladas, derivatización, radiación por microondas, cacao (es)
cromatografia líquida, sistemas de colunas acopladas, derivatização, radiação de microondas, cacau (pt)
Descargas
Amino acids (AA) composition in cocoa beans can predict the synthesis of compounds which affect cocoa flavor. Thus, their determination is of great interest for the community implied in the commercialization and production of cocoa. In consequence, in this work, the analysis of AA produced during cocoa beans fermentation and roasting was carried out. A high-performance liquid chromatographic method with DAD detection at 254 nm was optimized and validated for their selective determination in six varieties of cocoa beans with different genotypes, all of them grown in Venezuela. AA were extracted by defatted milled cocoa powder ultrasonication using purified water at 70 ºC. Then, they were derivatized with phenyl isothiocyanate, and their derivatives were separated, using a reversed-phase column with gradient elution, achieving a satisfactory resolution among the peaks (greater than 1.0) in less than 29 min. 110 cocoa samples were analyzed. Results showed a significant content of free AA, ranging from 3.87 to 5.97 g/kg in absence of fermentation with a predominance of acidic AA. Moreover, there is a progressive increase in the AA content while fermentation process occurs, with a predominance of hydrophobic AA such as alanine, valine, isoleucine, leucine, phenylalanine, and tyrosine. On the other hand, all cocoa types showed a partial degradation of free AA during the roasting step, especially the hydrophobic ones.
La determinación de aminoácidos (AA) en granos de cacao es de gran interés ya que estos son considerados como unos de los precursores de su sabor y aroma. Por esta razón, el presente trabajo tuvo como objetivo optimizar y validar un método por cromatografía líquida con detección DAD a 254 nm para la determinación selectiva de AA durante la fermentación y tostado en seis variedades de granos de cacao con diferentes genotipos, todos estos cultivados en Venezuela. Los AA se extrajeron del polvo de cacao molido y desgrasado con agua pura a 70 ºC, utilizando la técnica de ultrasonido. Luego, se derivatizaron con fenilotiocianato para separar sus derivados con buena resolución en menos de 29 min en una columna de fase reversa, utilizando gradiente de elución. Se analizaron 110 muestras de cacao. Los resultados mostraron un contenido significativo de AA libres, entre 3,87 y 5,97 g/kg, en ausencia de fermentación con predominio de AA ácidos, y un aumento progresivo en el contenido de AA, mientras ocurre el proceso de fermentación, con un predominio de AA hidrófobos como alanina, valina, isoleucina, leucina, fenilalanina y tirosina. Además, todos los tipos de cacao mostraron una degradación parcial de AA libres durante la etapa de tostado, especialmente los AA hidrófobos.
A determinação dos aminoácidos (AA) nos grãos de cacau é importante, pois são considerados um dos precursores de seu sabor e aroma. Neste trabalho, um método foi otimizado e validado por cromatografia líquida com detecção DAD a 254 nm para a determinação seletiva de AA durante a fermentação e torrefação em seis variedades de grãos de cacau com diferentes genótipos, todos cultivados na Venezuela. Os AAs foram extraídos do pó de cacau moído e desengordurados com água pura a 70 ° C usando a técnica de ultrassom. Em seguida, foram derivatizados com feniltiocianato, e os derivados foram separados com boa resolução em menos de 29 minutos em uma coluna de fase invertida usando eluição em gradiente. Foram analisadas 110 amostras de cacau. Os resultados mostraram um conteúdo significativo de AA livre entre 3,87 e 5,97 g/kg na ausência de fermentação com predominância de AA ácidos e um aumento progressivo no conteúdo de AA enquanto o processo de fermentação ocorre com predominância de AA hidrófobos como alanina, valina, isoleucina, leucina, fenilalanina e tirosina. Além disso, todos os tipos de cacau apresentaram uma degradação parcial do AA livre durante a fase de torrefação, principalmente o AA hidrofóbico.

Química aplicada y analítica
RP-HPLC-DAD determination of free amino acids in cocoa samples during fermentation and roasting
Determinación de aminoácidos libres en muestras de cacao durante la fermentación y el tostado por cromatografía líquida con detección DAD
Determinação de aminoácidos livres em amostras de cacau durante a fermentação e torrefação por cromatografia líquida com detecção DAD
RP-HPLC-DAD determination of free amino acids in cocoa samples during fermentation and roasting
Revista Colombiana de Química, vol. 49, núm. 1, 2020
Universidad Nacional de Colombia
Recepción: 11 Febrero 2019
Aprobación: 09 Octubre 2019
Abstract: Amino acids (AA) composition in cocoa beans can predict the synthesis of compounds which affect cocoa flavor. Thus, their determination is of great interest for the community implied in the commercialization and production of cocoa. In consequence, in this work, the analysis of AA produced during cocoa beans fermentation and roasting was carried out. A high-performance liquid chromatographic method with DAD detection at 254 nm was optimized and validated for their selective determination in six varieties of cocoa beans with different genotypes, all of them grown in Venezuela. AA were extracted by defatted milled cocoa powder ultrasonication using purified water at 70 ºC. Then, they were derivatized with phenyl isothiocyanate, and their derivatives were separated, using a reversed-phase column with gradient elution, achieving a satisfactory resolution among the peaks (greater than 1.0) in less than 29 min. 110 cocoa samples were analyzed. Results showed a significant content of free AA, ranging from 3.87 to 5.97 g/kg in absence of fermentation with a predominance of acidic AA. Moreover, there is a progressive increase in the AA content while fermentation process occurs, with a predominance of hydrophobic AA such as alanine, valine, isoleucine, leucine, phenylalanine, and tyrosine. On the other hand, all cocoa types showed a partial degradation of free AA during the roasting step, especially the hydrophobic ones.
Keywords: liquid chromatography, column-switching, derivatization, microwave radiation, cocoa.
Resumen: La determinación de aminoácidos (AA) en granos de cacao es de gran interés ya que estos son considerados como unos de los precursores de su sabor y aroma. Por esta razón, el presente trabajo tuvo como objetivo optimizar y validar un método por cromatografía líquida con detección DAD a 254 nm para la determinación selectiva de AA durante la fermentación y tostado en seis variedades de granos de cacao con diferentes genotipos, todos estos cultivados en Venezuela. Los AA se extrajeron del polvo de cacao molido y desgrasado con agua pura a 70 ºC, utilizando la técnica de ultrasonido. Luego, se derivatizaron con fenilotiocianato para separar sus derivados con buena resolución en menos de 29 min en una columna de fase reversa, utilizando gradiente de elución. Se analizaron 110 muestras de cacao. Los resultados mostraron un contenido significativo de AA libres, entre 3,87 y 5,97 g/kg, en ausencia de fermentación con predominio de AA ácidos, y un aumento progresivo en el contenido de AA, mientras ocurre el proceso de fermentación, con un predominio de AA hidrófobos como alanina, valina, isoleucina, leucina, fenilalanina y tirosina. Además, todos los tipos de cacao mostraron una degradación parcial de AA libres durante la etapa de tostado, especialmente los AA hidrófobos.
Palabras clave: cromatografía líquida, sistemas de columnas acopladas, derivatización, radiación por microondas, cacao.
Resumo: A determinação dos aminoácidos (AA) nos grãos de cacau é importante, pois são considerados um dos precursores de seu sabor e aroma. Neste trabalho, um método foi otimizado e validado por cromatografia líquida com detecção DAD a 254 nm para a determinação seletiva de AA durante a fermentação e torrefação em seis variedades de grãos de cacau com diferentes genótipos, todos cultivados na Venezuela. Os AAs foram extraídos do pó de cacau moído e desengordurados com água pura a 70 ° C usando a técnica de ultrassom. Em seguida, foram derivatizados com feniltiocianato, e os derivados foram separados com boa resolução em menos de 29 minutos em uma coluna de fase invertida usando eluição em gradiente. Foram analisadas 110 amostras de cacau. Os resultados mostraram um conteúdo significativo de AA livre entre 3,87 e 5,97 g/kg na ausência de fermentação com predominância de AA ácidos e um aumento progressivo no conteúdo de AA enquanto o processo de fermentação ocorre com predominância de AA hidrófobos como alanina, valina, isoleucina, leucina, fenilalanina e tirosina. Além disso, todos os tipos de cacau apresentaram uma degradação parcial do AA livre durante a fase de torrefação, principalmente o AA hidrofóbico.
Palavras-chave: cromatografia líquida, sistemas de colunas acopladas, derivatização, radiação de microondas, cacau.
Introduction
Cocoa beans are originated as seeds in fruit pods of the tropical crop Theobroma cacao L., that is cultivated in the humid lowland tropics generally by small-scale producers. There are three main cocoa types: Forastero (bulk grade), Criollo (fine grade), and hybrid Trinitario (fine grade). These varieties offer wide differences in their final flavor due to factors such as their genotype, soil, climate, and harvest conditions, and to the post-harvest technology used in fermentation, drying, and roasting stages [1].
Fermentation plays a major role in the development of aroma precursors like free amino acids, peptides, and reducing sugars, due to the microbial activity and endogenous enzymes of cocoa beans. Free amino acids and peptides are formed by proteolytic reactions induced by endogenous proteases (aspartic proteinase and carboxypeptidase) during cocoa beans storage [2, 3]. Contrarily, reducing sugars such as fructose and glucose are products of the hydrolysis of sucrose [4, 5]. Both amino acids and reducing sugars are precursors in Maillard’s reaction. Thus, they are responsible of the formation of the compounds which affect cocoa flavor during roasting [6, 7, 8, 9]. Therefore, determination of the amino acid profile is extremely important to assess the cocoa nutritional value as well as to predict the development of flavor compounds during the manufacture of cocoa-based products. An optimum fermentation originates good flavor and aroma, while an inadequate process might considerably demerit the product [4, 10]. Voigt et al. [2, 3] concluded that peptides and hydrophobic amino acids are important cocoa-specific flavor precursors. On the other hand, Kirchhoff et al. [11, 12] pointed out a correlation between the accumulation of free amino acids and the generation of essential aroma precursors, also noticed that they are formed by pH-dependent proteolysis processes. Moreover, Afoakwa et al. [1] observed that the nature of the amino components is critical for cocoa flavor due to their influence in the formation of heterocyclic compounds which affect the resulting aroma.
Therefore, to evaluate the cocoa quality is mandatory to study not only the compounds directly related with the flavor, but also their precursors. Thus, the analysis of amino acids during fermentation, drying and roasting stages could provide useful information to improve the post-harvest treatments and obtain a high-quality cocoa.
Venezuela is considered a producer of fine cocoa with excellent quality and a characteristic flavor. In recent years, measures have been implemented to increase production and to improve the economic competitiveness of cocoa. Several researchers [13-22] have been focused on the study of the chemical and physical parameters involved in the fermentation and drying processes, with the aim of improving post-harvest processes to increase the cocoa quality, especially of the Criollo variety. However, amino acids behavior during the different post-harvest stages has not been studied, although they are essential for cocoa flavor development.
Therefore, the main objective of this work was the determination of total and free amino acids content in Venezuelan cocoa during the fermentation and roasting stages. For this purpose, a high-performance liquid chromatographic method with DAD detection was optimized and validated.
Materials and methods
Reagents, solvents and standards
All solvents were of HPLC grade and all reagents were of analytical grade, unless indicated otherwise. Ethanol (EtOH) and acetonitrile (MeCN) were purchased from J.T. Baker (Phillipsburg, NJ, USA). All amino acids were obtained from Sigma-Aldrich (St. Louis, MO, USA). Phenylisothiocyanate (PITC) and triethylamine (TEA) (99% w/w) were obtained from Sigma-Aldrich. Milli-Q ultrapure water with a specific resistivity of 18 Mcm−1 was used for standards and samples preparation, reagents dilution and for cleaning purposes.
Acetate buffer was prepared with sodium acetate solution 0.07 mol L-1 and 99.7% w/w acetic acid from Fisher obtaining a final concentration of 0.05 mol L-1 and a pH=5.1. Individual stock standard solutions with 10000 µg mL-1 of each amino acid were prepared in reagent-grade water. All solutions were stored under refrigeration at 4 ºC. Working solutions were weekly prepared by dilution of stock standard solutions in water.
Cocoa samples
To study the applicability of the proposed method, Criollo, Trinitario, and Forastero cocoa beans were fermented (1-7 days) and then roasted prior to their analysis.
Cocoa pods of the varieties ICS-1 (Trinitario) and IMC-67 (Forastero) were obtained from genetically identified trees grown on the Cocoa Genebank of Ocumare de la Costa (INIA-Aragua, Venezuela). Criollo Guasare, Zea, and San Juan were provided by the Cocoa Genebank of San Juan de Lagunillas (INIA-Mérida, Venezuela), and Porcelana was obtained from the Cocoa Genebank of the Local Chama Station (Corpozulia, Venezuela).
Samples were processed as follows: cocoa seeds were subjected to fermentation process according to their variety. This process was performed in triplicate into 60 mLwooden boxes. The fermenting mass was turned over every 24 h. Each day three replicates were collected and analyzed. After fermentation, a drying process was performed for 6 days. Then, some of the samples were grounded, sieved (≤ 42 mesh), and defatted, according to the Soxhlet extraction method [23]. These were stored at -20 °C until analysis. Besides, with the aim of studying the roasting effect on the amino acids and reducing sugars profile, some fermented and dried samples were first roasted and then grounded and defatted. All samples were conserved in sealed PTFE containers, which were kept under refrigeration (4 °C) until analysis.
Extraction and derivatization of the amino acids
To carry out the total free amino acids extraction, 0.800 g of defatted cocoa powder were ultrasonicated for 15 min at 70 ºC in 15 mL of purified water. The obtained extract was cooled to room temperature and centrifuged at 3500 rpm for 15 min. Then the supernatant was transferred to a 25 mL volumetric flask filled with water and then filtered through a 0.22 μm membrane (Membrane support-220, Waters, USA).
Free amino acids were derivatized with PITC, following the method described by Bidlingmeyer et al. [24] with some modifications. Thus, 20 µL of sample were placed into a vial and mixed with 20 µL of ethanol and 20 µL of the derivative reagent solution (1:1:7 v/v/v PITC:TEA:Ethanol ). Then, the vial was capped and exposed to microwave radiation in a domestic microwave oven (340 W for 40 s). Finally, 20 µL of the PTC-AA extract were injected into the HPLC system.
Amino acid derivatives analysis
After extraction and derivatization procedures, amino acid derivatives (PTC-AA) were present in a complex mixture, making necessary a sample cleanup to obtain chromatograms with good resolution.
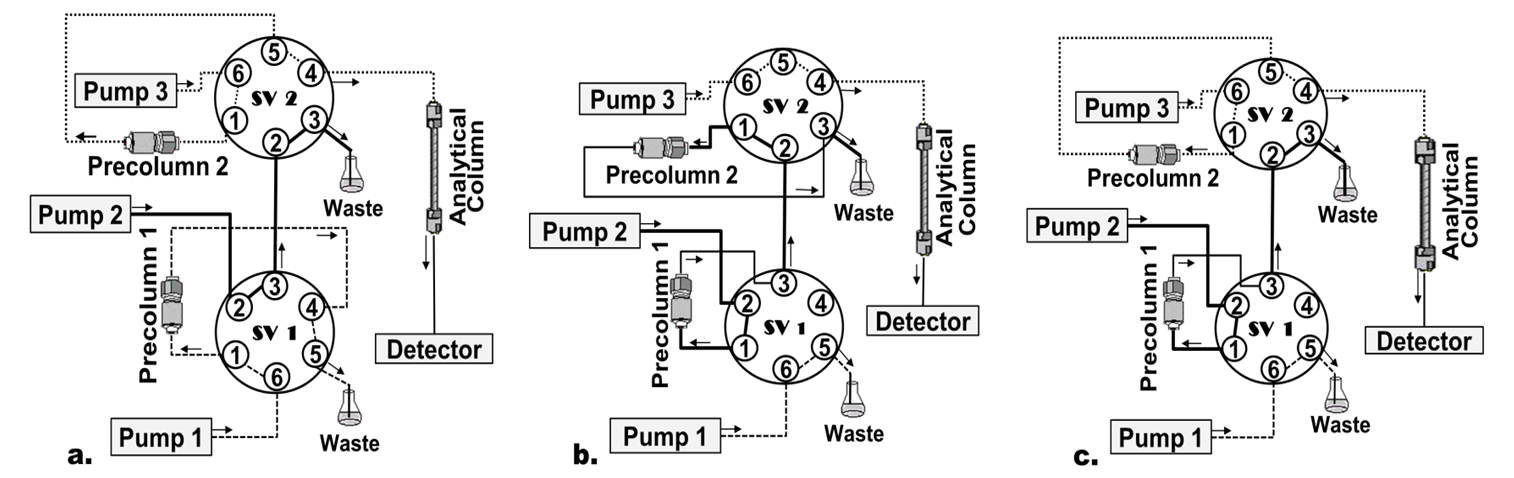
A column-switching HPLC system in the circuit transfer mode was used for sample cleanup and PTC-AA separation and detection. The chromatographic system was composed by a quaternary pump, a column oven, an autosampler, a diode array detector (DAD) (Series 200 HPLC System, PerkinElmer; Norwalk, CT), and a digital workstation with TotalChrom software (Version 6.3) which was used as controller and data manager. Two additional Waters pumps (model 515) (Milford, MA, USA, Pump 1, Pump 2) were used to propel the extraction and transfer mobile phases. Injections were made with a Rheodyne type 7125 six-port valve equipped with a 100 μL loop. Two column-switching six-port valves (Supelco, Bellefonte, PA, USA; SV 1, SV 2) were attached to the system and controlled by the workstation. Three columns were used in the system: 1) Precolumn 1: Oasis HLB column 20 x 3.9 mm; dp (particle diameter): 25 μm, used as extraction column; 2) Precolumn 2: Supelcosil™ LC-18 20 x 3 mm; dp: 5 µm, which replaced the loop injection previous to the analytical column; 3) Advantage Kromasil C18 column 150 x 3.0 mm i.d.; dp: 3 μm, used as analytical column. Figure 1 shows a schematic depiction of the column-switching system.
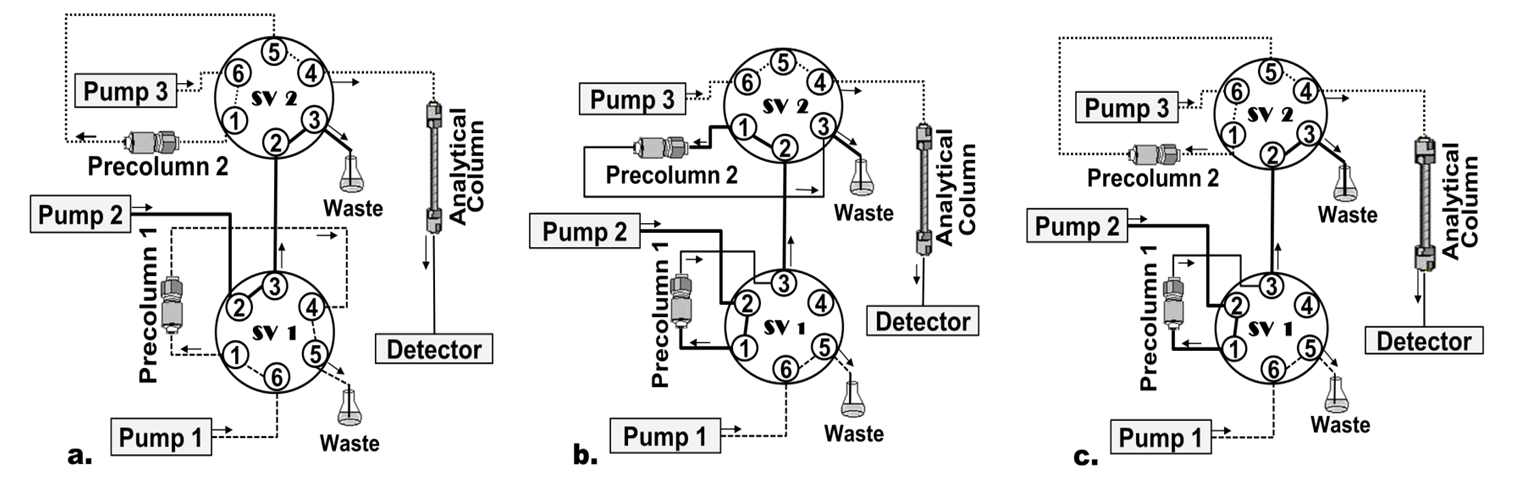
Figure 1
Schematic diagram of the columnswitching HPLC system The direction of flow is indicated by arrows a Sample charging precolumn 1 SV 1 load position SV 2 inject position b Sample charging precolumn 2 SV 1 inject position SV 2 load position c Final elution and injection into the HPLC system SV 1 load position SV 2 inject position
Figure 1. Schematic diagram of the column-switching HPLC system. The direction of flow is indicated by arrows. a. Sample charging precolumn 1: SV 1: load position, SV 2: inject position; b. Sample charging precolumn 2: SV 1: inject position, SV 2: load position; c. Final elution and injection into the HPLC system: SV 1: load position, SV 2: inject position.
Analysis procedure
With SV 1 in load position (Figure 1a), 100 µL of PTC-AA extract were propelled by pump 1 at a flow-rate of 1 mL min-1, injected into the extraction column (precolumn 1), and washed with 2% acetonitrile in acetate buffer. PTC-AA were retained while endogenous sample components were eluted to waste. After 0.5 min, SV 1 was automatically rotated to inject position (Figure 1b) allowing pump 2 to propel the mobile phase (90% ethanol in acetate buffer) at 1.8 mL min-1. This way, enriched PTC-AA were eluted from extraction column and injected into precolumn 2.
After 0.13 min, SV 2 was changed to inject position and PCT-AA extract with 2% acetonitrile in acetate buffer was pushed by the pump 3 at 1.7 mL min-1 into the analytical column to perform the analytes separation. Simultaneously, SV 1 was changed to load position to carry out the re-equilibration of the extraction column (precolumn 1) before the next injection (Figure 1c).
Analytes separation was performed at 45 °C using two different mobile phases, mobile phase A: 2% acetonitrile in acetate buffer and mobile phase B: acetonitrile-water (60:40 v/v) in a gradient mode elution at flow rate of 1.7 mL min-1 and with the following program: 0 % solvent B (100% A) (5 min) to 35% B at 1.5 %B min-1 and up to 100% B (3 min) at 32.5 %B min-1.
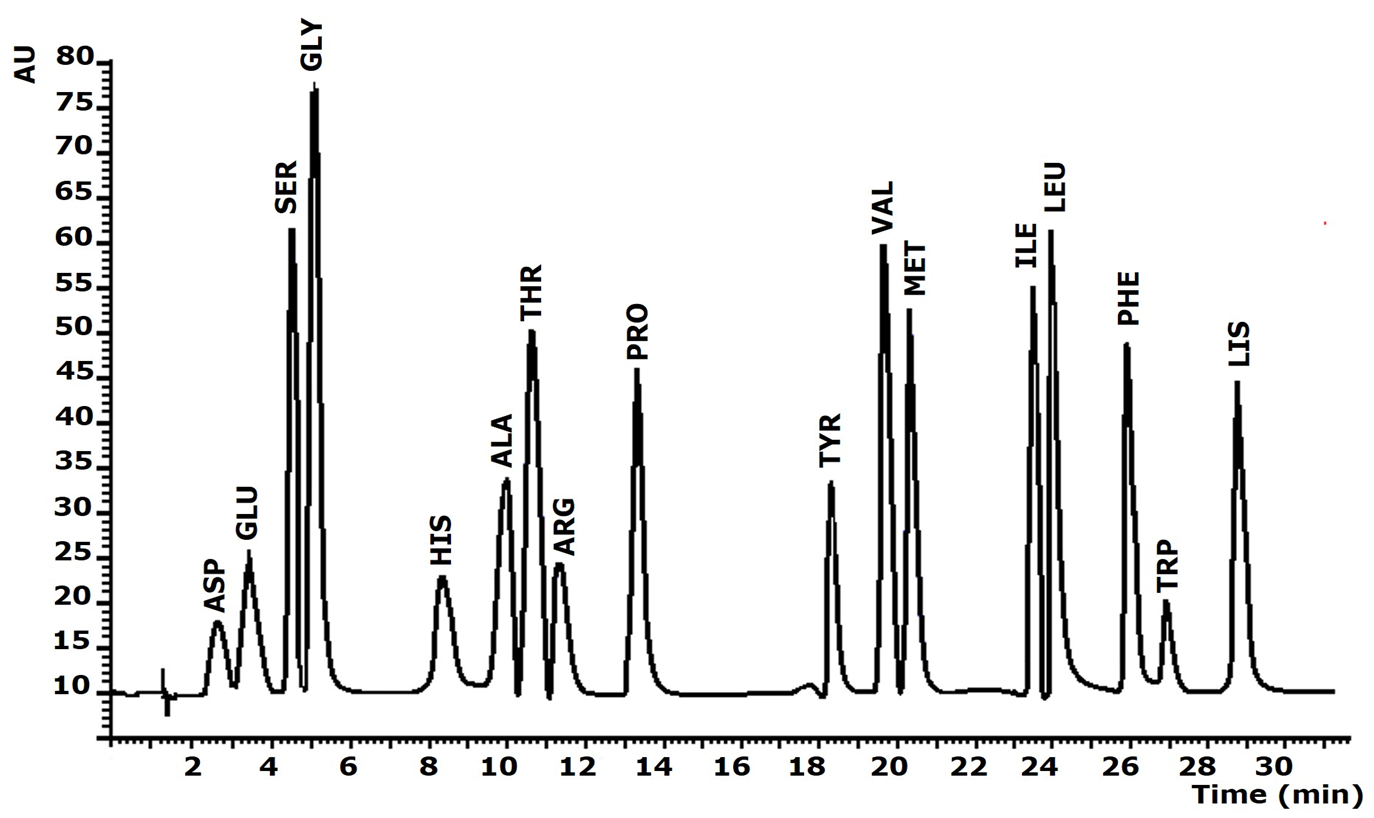
A summary of the system operating conditions is shown in Table 1. Under these conditions, analytes were separated in less than 29 min, with good resolution and no matrix effect. A chromatogram of the separation of the AA derivatives is shown in Figure 2.
Table 1. Optimized experimental conditions of the procedure.
| Step | System component | Conditions |
| Sample injection (t = 0 min) | Rheodyne 7125 valve: inject position Switching-valve 1: load position Switching-valve 2: inject position | Injection volume: 100 µL |
| Sample extraction (t =0.50 min) | Extraction precolumn 1:Oasis HLB (20 x 3.9 mm; dp: 25 μm) | Mobile phase: 2% acetonitrile in 0.05 mol L-1 acetate buffer (pH= 5.1) Flow rate: 1.0 mL min-1 |
| Analytes injection into Precolumn 2 (t =0.13 min) | Precolumn 2: Supelcosil LC-18 (20 x 3 mm; dp: 5 µm) Switching-valve 1: inject position Switching-valve 2: load position | Mobile phase: 90% ethanol in 0.05mol L-1 acetate buffer (pH= 5.1) Flow rate: 1.8 mL min-1 |
| Analytes injection into the analytical column Precolumn 1: re-conditioning before next injection | Advantage Kromasil C18 100A (150mm x 3.0 mm; 3µm) Switching-valve 1: load position Switching-valve 2: inject position | Solvent A: 2% actonitrile in 0.05mol L-1 acetate buffer (pH= 5.1) Flow rate: 1.7 mL min-1 Mobile phase: 2% acetonitrile in 0.05 mol L-1 acetate buffer (pH= 5.1) Flow rate: 1.0 mL min-1 |
| Chromatographic separation (t = 29 min) | Switching-valve 2: inject position | Solvent A: 2% actonitrile in 0.05 mo L-1 acetate buffer (pH= 5.1) Solvent B: 60% acetonitrile in water Gradient program: 0 % solvent B (100% A) (5 min) to 35% B at 1.5 B% min-1, and up to 100% B (3 min) at 32.5 B% min-1 Flow- ate: 1.7 mL min-1 Detection: DAD a 254nm |
Results and discussion
Microwave assisted derivatization optimization
PITC reacts with primary and secondary AA [25, 26]. The reaction occurred at room temperature and the best results were obtained with a reaction time of 15 min.
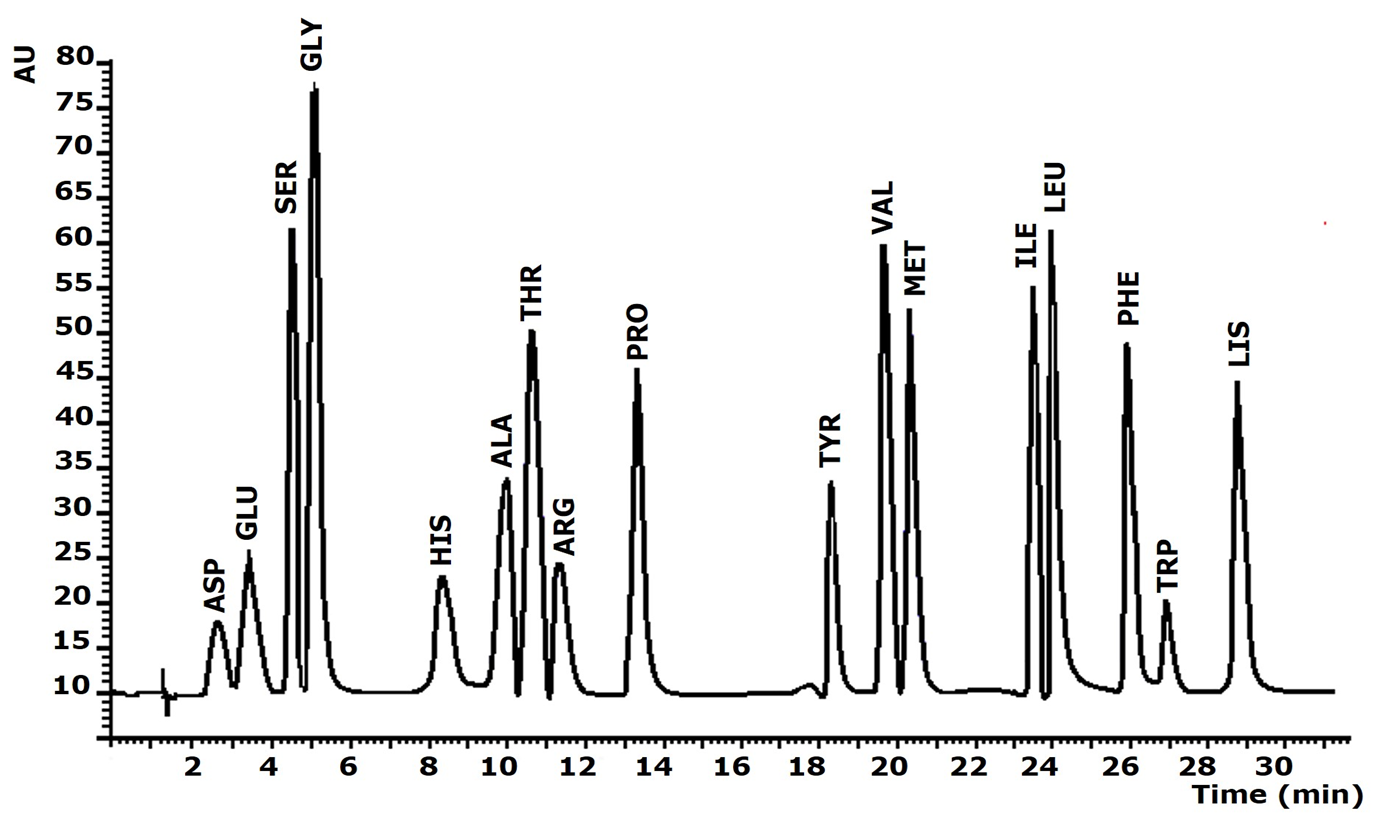
Figure 2
Chromatogram for PTCAA separation obtained under the optimized procedure for a standard solution of AA at concentration of 10 µg mL1 experimental conditions in Table 1
Figure 2. Chromatogram for PTC-AA separation obtained under the optimized procedure for a standard solution of AA at concentration of 10 µg mL-1 (experimental conditions in Table 1).
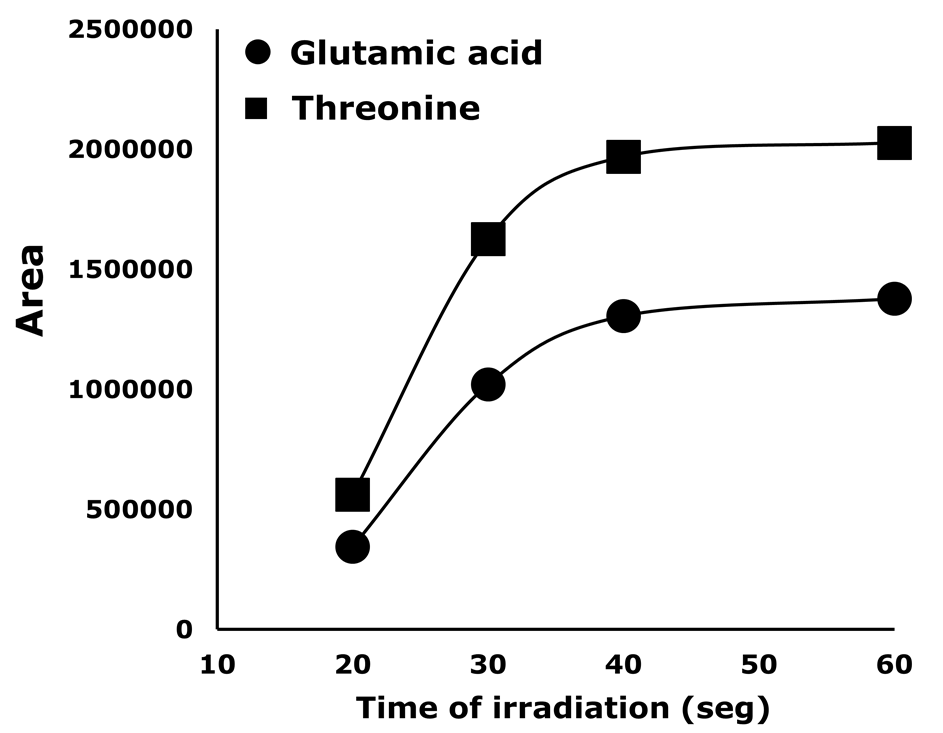
Aiming to decrease the analysis time, microwave radiation was used to assist the reaction [27]. Parameters such as microwave power and reaction time should be studied when this strategy is used. Therefore, time reaction was studied for two AA (glutamic acid and threonine) while maintaining microwave power at 340 W. The obtained results are shown in Figure 3.
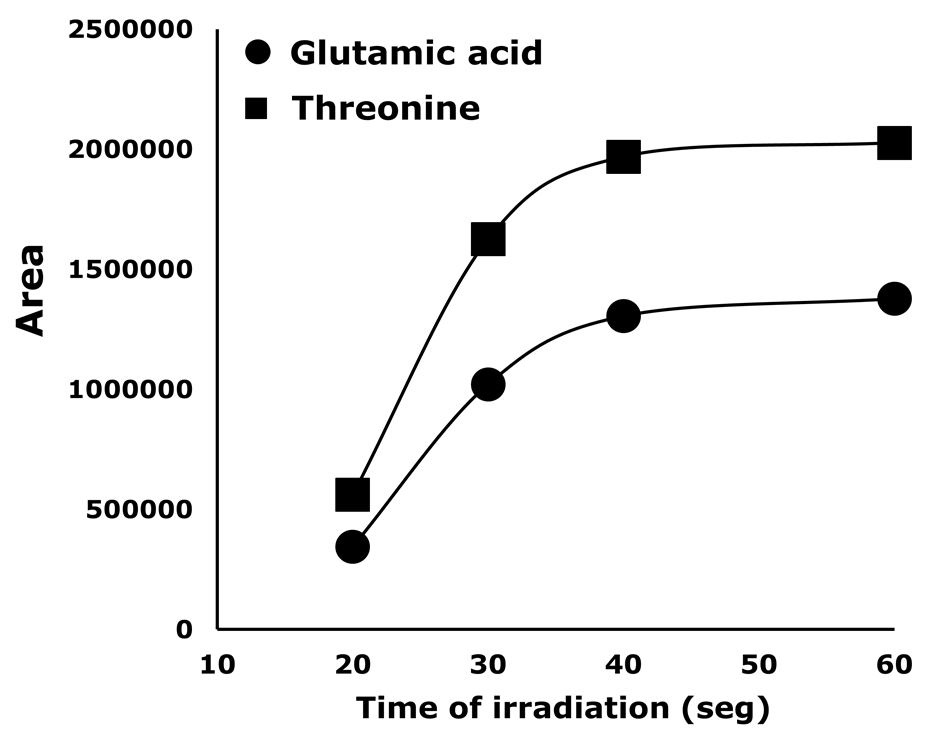
Figure 3
Effect of the irradiation time at microwave power of 340 W
Figure 3. Effect of the irradiation time at microwave power of 340 W.
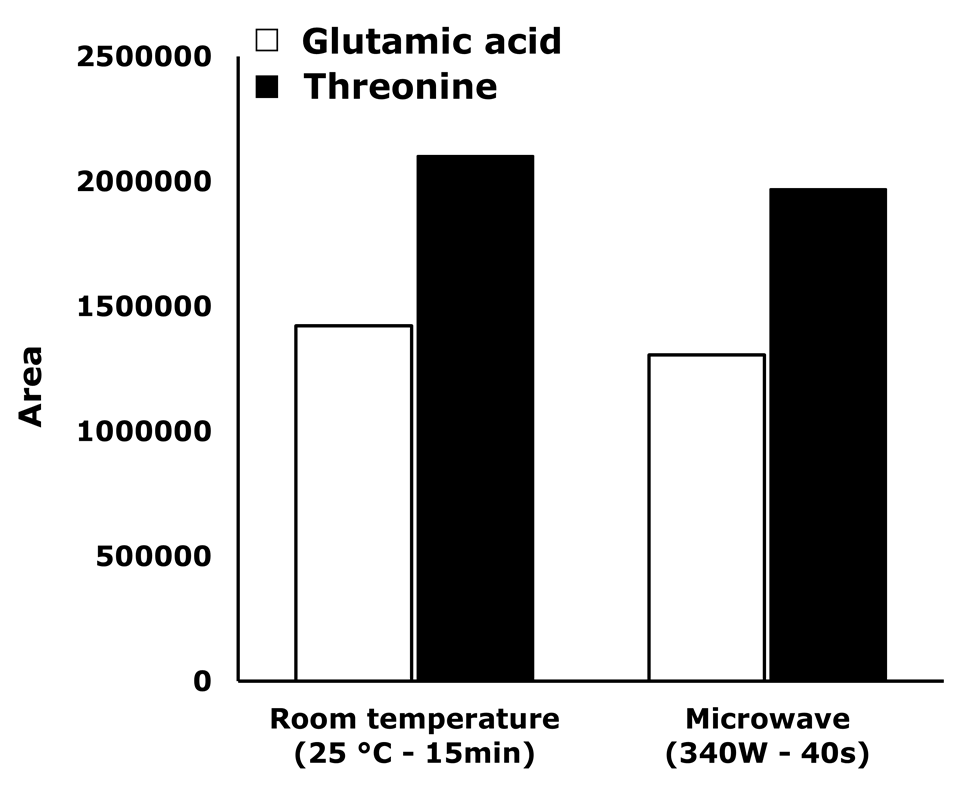
As can be seen, irradiation time and peak area increased proportionally until 40 s. In consequence, 40 s was set as optimum derivatization time. Additionally, in Figure 4 a comparison between the derivatization reaction performance under standard conditions and assisted by microwave irradiation is presented. Although a similar efficiency is obtained with both methods, the reaction time is drastically decreased by using the microwave (from 15 min to 40 s).
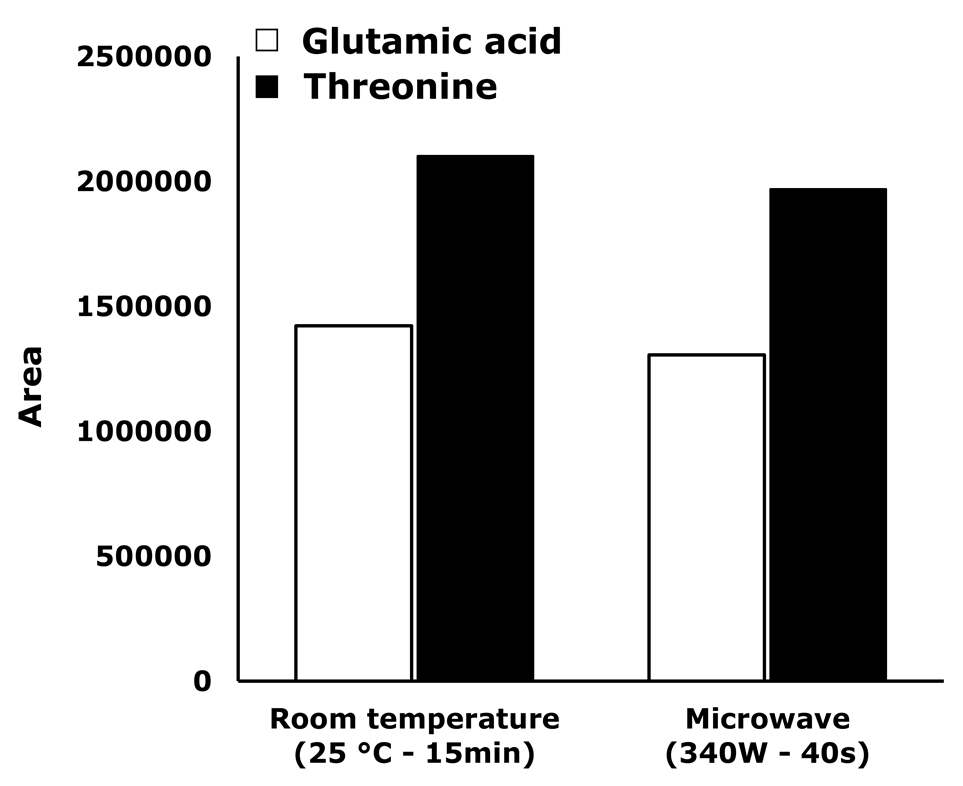
Figure 3
Effect of the irradiation time at microwave power of 340 W
Figure 4. Comparison of derivatization performance at standard conditions and assisted by microwave irradiation.
Solid phase extraction
A Waters Oasis HLB precolumn was used for sample clean up and analytes extraction and preconcentration. This column is a universal polymeric reversed phase sorbent which allows to obtain quantitative and reproducible recoveries of the analytes due to the presence of a balanced composition of two monomers: .-vinilpirrolidone (hydrophilic) and divinylbencene (hydrophobic) [28]. The retention mechanisms are dipole-dipole interactions and Van der Waals forces for polar and non-polar compounds, respectively. Experimental parameters were optimized to achieve the maximum efficiency during extraction. The obtained results can be seen in Table 1.
Method validation
Accuracy, precision, linearity and matrix effect studies were carried out to validate the proposed method. The accuracy of the procedure was evaluated by performing recovery tests using spiked cocoa samples at different amino acid concentration levels. In all cases, recovery values ranged between 89% and 109%, with a relative standard deviation (%RSD) lower than 3.85% (. = 5), which indicates an excellent efficiency for the amino acids extraction and derivatization procedures.
The within-day and between-day precisions, expressed as %RSD, were established at three different amino acid concentration levels (low, middle and high) for aqueous standard solutions and for spiked cocoa samples. RSD < 2.9% (. = 5) were obtained for all the analytes, confirming the satisfactory precision of the proposed procedure.
Calibration curves for each AA were obtained from both standard aqueous solutions and spiked cocoa samples (standard additions) in a wide range of concentration (0.1 - 15.0 µg mL-1). Each calibration set included seven data points and each point was run at least three times. Good linearity between peak area and concentrations was achieved with correlation coefficients R. > 0.9900 in all cases. Sample matrix effect was studied by comparing the slopes of the calibration curves of the standard aqueous solutions with the slopes obtained when analyzing the spiked samples. A paired student t-test indicated that the slopes were not statistically different (P < 0.05), which denoted the absence of any matrix effect. Limits of detection, defined as a signal/noise ratio of 3:1, were between 0.04 and 0.20 µg mL-1 for all amino acids.
Free amino acids content in cocoa samples
Results of the free amino acid contents in the analyzed cocoa samples are shown in Table 2.
Table 2. Free amino acids content (g/kg) for unfermented and fermented cocoa samples.
| DF | 0 | 3 | 0 | 3 | 0 | 3 | 0 | 3 | 0 | 7 | 0 | 7 | 0 | 7 | 0 | 7 | 0 | 7 | 0 | 7 |
| Amino acids (g/kg) | Por | Por | Zea | Zea | Gua | Gua | SJ | SJ | IMC-67a | IMC-67a | IMC-67b | IMC-67b | IMC-67c | IMC-67c | IMC-67d | IMC-67d | ISC – 1a | ISC – 1a | ISC – 1b | ISC – 1b |
| Acidic | 1.35 | 0.37 | 2.23 | 0.98 | 0.66 | 1.08 | 3.38 | 1.46 | 1.37 | 1.15 | 1.03 | 1.02 | 1.79 | 0.94 | 0.75 | 0.47 | 1.20 | 0.46 | 1.26 | 0.79 |
| Asp | 0.56 | 0.26 | 0.56 | 0.15 | 0.30 | 0.59 | 1.23 | 0.46 | 0.58 | 0.17 | 0.27 | 0.27 | 0.50 | 0.18 | 0.22 | 0.12 | 0.42 | 0.17 | 0,51 | 0.24 |
| Glu | 0.79 | 0.12 | 1.67 | 0.74 | 0.36 | 0.49 | 2.15 | 1.00 | 0.79 | 0.97 | 0.76 | 0.75 | 1.23 | 0.76 | 0.53 | 0.36 | 0.75 | 0.28 | 0.75 | 0.53 |
| Hydro phobic | 2.26 | 9.51 | 2.51 | 7.53 | 1.87 | 6.36 | 1.78 | 5.02 | 1.54 | 8.22 | 0.94 | 11.71 | 1.93 | 8.77 | 2.02 | 9.92 | 1.42 | 6.48 | 1.42 | 7.32 |
| Leu | 0.29 | 1.85 | 0.45 | 1.55 | 0.20 | 1.50 | 0.11 | 1.04 | 0.13 | 2.26 | 0.10 | 2.66 | 0.19 | 1.83 | 0.22 | 2.21 | 0,08 | 1.66 | 0.17 | 1.30 |
| Ala | 0.33 | 1.76 | 0.51 | 1.67 | 0.17 | 0.47 | 0.11 | 0.89 | 0.19 | 0.65 | 0.13 | 1.12 | 0.29 | 1.22 | 0.32 | 1.77 | 0,11 | 0.89 | 0.26 | 0.79 |
| Phe | 0.41 | 1.15 | 0.43 | 1.00 | 0.28 | 1.10 | 0.06 | 0.83 | 0.16 | 0.81 | 0.12 | 1.51 | 0.27 | 1.18 | 0.10 | 1.40 | 0,10 | 1.08 | 0.18 | 1.47 |
| Tyr | 0.24 | 0.32 | 0.12 | 0.42 | 0.48 | 0.82 | 0.17 | 0.58 | 0.32 | 0.49 | 0.17 | 0.99 | 0.28 | 0.77 | 0.49 | 0.63 | 0.42 | 0.77 | 0.07 | 1.05 |
| Val | 0.14 | 1.30 | 0.23 | 0.47 | 0.10 | 0.46 | 0.10 | 0.34 | 0.08 | 0.59 | 0.06 | 1.36 | 0.12 | 0.53 | 0.12 | 1.01 | 0.06 | 0.36 | 0.08 | 0.45 |
| Ile | 0.16 | 1.09 | 0.31 | 0.80 | 0.14 | 0.46 | 0.06 | 0.29 | 0.10 | 1.27 | 0.06 | 1.44 | 0.13 | 0.60 | 0.15 | 0.80 | 0.09 | 0.32 | 0.08 | 0.56 |
| Pro | 0.52 | 1.34 | 0.29 | 1.08 | 0.37 | 1.19 | 1.14 | 0.79 | 0.50 | 1.21 | 0.25 | 1.67 | 0.50 | 1.45 | 0.46 | 1.28 | 0.46 | 1.13 | 0.51 | 1.17 |
| Met | 0.01 | 0.18 | 0.02 | 0.14 | 0.01 | 0.12 | 0.01 | 0.13 | 0.01 | 0.31 | 0.01 | 0.36 | 0.01 | 0.35 | 0.01 | 0.23 | 0.06 | 0.03 | 0.04 | 0.03 |
| Trp | 0.17 | 0.51 | 0.16 | 0.40 | 0.13 | 0.25 | 0.03 | 0.14 | 0.06 | 0.63 | 0.05 | 0.60 | 0.11 | 0.80 | 0.11 | 0.55 | 0.07 | 0.22 | 0.04 | 0.45 |
| Others | 1.00 | 3.84 | 1.22 | 3.23 | 1.34 | 3.35 | 0.18 | 2.57 | 0.27 | 3.68 | 0.36 | 4.18 | 0.51 | 2.97 | 2.17 | 3.37 | 1.34 | 2.65 | 0.64 | 2.95 |
| Lys | 0.79 | 1.58 | 0.91 | 1.58 | 0.44 | 1.94 | 0.10 | 1.78 | 0.11 | 1.19 | 0.10 | 1.19 | 0.27 | 0.87 | 0.29 | 0.91 | 0.16 | 1.85 | 0.31 | 0.90 |
| Ser | 0.10 | 0.98 | 0.18 | 0.82 | 0.82 | 0.80 | 0.05 | 0.28 | 0.12 | 1.00 | 0.10 | 1.47 | 0.13 | 0.89 | 1.35 | 1.23 | 1.02 | 0.38 | 0.20 | 1.04 |
| Gly | 0.04 | 0.51 | 0.04 | 0.28 | 0.03 | 0.17 | 0.01 | 0.15 | 0.02 | 0.49 | 0.14 | 0.62 | 0.03 | 0.44 | 0.45 | 0.55 | 0.01 | 0,08 | 0.04 | 0.31 |
| His | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| Arg | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND | ND |
| Thr | 0.07 | 0.78 | 0.10 | 0.55 | 0.05 | 0.45 | 0.02 | 0.36 | 0.02 | 1.00 | 0.02 | 0.89 | 0.07 | 0.75 | 0.08 | 0.66 | 0.13 | 0.33 | 0.15 | 0.69 |
| Total | 4.61 | 13.7 | 5.97 | 11.7 | 3.87 | 10.8 | 5.34 | 9.05 | 3.18 | 13.1 | 2.33 | 16.9 | 4.18 | 12.7 | 4.95 | 13.8 | 3.96 | 9.61 | 4.70 | 11.1 |
Abbreviations. DF: Day of fermentation Por: Porcelana; Gua: Guasare; SJ: San Juan; IMC-67.: 1st season of harvest in year 1; IMC-67.: 2nd season of harvest in year 1; IMC-67.:3rd season of harvest in year 1; IMC-67.: 3rd season of harvest in year 2; ISC-1.: 1st season of harvest in year 1; ISC-1st : 1º season of harvest in year 2; ND: Not detected.
In Figure 5, total, hydrophobic, acidic, and others amino acids content versus fermentation and roasting time is shown.
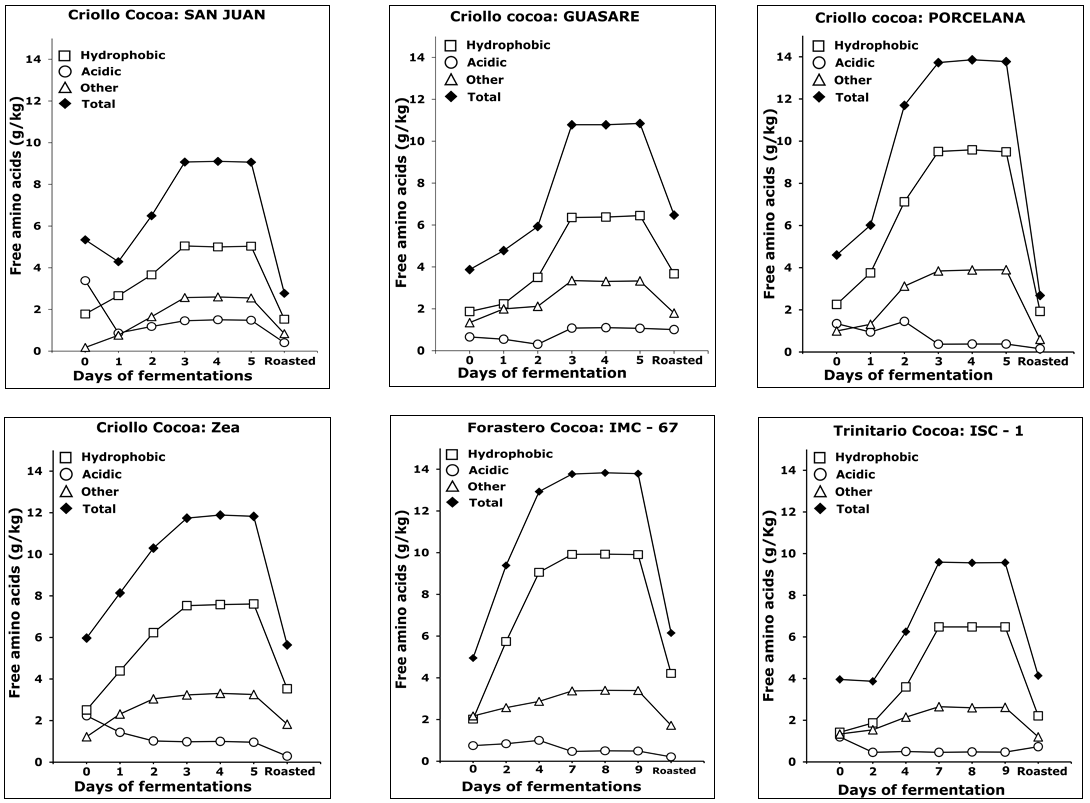
Figure 5. Trends of different group of amino acids in three types of cocoa (Criollo, Forastero and Trinitario) during fermentation and roasting.
As can be seen, a progressive increase of total free amino acids was observed during fermentation, with a maximum on the 3rd day for Criollos, and on the 7th day for Forasteros and Trinitarios, while a significant decrease during roasting is observed for all types. This trend agrees with previously reported studies in this matter [11, 12, 29], confirming that amino acids are enzymatically released from the storage proteins during fermentation. It is also noticeable that all cocoas, in the absence of fermentation, showed an appreciable content of amino acids. Thus, the ratio of hydrophobic to acidic free amino acids provides information of the fermentation degree, since it changes while fermentation advances. The hydrophobic amino acid content increases linearly with the fermentation time, derived from the proteolytic activity over major storage proteins in cocoa seeds. Particularly, hydrophobic amino acids such as leucine, phenylalanine, valine, alanine, and isoleucine showed the greatest increase (Table 2). Voigt et al. [2] studied the proteolytic formation of the cocoa-specific aroma precursors in vitro using the vicilin-class globulin (VCG) from cocoa seeds as protein substrate and different proteases. They reported that the specific mixture of oligopeptides and hydrophobic free amino acids are formed by degradation of the cocoa globulin because of the action of two enzymes: the endogenous aspartic endoprotease and the carboxypeptidase. The aspartic endoprotease (pH 3.5) hydrolyses the peptide bonds in proteins producing oligopeptides with hydrophobic amino acid residues at their carboxyl terminal. These are substrates for the carboxypeptidase (pH 5.8), which acts on terminals originating the free hydrophobic amino acids [3, 27, 30].
As was previously described, unfermented cocoa samples contained low amounts of free total amino acids, ranging between 2.33 and 5.97 g kg-1 (Table 2) and with a predominance of acidic amino acids, which represented 17 - 64%, 41 - 44%, and 30 - 38 % of the total amino acids content for Criollo, Forastero, and Trinitario, respectively. Variation of acidic, hydrophobic, and others amino acids during fermentation for all types of cocoa is detailed in Figure 6.
In contrast, fermented beans presented a higher amount of total free amino acids (9.05 - 16.9 g kg-1) with a significant increase of the hydrophobic amino acids content (56 - 72%) and a decrease of the acidic ones of more than 25%. For example, the cocoa beans of IMC-67 1st season, 1st year (Forastero) had an acid:hydrophobic:others amino acid ratio of 43:49:8 in unfermented beans and 9:63:28 in the fermented ones (Figure 6 b). This trend agrees with the results reported by other researchers [4, 11], which reported total free amino acid amounts within the range of 2.4 - 5.1 g kg-1 for unfermented cocoa beans with an acidic:hydrophopic:other ratio of 18:30:52, and 9.4 - 14.5 g kg-1 (6:46:48 and 58:16:26) for fermented cocoa beans from Malaysia and Ghana.
The relative differences between our results and the reported ones could be attributed to proteolytic activity, which depends on the conditions of the fermentation process, including the mass and aeration during fermentation with a direct influence over the pH and temperature of the process, as well as the maturity, harvesting season, variety, and origin of the beans [1, 4]. Figure 6a shows the amino acid composition in different varieties of Criollo cocoa (first and last day of fermentation). The variations in this composition can be justified by their genetic origin. Moreover, differences in the cocoa beans from Zea and San Juan, which are genetically equivalent, can be attributed to soil characteristics and climatic conditions during plant development since these were harvested in different locations [20]. A similar behavior is observed for Forastero IMC-67 samples that were collected in intervals of two crop cycles per year (Figure 6 b). Once again, conditions as climate, the amount of sunshine and rainfall, temperature, soil conditions, ripening, and post-harvesting practices directly affected the amino acids composition [31].
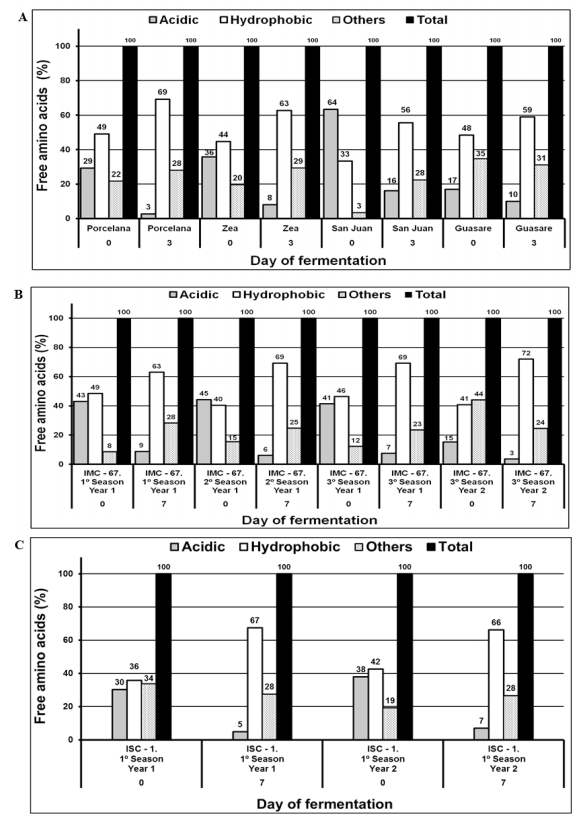
Figure 6. Composition (%) of amino acids on the first and last day of fermentation; a) Cocoa Criollo, b) Cocoa Forastero and c) Cocoa Trinitario.
Roasting of cocoa is an essential step for the chocolate flavor development. At this stage, the Maillard’s reaction takes place, involving free amino acids, peptides, and reducing sugars [32, 33]. Figure 5 also shows a significant decrease of free amino acids during roasting, being greater for the hydrophobic ones. Voigt et al. [2] reported that the hydrophobic amino acids leucine, alanine, phenylalanine, and tyrosine, released by proteinase activities in fermentation, are important contributors, as well as the reducing sugars fructose and glucose derived from sucrose hydrolysis [4, 34]. This behavior has been associated to a significant increase in pyrazines after roasting, which are the compounds responsible for cocoa flavor intensity.
In this research, a comparative study between the free amino acid profile (last day of fermentation and roasting) and the content of pyrazines obtained was conducted, according to the procedure reported by Brunetto et al [35]. Figure 7 shows the amino acids content generated during the fermentation and their decrease due to their transformation into pyrazines. Furthermore, among the analyzed Venezuelan cocoas, the amino acids behavior in Porcelana variety can be highlighted, since an important decrease of hydrophobic amino acids can be observed as well as a significant increase in pyrazine content during roasting stage, similar to the behavior observes on the Forastero variety. Several researchers confirm that free amino acids act as substrate in the formation of pyrazines [9, 36]. Therefore, pyrazines levels can be used as an index of the roasting process [37].
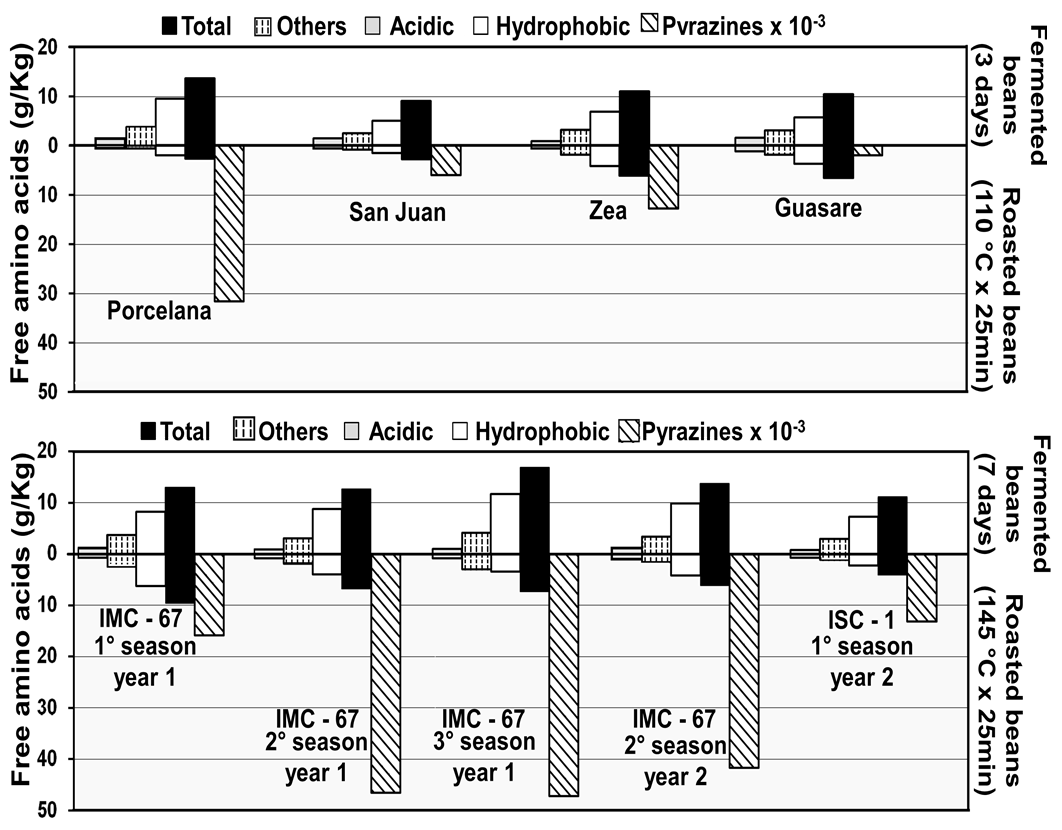
Figure 7. Effect of roasting over amino acids profile in different types of cocoa beans.
Conclusions
The obtained results have proved that the developed reversed-phase HPLC method with PTC-AA DADis a useful tool for primary and secondary amino acids determination in cocoa samples. Amino acids extraction from cocoa samples with water in an ultrasonic bath at 70 ºC provided a satisfactory extraction efficiency with recoveries higher than 89%. The pre-column amino acids derivatization time was significantly reduced using a domestic microwave oven. Besides, exploiting a column-switching system with an OASIS pre-column allowed the efficient and fast cleaning of PTC-AA extract, thereby reducing matrix interferences. Different validation parameters showed that with the developed method rapid and highly reproducible results can be obtained.
The proposed method was successfully applied for the evaluation of amino acids content in Trinitario, Forastero, and Criollo cocoa samples grown in different seasons of the year. All the analyzed samples had a significant content of free amino acids, ranging from 3.87 to 5.97 g kg-1 in the absence of fermentation. In general, amino acid degradation was observed on the first day of fermentation, whereas some samples showed progressive increase as the fermentation process carried on, with a predominance of hydrophobic amino acids such as leucine, phenylalanine, valine, alanine, and isoleucine. Moreover, during the roasting step a fraction of the amino acids, especially the hydrophobic ones, is partially degraded by the Maillard’s reaction to form the compounds associated with the aroma and flavor of cocoa.
References
[1] E.M. Afoakwa, A. Paterson, M. Fowler, and A. Ryan, “Flavor formation and character in cocoa and chocolate: A critical review”, Critical Reviews in Food Science and Nutrition, vol. 48, pp. 840–857, 2008. DOI: 10.1080/10408390701719272.
[2] J. Voigt, B. Biehl, H. Heinrichs, S. Kamaruddin, G. Marsoner, and A. Hugi, “In-vitro formation of cocoa specific aroma precursors: aroma-related peptides generated from cocoa seed protein by co-operation of an aspartic endoprotease and a carboxypeptidase”, Food Chemisty, vol. 49, pp. 173-80, 1994. DOI: https://doi.org/10.1016/0308-8146(94)90155-4
[3] J. Voigt and B. Biehl, “Precursors of the cocoa-specific aroma components are derived from the vicilin-class (7S) globulin of the cocoa seeds by proteolytic processing”, Botanical Acta, vol. 108, pp. 283-289, 1995. DOI: 10.1111/j.1438-8677.1995.tb00496.x
H. Puziah, S. Jinap, K.S.M. Sharifah, and A. Asbi, “Changes in free amino acid, peptide-N, sugar and pyrazine concentration during fermentation”, Journal of the Science Food and Agriculture, vol. 78, pp. 535-542, 1998. DOI: https://doi.org/10.1002/(SICI)1097-0010(199812)78:4<535::AID-JSFA151>3.0.CO;2-6.
[5] G.A. Reineccius, D.A. Andersen, T.E. Kavanagh, and P.G. Keeney, “Identification and quantification of the free sugars in cocoa beans”, Journal of Agricultural and Food Chemistry, vol. 20, n° 2, pp. 199-202, 1972. DOI: https://doi.org/10.1021/jf60180a033.
[6] L. Lima, H. Almeida, M. Nout, and M. Zwietering, “Theobroma cacao L: The food of the Gods. Quality determinants of comercial cocoa beans, with particular reference to the impact of fermentation”, Critical Reviews in Food Science and Nutrition, vol. 51, pp. 731-761, 2011. DOI: 10.1080/10408391003799913.
[7] N. Camu, T. De Winter, S. Addo, J. Takrama, H. Bernaert, and L. De Vuyst, “Fermentation of cocoa beans: influence of microbial activities and polyphenol concentration of the flavor of chocolate”, Journal of the Science of Food and Agriculture, vol. 88, pp. 2288-2297, 2008. DOI: 10.1002/jsfa.
[8] J. Serra Bonvehi, “Investigation of aromatic compounds in roasted cocoa power”, European Food Research and Technology, vol. 221, pp. 19-29, 2005. DOI: 10.1007/s00217-005-1147-y.
[9] E. Cros and N. Jeanjean, “Formation de l’arôme cacao. Cacao et chocolat. Production, utilisation, caractéristiques". J. Pontillon. Paris. Ed. Tec & Doc, 1998, pp. 188-206.
[10] T.A. Rohan, “Precursors of chocolate aroma: a comparative study of fermented and unfermented cocoa beans”, Journal of Food Science, vol. 29, n° 4, pp. 456-459, 1964. DOI: https://doi.org/10.1111/j.1365-2621.1964.tb01760.x.
[11] P.M. Kirchhoff, B. Biehl, and G. Crone, “Peculiarity of the accumulation of free amino acids during cocoa fermentation”, Food Chemistry, vol. 31, pp. 295-311, 1989.
[12] P.M. Kirchhoff, B. Biehl, H. Ziegeler-Berghausen, M. Hammoor, and R. Lieberei, "Kinetics of the formation of free amino acids in cocoa seeds during fermentation”, Food Chemistry, vol. 34, pp. 161-179, 1989.
[13] E. Portillo, L. Graziani de Fariñasand E. Bentancourt, “Chemical analysis of Criollo Porcelana Cocoa (Theobroma cocoa L.) in the South of Maracaibo Lake”, Revista Facultad de Agronomía Universidad del Zulia, Venezuela, vol. 24, pp. 522-546, 2007.
[14] E. Portillo, D. Fuenmayor, B. Rodríguez, and R. Díaz, “Effect of the fermentation on the content of purines in Criollo and Hybrid cocoa (Theobroma cocoa L) in the south of Maracaibo´s lake”, Revista Facultad de Agronomía Universidad del Zulia, Venezuela, vol. 28, n°1, pp. 228-237, 2011.
[15] L. Graziani de Fariñas, L. Ortiz de Bertorelli, M. Lemus, and P. Parra, “Effect of blending two types of cocoa seeds on chemical properties during fermentation”, Agronomía Tropical, vol. 52, n° 3, pp. 325-342, 2002.
[16] L. Graziani de Fariñas, L. Ortiz de Bertorelli, and P. Parra, “Chemical characteristics of seeds of different types of cocoa from the locality of Cumboto, Aragua”, Agronomía Tropical, vol. 53, n°2, pp. 133-144, 2003.
[17] L. Ortiz de Bertorelli, G. Camacho, and L. Graziani de Fariñas, “Effect of sun drying on the quality of fermented cocoa beans”, Agronomía Tropical, vol. 54, n°1, pp. 31-43, 2004.
[18] L. Ortiz de Bertorelli, L. Graziani de Fariñas, and R.L. Gervaine, “Evaluation of some chemical characteristics of cocoa beans in fermentation”, Agronomía Tropical, vol. 51, n°1, pp. 73-79, 2009.
[19] L. Ortiz de Bertorelli, L. Graziani de Fariñas, and R.L. Gervaine, “Some factors affecting characteristics of fermented and sun dried cocoa grain”, Agronomía Tropical, vol. 59, n°2, pp. 119-127, 2009.
[20] A. Zambrano, et al., “Chemical evaluation of aroma and flavor of precursors of the Criollo Merideño cocoa during fermentation in two edaphoclimatic conditions”, Agronomía Tropical, vol. 60, n° 2, pp. 211-219, 2010.
[21] M.R. Brunetto, et al., “Determination of theobromine, theophylline and caffeine in cocoa samples by high-performance liquid chromatographic method with on-line simple cleanup in a switching-column system”, Food Chemistry, vol. 100, pp. 459-467, 2007. DOI: 10.1016/j.foodchem.2005.10.007.
[22] C. Álvarez, E. Pérez and M.C. “Physical-chemical characterization of fermented, dried and roasted cocoa beans cultivated in the region of Cuyagua, Aragua State”, Agronomía Tropical, vol. 57, n°4, pp. 249-256, 2007.
[23] International Office of Cocoa, Chocolate and Sugar Confectionary (IOCCC). 1996. “Determination of the Fat Content of Cocoa Powder by Soxhlet Extraction”, IOCCC. Analytical Method N° 37.
[24] B.A. Bidlingmeyer, S.A. Cohen, and T.L. Tarvin, “Rapid analysis of amino acids using pre-column derivatization”, Journal of Chromatography, vol. 336, pp. 93-104, 1984. DOI: https://doi.org/10.1016/S0378-4347(00)85133-6.
[25] M. Hariharan, S. Naga, and T. VanNoord, “Systematic approach to the development of plasma amino acid analysis by high-performance liquid chromatography with ultraviolet detection with precolumn derivatization using phenyl isothiocyanate”, Journal of Chromatography, vol. 621, pp. 15-22, 1993. DOI: https://doi.org/10.1016/0378-4347(93)80071-B
[26] L. Zhang, et al., “One single amino acid for estimation the content of total free amino acids in Qingkailinginjection using high-performance liquid chromatography-diode array detection” Journal of Analytical Methods in Chemistry, 2014. DOI: https://doi.org/10.1155/2014/951075.
[27] L. Chen, D. Song, Y. Tian, L. Ding, A. Yu, and H. Zhang. Application of on-line microwave sample-preparation techniques. Trends Anal. Chem. Vol. 27, pp. 151–159, 2008. DOI: 10.1016/j.trac.2008.01.003.
[28] Oasis Applications Notebook. Waters Corporation, 2003. https://www.waters.com/waters/library.htm?lid=1528415&cid=511436&locale=en_US.
[29] S. Jinap, H.N. Lioe, I. Yusep, S. Nazamid, and B. Jamilah, “Role of carboxypeptidases to the free amino acid composition, methylpyrazine formation and sensory characteristic of underfermented cocoa beans”, International Food Research Journal, vol. 17, pp. 763-774, 2010.
[30] G. Bytof, B. Biehl, H. Heinrichs, and J. Voigt, “Specificity and stability of the carboxypeptidase activity in ripe, ungerminated seeds of Theobroma cacao L”, Food Chemistry, vol. 54, n°1, pp. 15-21, 1995. DOI: https://doi.org/10.1016/0308-8146(95)92657-6.
[31] A. Oyekale, M. Bolaji, and O. Olowa, “The effect of climate change in cocoa production and vulnerability assessment in Nigeria”, Agricultural Journal, vol. 4, n°2, pp. 77-85, 2009. DOI: aj.2009.77.85.
[32] T.A. Rohan and T. Stewart, “The precursors of chocolate aroma: changes in the free amino acids during the roasting of cocoa beans”, Journal of Food Science, vol. 31, n°2, pp. 202-205, 1966. DOI: https://doi.org/10.1111/j.1365-2621.1966.tb00479.x.
[33] T.A. Rohan and T. Stewart, “The precursors of chocolate aroma: production of free amino acids during fermentation cocoa beans”, Journal of Food Science, vol. 32, pp. 395-398, 1967. DOI: https://doi.org/10.1111/j.1365-2621.1967.tb09693.x.
[34] T.A. Rohan and T. Stewart, “The Precursors of Chocolate Aroma: changes in the sugars during the roasting of cocoa beans”, Journal of Food Science, vol. 31, n°2, pp. 206-209, 1966. DOI: https://doi.org/10.1111/j.1365-2621.1966.tb00480.x.
[35] M.R. Brunetto, et al., “Headspace gas chromatography-mass spectrometry determination of alkylpyrazines in cocoa liquor samples”, Food Chemistry, vol. 112, pp. 253-257, 2009. DOI: 10.1016/j.foodchem.2008.05.082.
[36] E. Portillo, et al., “Aroma formation of Criollo cocoa (Theobroma cacao L.) in function of the post-harvest treatment in Venezuela”, Revista Agrícola de la Universidad de Oriente, Venezuela, vol. 9, n°2, pp. 458-468, 2009.
S. Jinap, W. I. Wan Rosli, A.R. Russly, and L.M. Nordin, “Effect of roasting time and temperature on volatile component profile during nib roasting of cocoa beans (Theobroma cacao)”, Journal of the Science of Food and Agriculture, vol. 77, pp. 441-448, 1998. DOI: https://doi.org/10.1002/(SICI)1097-0010(199808)77:4<441::AID-JSFA46>3.0.CO;2-%23.
Notas de autor
mariadelrosario2327@gmail.com
Recibido: 11 de febrero de 2019; Aceptado: 9 de octubre de 2019
Abstract
Amino acids (AA) composition in cocoa beans can predict the synthesis of compounds which affect cocoa flavor. Thus, their determination is of great interest for the community implied in the commercialization and production of cocoa. In consequence, in this work, the analysis of AA produced during cocoa beans fermentation and roasting was carried out. A high-performance liquid chromatographic method with DAD detection at 254 nm was optimized and validated for their selective determination in six varieties of cocoa beans with different genotypes, all of them grown in Venezuela. AA were extracted by defatted milled cocoa powder ultrasonication using purified water at 70 ºC. Then, they were derivatized with phenyl isothiocyanate, and their derivatives were separated, using a reversed-phase column with gradient elution, achieving a satisfactory resolution among the peaks (greater than 1.0) in less than 29 min. 110 cocoa samples were analyzed. Results showed a significant content of free AA, ranging from 3.87 to 5.97 g/kg in absence of fermentation with a predominance of acidic AA. Moreover, there is a progressive increase in the AA content while fermentation process occurs, with a predominance of hydrophobic AA such as alanine, valine, isoleucine, leucine, phenylalanine, and tyrosine. On the other hand, all cocoa types showed a partial degradation of free AA during the roasting step, especially the hydrophobic ones.
Keywords
liquid chromatography, column-switching, derivatization, microwave radiation, cocoa.Resumen
La determinación de aminoácidos (AA) en granos de cacao es de gran interés ya que estos son considerados como unos de los precursores de su sabor y aroma. Por esta razón, el presente trabajo tuvo como objetivo optimizar y validar un método por cromatografía líquida con detección DAD a 254 nm para la determinación selectiva de AA durante la fermentación y tostado en seis variedades de granos de cacao con diferentes genotipos, todos estos cultivados en Venezuela. Los AA se extrajeron del polvo de cacao molido y desgrasado con agua pura a 70 ºC, utilizando la técnica de ultrasonido. Luego, se derivatizaron con fenilotiocianato para separar sus derivados con buena resolución en menos de 29 min en una columna de fase reversa, utilizando gradiente de elución. Se analizaron 110 muestras de cacao. Los resultados mostraron un contenido significativo de AA libres, entre 3,87 y 5,97 g/kg, en ausencia de fermentación con predominio de AA ácidos, y un aumento progresivo en el contenido de AA, mientras ocurre el proceso de fermentación, con un predominio de AA hidrófobos como alanina, valina, isoleucina, leucina, fenilalanina y tirosina. Además, todos los tipos de cacao mostraron una degradación parcial de AA libres durante la etapa de tostado, especialmente los AA hidrófobos.
Palabras clave
cromatografía líquida, sistemas de columnas acopladas, derivatización, radiación por microondas, cacao.Resumo
A determinação dos aminoácidos (AA) nos grãos de cacau é importante, pois são considerados um dos precursores de seu sabor e aroma. Neste trabalho, um método foi otimizado e validado por cromatografia líquida com detecção DAD a 254 nm para a determinação seletiva de AA durante a fermentação e torrefação em seis variedades de grãos de cacau com diferentes genótipos, todos cultivados na Venezuela. Os AAs foram extraídos do pó de cacau moído e desengordurados com água pura a 70 ° C usando a técnica de ultrassom. Em seguida, foram derivatizados com feniltiocianato, e os derivados foram separados com boa resolução em menos de 29 minutos em uma coluna de fase invertida usando eluição em gradiente. Foram analisadas 110 amostras de cacau. Os resultados mostraram um conteúdo significativo de AA livre entre 3,87 e 5,97 g/kg na ausência de fermentação com predominância de AA ácidos e um aumento progressivo no conteúdo de AA enquanto o processo de fermentação ocorre com predominância de AA hidrófobos como alanina, valina, isoleucina, leucina, fenilalanina e tirosina. Além disso, todos os tipos de cacau apresentaram uma degradação parcial do AA livre durante a fase de torrefação, principalmente o AA hidrofóbico.
Palavras-chave
cromatografia líquida, sistemas de colunas acopladas, derivatização, radiação de microondas, cacau.Introduction
Cocoa beans are originated as seeds in fruit pods of the tropical crop Theobroma cacao L., that is cultivated in the humid lowland tropics generally by small-scale producers. There are three main cocoa types: Forastero (bulk grade), Criollo (fine grade), and hybrid Trinitario (fine grade). These varieties offer wide differences in their final flavor due to factors such as their genotype, soil, climate, and harvest conditions, and to the post-harvest technology used in fermentation, drying, and roasting stages [1].
Fermentation plays a major role in the development of aroma precursors like free amino acids, peptides, and reducing sugars, due to the microbial activity and endogenous enzymes of cocoa beans. Free amino acids and peptides are formed by proteolytic reactions induced by endogenous proteases (aspartic proteinase and carboxypeptidase) during cocoa beans storage [2, 3]. Contrarily, reducing sugars such as fructose and glucose are products of the hydrolysis of sucrose [4, 5]. Both amino acids and reducing sugars are precursors in Maillard’s reaction. Thus, they are responsible of the formation of the compounds which affect cocoa flavor during roasting [6, 7, 8, 9]. Therefore, determination of the amino acid profile is extremely important to assess the cocoa nutritional value as well as to predict the development of flavor compounds during the manufacture of cocoa-based products. An optimum fermentation originates good flavor and aroma, while an inadequate process might considerably demerit the product [4, 10]. Voigt et al. [2, 3] concluded that peptides and hydrophobic amino acids are important cocoa-specific flavor precursors. On the other hand, Kirchhoff et al. [11, 12] pointed out a correlation between the accumulation of free amino acids and the generation of essential aroma precursors, also noticed that they are formed by pH-dependent proteolysis processes. Moreover, Afoakwa et al. [1] observed that the nature of the amino components is critical for cocoa flavor due to their influence in the formation of heterocyclic compounds which affect the resulting aroma.
Therefore, to evaluate the cocoa quality is mandatory to study not only the compounds directly related with the flavor, but also their precursors. Thus, the analysis of amino acids during fermentation, drying and roasting stages could provide useful information to improve the post-harvest treatments and obtain a high-quality cocoa.
Venezuela is considered a producer of fine cocoa with excellent quality and a characteristic flavor. In recent years, measures have been implemented to increase production and to improve the economic competitiveness of cocoa. Several researchers [13-22] have been focused on the study of the chemical and physical parameters involved in the fermentation and drying processes, with the aim of improving post-harvest processes to increase the cocoa quality, especially of the Criollo variety. However, amino acids behavior during the different post-harvest stages has not been studied, although they are essential for cocoa flavor development.
Therefore, the main objective of this work was the determination of total and free amino acids content in Venezuelan cocoa during the fermentation and roasting stages. For this purpose, a high-performance liquid chromatographic method with DAD detection was optimized and validated.
Materials and methods
Reagents, solvents and standards
All solvents were of HPLC grade and all reagents were of analytical grade, unless indicated otherwise. Ethanol (EtOH) and acetonitrile (MeCN) were purchased from J.T. Baker (Phillipsburg, NJ, USA). All amino acids were obtained from Sigma-Aldrich (St. Louis, MO, USA). Phenylisothiocyanate (PITC) and triethylamine (TEA) (99% w/w) were obtained from Sigma-Aldrich. Milli-Q ultrapure water with a specific resistivity of 18 Mcm−1 was used for standards and samples preparation, reagents dilution and for cleaning purposes.
Acetate buffer was prepared with sodium acetate solution 0.07 mol L-1 and 99.7% w/w acetic acid from Fisher obtaining a final concentration of 0.05 mol L-1 and a pH=5.1. Individual stock standard solutions with 10000 µg mL-1 of each amino acid were prepared in reagent-grade water. All solutions were stored under refrigeration at 4 ºC. Working solutions were weekly prepared by dilution of stock standard solutions in water.
Cocoa samples
To study the applicability of the proposed method, Criollo, Trinitario, and Forastero cocoa beans were fermented (1-7 days) and then roasted prior to their analysis.
Cocoa pods of the varieties ICS-1 (Trinitario) and IMC-67 (Forastero) were obtained from genetically identified trees grown on the Cocoa Genebank of Ocumare de la Costa (INIA-Aragua, Venezuela). Criollo Guasare, Zea, and San Juan were provided by the Cocoa Genebank of San Juan de Lagunillas (INIA-Mérida, Venezuela), and Porcelana was obtained from the Cocoa Genebank of the Local Chama Station (Corpozulia, Venezuela).
Samples were processed as follows: cocoa seeds were subjected to fermentation process according to their variety. This process was performed in triplicate into 60 mLwooden boxes. The fermenting mass was turned over every 24 h. Each day three replicates were collected and analyzed. After fermentation, a drying process was performed for 6 days. Then, some of the samples were grounded, sieved (≤ 42 mesh), and defatted, according to the Soxhlet extraction method [23]. These were stored at -20 °C until analysis. Besides, with the aim of studying the roasting effect on the amino acids and reducing sugars profile, some fermented and dried samples were first roasted and then grounded and defatted. All samples were conserved in sealed PTFE containers, which were kept under refrigeration (4 °C) until analysis.
Extraction and derivatization of the amino acids
To carry out the total free amino acids extraction, 0.800 g of defatted cocoa powder were ultrasonicated for 15 min at 70 ºC in 15 mL of purified water. The obtained extract was cooled to room temperature and centrifuged at 3500 rpm for 15 min. Then the supernatant was transferred to a 25 mL volumetric flask filled with water and then filtered through a 0.22 μm membrane (Membrane support-220, Waters, USA).
Free amino acids were derivatized with PITC, following the method described by Bidlingmeyer et al. [24] with some modifications. Thus, 20 µL of sample were placed into a vial and mixed with 20 µL of ethanol and 20 µL of the derivative reagent solution (1:1:7 v/v/v PITC:TEA:Ethanol ). Then, the vial was capped and exposed to microwave radiation in a domestic microwave oven (340 W for 40 s). Finally, 20 µL of the PTC-AA extract were injected into the HPLC system.
Amino acid derivatives analysis
After extraction and derivatization procedures, amino acid derivatives (PTC-AA) were present in a complex mixture, making necessary a sample cleanup to obtain chromatograms with good resolution.
A column-switching HPLC system in the circuit transfer mode was used for sample cleanup and PTC-AA separation and detection. The chromatographic system was composed by a quaternary pump, a column oven, an autosampler, a diode array detector (DAD) (Series 200 HPLC System, PerkinElmer; Norwalk, CT), and a digital workstation with TotalChrom software (Version 6.3) which was used as controller and data manager. Two additional Waters pumps (model 515) (Milford, MA, USA, Pump 1, Pump 2) were used to propel the extraction and transfer mobile phases. Injections were made with a Rheodyne type 7125 six-port valve equipped with a 100 μL loop. Two column-switching six-port valves (Supelco, Bellefonte, PA, USA; SV 1, SV 2) were attached to the system and controlled by the workstation. Three columns were used in the system: 1) Precolumn 1: Oasis HLB column 20 x 3.9 mm; dp (particle diameter): 25 μm, used as extraction column; 2) Precolumn 2: Supelcosil™ LC-18 20 x 3 mm; dp: 5 µm, which replaced the loop injection previous to the analytical column; 3) Advantage Kromasil C18 column 150 x 3.0 mm i.d.; dp: 3 μm, used as analytical column. Figure 1 shows a schematic depiction of the column-switching system.
Figure 1: Schematic diagram of the columnswitching HPLC system The direction of flow is indicated by arrows a Sample charging precolumn 1 SV 1 load position SV 2 inject position b Sample charging precolumn 2 SV 1 inject position SV 2 load position c Final elution and injection into the HPLC system SV 1 load position SV 2 inject position
Figure 1. Schematic diagram of the column-switching HPLC system. The direction of flow is indicated by arrows. a. Sample charging precolumn 1: SV 1: load position, SV 2: inject position; b. Sample charging precolumn 2: SV 1: inject position, SV 2: load position; c. Final elution and injection into the HPLC system: SV 1: load position, SV 2: inject position.
Analysis procedure
With SV 1 in load position (Figure 1a), 100 µL of PTC-AA extract were propelled by pump 1 at a flow-rate of 1 mL min-1, injected into the extraction column (precolumn 1), and washed with 2% acetonitrile in acetate buffer. PTC-AA were retained while endogenous sample components were eluted to waste. After 0.5 min, SV 1 was automatically rotated to inject position (Figure 1b) allowing pump 2 to propel the mobile phase (90% ethanol in acetate buffer) at 1.8 mL min-1. This way, enriched PTC-AA were eluted from extraction column and injected into precolumn 2.
After 0.13 min, SV 2 was changed to inject position and PCT-AA extract with 2% acetonitrile in acetate buffer was pushed by the pump 3 at 1.7 mL min-1 into the analytical column to perform the analytes separation. Simultaneously, SV 1 was changed to load position to carry out the re-equilibration of the extraction column (precolumn 1) before the next injection (Figure 1c).
Analytes separation was performed at 45 °C using two different mobile phases, mobile phase A: 2% acetonitrile in acetate buffer and mobile phase B: acetonitrile-water (60:40 v/v) in a gradient mode elution at flow rate of 1.7 mL min-1 and with the following program: 0 % solvent B (100% A) (5 min) to 35% B at 1.5 %B min-1 and up to 100% B (3 min) at 32.5 %B min-1.
A summary of the system operating conditions is shown in Table 1. Under these conditions, analytes were separated in less than 29 min, with good resolution and no matrix effect. A chromatogram of the separation of the AA derivatives is shown in Figure 2.
Table 1. Optimized experimental conditions of the procedure.
Table 1: Optimized experimental conditions of the procedure
Step
System component
Conditions
Sample injection (t = 0 min)
Rheodyne 7125 valve: inject position Switching-valve 1: load position Switching-valve 2: inject position
Injection volume: 100 µL
Sample extraction (t =0.50 min)
Extraction precolumn 1:Oasis HLB (20 x 3.9 mm; dp: 25 μm)
Mobile phase: 2% acetonitrile in 0.05 mol L-1 acetate buffer (pH= 5.1) Flow rate: 1.0 mL min-1
Analytes injection into Precolumn 2 (t =0.13 min)
Precolumn 2: Supelcosil LC-18 (20 x 3 mm; dp: 5 µm) Switching-valve 1: inject position Switching-valve 2: load position
Mobile phase: 90% ethanol in 0.05mol L-1 acetate buffer (pH= 5.1) Flow rate: 1.8 mL min-1
Analytes injection into the analytical column Precolumn 1: re-conditioning before next injection
Advantage Kromasil C18 100A (150mm x 3.0 mm; 3µm) Switching-valve 1: load position Switching-valve 2: inject position
Solvent A: 2% actonitrile in 0.05mol L-1 acetate buffer (pH= 5.1) Flow rate: 1.7 mL min-1 Mobile phase: 2% acetonitrile in 0.05 mol L-1 acetate buffer (pH= 5.1) Flow rate: 1.0 mL min-1
Chromatographic separation (t = 29 min)
Switching-valve 2: inject position
Solvent A: 2% actonitrile in 0.05 mo L-1 acetate buffer (pH= 5.1) Solvent B: 60% acetonitrile in water Gradient program: 0 % solvent B (100% A) (5 min) to 35% B at 1.5 B% min-1, and up to 100% B (3 min) at 32.5 B% min-1 Flow- ate: 1.7 mL min-1 Detection: DAD a 254nm
Results and discussion
Microwave assisted derivatization optimization
PITC reacts with primary and secondary AA [25, 26]. The reaction occurred at room temperature and the best results were obtained with a reaction time of 15 min.
Figure 2: Chromatogram for PTCAA separation obtained under the optimized procedure for a standard solution of AA at concentration of 10 µg mL1 experimental conditions in Table 1
Figure 2. Chromatogram for PTC-AA separation obtained under the optimized procedure for a standard solution of AA at concentration of 10 µg mL-1 (experimental conditions in Table 1).
Aiming to decrease the analysis time, microwave radiation was used to assist the reaction [27]. Parameters such as microwave power and reaction time should be studied when this strategy is used. Therefore, time reaction was studied for two AA (glutamic acid and threonine) while maintaining microwave power at 340 W. The obtained results are shown in Figure 3.
Figure 3: Effect of the irradiation time at microwave power of 340 W
Figure 3. Effect of the irradiation time at microwave power of 340 W.
As can be seen, irradiation time and peak area increased proportionally until 40 s. In consequence, 40 s was set as optimum derivatization time. Additionally, in Figure 4 a comparison between the derivatization reaction performance under standard conditions and assisted by microwave irradiation is presented. Although a similar efficiency is obtained with both methods, the reaction time is drastically decreased by using the microwave (from 15 min to 40 s).
Figure 3: Effect of the irradiation time at microwave power of 340 W
Figure 4. Comparison of derivatization performance at standard conditions and assisted by microwave irradiation.
Solid phase extraction
A Waters Oasis HLB precolumn was used for sample clean up and analytes extraction and preconcentration. This column is a universal polymeric reversed phase sorbent which allows to obtain quantitative and reproducible recoveries of the analytes due to the presence of a balanced composition of two monomers: .-vinilpirrolidone (hydrophilic) and divinylbencene (hydrophobic) [28]. The retention mechanisms are dipole-dipole interactions and Van der Waals forces for polar and non-polar compounds, respectively. Experimental parameters were optimized to achieve the maximum efficiency during extraction. The obtained results can be seen in Table 1.
Method validation
Accuracy, precision, linearity and matrix effect studies were carried out to validate the proposed method. The accuracy of the procedure was evaluated by performing recovery tests using spiked cocoa samples at different amino acid concentration levels. In all cases, recovery values ranged between 89% and 109%, with a relative standard deviation (%RSD) lower than 3.85% (. = 5), which indicates an excellent efficiency for the amino acids extraction and derivatization procedures.
The within-day and between-day precisions, expressed as %RSD, were established at three different amino acid concentration levels (low, middle and high) for aqueous standard solutions and for spiked cocoa samples. RSD < 2.9% (. = 5) were obtained for all the analytes, confirming the satisfactory precision of the proposed procedure.
Calibration curves for each AA were obtained from both standard aqueous solutions and spiked cocoa samples (standard additions) in a wide range of concentration (0.1 - 15.0 µg mL-1). Each calibration set included seven data points and each point was run at least three times. Good linearity between peak area and concentrations was achieved with correlation coefficients R. > 0.9900 in all cases. Sample matrix effect was studied by comparing the slopes of the calibration curves of the standard aqueous solutions with the slopes obtained when analyzing the spiked samples. A paired student t-test indicated that the slopes were not statistically different (P < 0.05), which denoted the absence of any matrix effect. Limits of detection, defined as a signal/noise ratio of 3:1, were between 0.04 and 0.20 µg mL-1 for all amino acids.
Free amino acids content in cocoa samples
Results of the free amino acid contents in the analyzed cocoa samples are shown in Table 2.
Table 2. Free amino acids content (g/kg) for unfermented and fermented cocoa samples.
Table 2: Free amino acids content gkg for unfermented and fermented cocoa samples
DF
0
3
0
3
0
3
0
3
0
7
0
7
0
7
0
7
0
7
0
7
Amino acids (g/kg)
Por
Por
Zea
Zea
Gua
Gua
SJ
SJ
IMC-67a
IMC-67a
IMC-67b
IMC-67b
IMC-67c
IMC-67c
IMC-67d
IMC-67d
ISC – 1a
ISC – 1a
ISC – 1b
ISC – 1b
Acidic
1.35
0.37
2.23
0.98
0.66
1.08
3.38
1.46
1.37
1.15
1.03
1.02
1.79
0.94
0.75
0.47
1.20
0.46
1.26
0.79
Asp
0.56
0.26
0.56
0.15
0.30
0.59
1.23
0.46
0.58
0.17
0.27
0.27
0.50
0.18
0.22
0.12
0.42
0.17
0,51
0.24
Glu
0.79
0.12
1.67
0.74
0.36
0.49
2.15
1.00
0.79
0.97
0.76
0.75
1.23
0.76
0.53
0.36
0.75
0.28
0.75
0.53
Hydro phobic
2.26
9.51
2.51
7.53
1.87
6.36
1.78
5.02
1.54
8.22
0.94
11.71
1.93
8.77
2.02
9.92
1.42
6.48
1.42
7.32
Leu
0.29
1.85
0.45
1.55
0.20
1.50
0.11
1.04
0.13
2.26
0.10
2.66
0.19
1.83
0.22
2.21
0,08
1.66
0.17
1.30
Ala
0.33
1.76
0.51
1.67
0.17
0.47
0.11
0.89
0.19
0.65
0.13
1.12
0.29
1.22
0.32
1.77
0,11
0.89
0.26
0.79
Phe
0.41
1.15
0.43
1.00
0.28
1.10
0.06
0.83
0.16
0.81
0.12
1.51
0.27
1.18
0.10
1.40
0,10
1.08
0.18
1.47
Tyr
0.24
0.32
0.12
0.42
0.48
0.82
0.17
0.58
0.32
0.49
0.17
0.99
0.28
0.77
0.49
0.63
0.42
0.77
0.07
1.05
Val
0.14
1.30
0.23
0.47
0.10
0.46
0.10
0.34
0.08
0.59
0.06
1.36
0.12
0.53
0.12
1.01
0.06
0.36
0.08
0.45
Ile
0.16
1.09
0.31
0.80
0.14
0.46
0.06
0.29
0.10
1.27
0.06
1.44
0.13
0.60
0.15
0.80
0.09
0.32
0.08
0.56
Pro
0.52
1.34
0.29
1.08
0.37
1.19
1.14
0.79
0.50
1.21
0.25
1.67
0.50
1.45
0.46
1.28
0.46
1.13
0.51
1.17
Met
0.01
0.18
0.02
0.14
0.01
0.12
0.01
0.13
0.01
0.31
0.01
0.36
0.01
0.35
0.01
0.23
0.06
0.03
0.04
0.03
Trp
0.17
0.51
0.16
0.40
0.13
0.25
0.03
0.14
0.06
0.63
0.05
0.60
0.11
0.80
0.11
0.55
0.07
0.22
0.04
0.45
Others
1.00
3.84
1.22
3.23
1.34
3.35
0.18
2.57
0.27
3.68
0.36
4.18
0.51
2.97
2.17
3.37
1.34
2.65
0.64
2.95
Lys
0.79
1.58
0.91
1.58
0.44
1.94
0.10
1.78
0.11
1.19
0.10
1.19
0.27
0.87
0.29
0.91
0.16
1.85
0.31
0.90
Ser
0.10
0.98
0.18
0.82
0.82
0.80
0.05
0.28
0.12
1.00
0.10
1.47
0.13
0.89
1.35
1.23
1.02
0.38
0.20
1.04
Gly
0.04
0.51
0.04
0.28
0.03
0.17
0.01
0.15
0.02
0.49
0.14
0.62
0.03
0.44
0.45
0.55
0.01
0,08
0.04
0.31
His
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
Arg
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
ND
Thr
0.07
0.78
0.10
0.55
0.05
0.45
0.02
0.36
0.02
1.00
0.02
0.89
0.07
0.75
0.08
0.66
0.13
0.33
0.15
0.69
Total
4.61
13.7
5.97
11.7
3.87
10.8
5.34
9.05
3.18
13.1
2.33
16.9
4.18
12.7
4.95
13.8
3.96
9.61
4.70
11.1
Abbreviations. DF: Day of fermentation Por: Porcelana; Gua: Guasare; SJ: San Juan; IMC-67.: 1st season of harvest in year 1; IMC-67.: 2nd season of harvest in year 1; IMC-67.:3rd season of harvest in year 1; IMC-67.: 3rd season of harvest in year 2; ISC-1.: 1st season of harvest in year 1; ISC-1st : 1º season of harvest in year 2; ND: Not detected.
In Figure 5, total, hydrophobic, acidic, and others amino acids content versus fermentation and roasting time is shown.
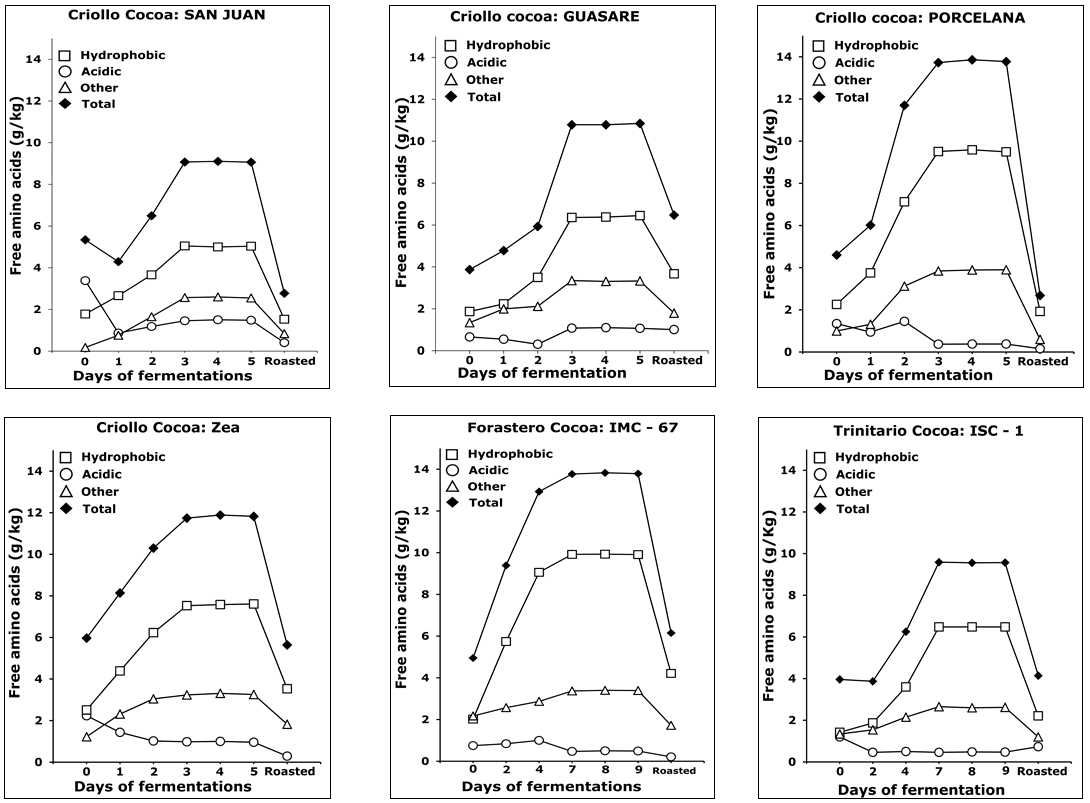
Figure 5. Trends of different group of amino acids in three types of cocoa (Criollo, Forastero and Trinitario) during fermentation and roasting.
As can be seen, a progressive increase of total free amino acids was observed during fermentation, with a maximum on the 3rd day for Criollos, and on the 7th day for Forasteros and Trinitarios, while a significant decrease during roasting is observed for all types. This trend agrees with previously reported studies in this matter [11, 12, 29], confirming that amino acids are enzymatically released from the storage proteins during fermentation. It is also noticeable that all cocoas, in the absence of fermentation, showed an appreciable content of amino acids. Thus, the ratio of hydrophobic to acidic free amino acids provides information of the fermentation degree, since it changes while fermentation advances. The hydrophobic amino acid content increases linearly with the fermentation time, derived from the proteolytic activity over major storage proteins in cocoa seeds. Particularly, hydrophobic amino acids such as leucine, phenylalanine, valine, alanine, and isoleucine showed the greatest increase (Table 2). Voigt et al. [2] studied the proteolytic formation of the cocoa-specific aroma precursors in vitro using the vicilin-class globulin (VCG) from cocoa seeds as protein substrate and different proteases. They reported that the specific mixture of oligopeptides and hydrophobic free amino acids are formed by degradation of the cocoa globulin because of the action of two enzymes: the endogenous aspartic endoprotease and the carboxypeptidase. The aspartic endoprotease (pH 3.5) hydrolyses the peptide bonds in proteins producing oligopeptides with hydrophobic amino acid residues at their carboxyl terminal. These are substrates for the carboxypeptidase (pH 5.8), which acts on terminals originating the free hydrophobic amino acids [3, 27, 30].
As was previously described, unfermented cocoa samples contained low amounts of free total amino acids, ranging between 2.33 and 5.97 g kg-1 (Table 2) and with a predominance of acidic amino acids, which represented 17 - 64%, 41 - 44%, and 30 - 38 % of the total amino acids content for Criollo, Forastero, and Trinitario, respectively. Variation of acidic, hydrophobic, and others amino acids during fermentation for all types of cocoa is detailed in Figure 6.
In contrast, fermented beans presented a higher amount of total free amino acids (9.05 - 16.9 g kg-1) with a significant increase of the hydrophobic amino acids content (56 - 72%) and a decrease of the acidic ones of more than 25%. For example, the cocoa beans of IMC-67 1st season, 1st year (Forastero) had an acid:hydrophobic:others amino acid ratio of 43:49:8 in unfermented beans and 9:63:28 in the fermented ones (Figure 6 b). This trend agrees with the results reported by other researchers [4, 11], which reported total free amino acid amounts within the range of 2.4 - 5.1 g kg-1 for unfermented cocoa beans with an acidic:hydrophopic:other ratio of 18:30:52, and 9.4 - 14.5 g kg-1 (6:46:48 and 58:16:26) for fermented cocoa beans from Malaysia and Ghana.
The relative differences between our results and the reported ones could be attributed to proteolytic activity, which depends on the conditions of the fermentation process, including the mass and aeration during fermentation with a direct influence over the pH and temperature of the process, as well as the maturity, harvesting season, variety, and origin of the beans [1, 4]. Figure 6a shows the amino acid composition in different varieties of Criollo cocoa (first and last day of fermentation). The variations in this composition can be justified by their genetic origin. Moreover, differences in the cocoa beans from Zea and San Juan, which are genetically equivalent, can be attributed to soil characteristics and climatic conditions during plant development since these were harvested in different locations [20]. A similar behavior is observed for Forastero IMC-67 samples that were collected in intervals of two crop cycles per year (Figure 6 b). Once again, conditions as climate, the amount of sunshine and rainfall, temperature, soil conditions, ripening, and post-harvesting practices directly affected the amino acids composition [31].
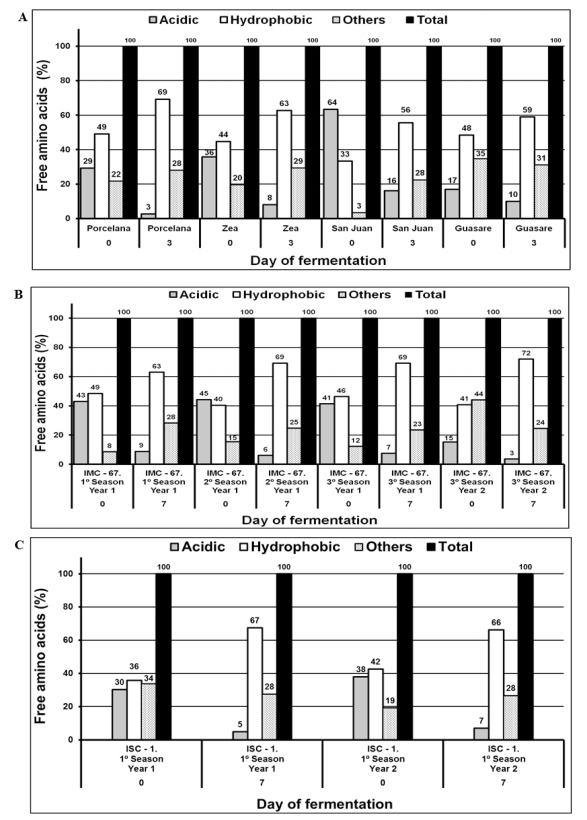
Figure 6. Composition (%) of amino acids on the first and last day of fermentation; a) Cocoa Criollo, b) Cocoa Forastero and c) Cocoa Trinitario.
Roasting of cocoa is an essential step for the chocolate flavor development. At this stage, the Maillard’s reaction takes place, involving free amino acids, peptides, and reducing sugars [32, 33]. Figure 5 also shows a significant decrease of free amino acids during roasting, being greater for the hydrophobic ones. Voigt et al. [2] reported that the hydrophobic amino acids leucine, alanine, phenylalanine, and tyrosine, released by proteinase activities in fermentation, are important contributors, as well as the reducing sugars fructose and glucose derived from sucrose hydrolysis [4, 34]. This behavior has been associated to a significant increase in pyrazines after roasting, which are the compounds responsible for cocoa flavor intensity.
In this research, a comparative study between the free amino acid profile (last day of fermentation and roasting) and the content of pyrazines obtained was conducted, according to the procedure reported by Brunetto et al [35]. Figure 7 shows the amino acids content generated during the fermentation and their decrease due to their transformation into pyrazines. Furthermore, among the analyzed Venezuelan cocoas, the amino acids behavior in Porcelana variety can be highlighted, since an important decrease of hydrophobic amino acids can be observed as well as a significant increase in pyrazine content during roasting stage, similar to the behavior observes on the Forastero variety. Several researchers confirm that free amino acids act as substrate in the formation of pyrazines [9, 36]. Therefore, pyrazines levels can be used as an index of the roasting process [37].
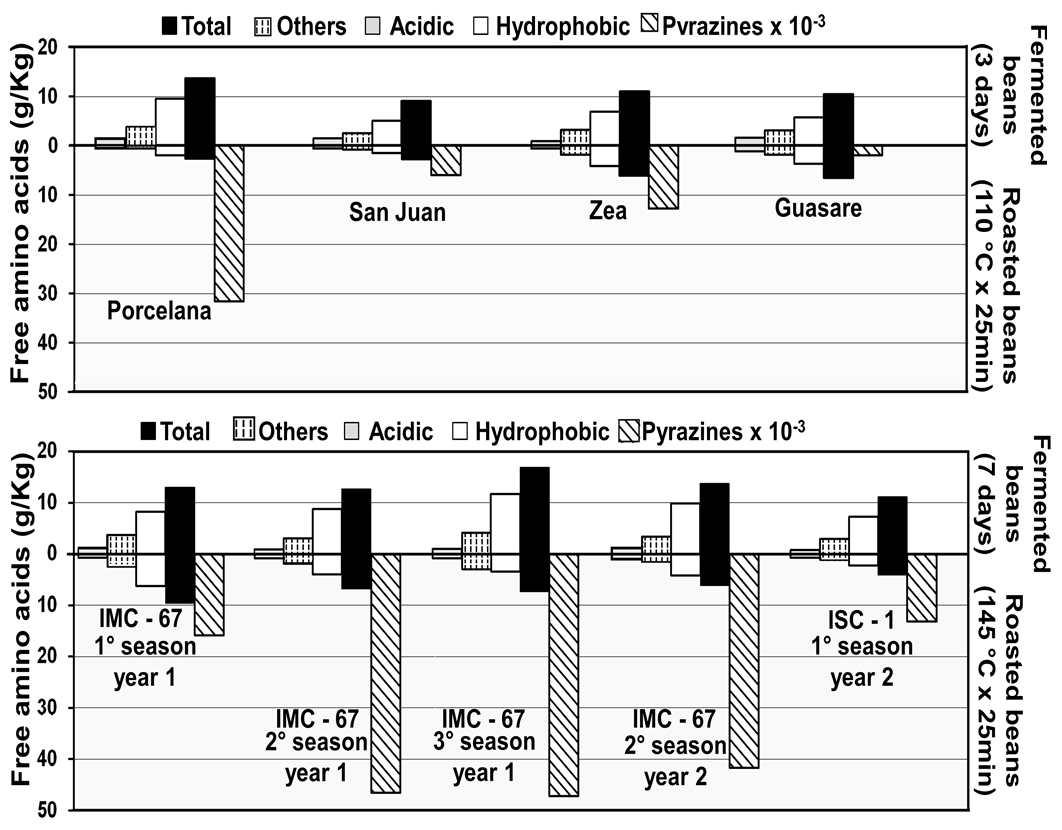
Figure 7. Effect of roasting over amino acids profile in different types of cocoa beans.
Conclusions
The obtained results have proved that the developed reversed-phase HPLC method with PTC-AA DADis a useful tool for primary and secondary amino acids determination in cocoa samples. Amino acids extraction from cocoa samples with water in an ultrasonic bath at 70 ºC provided a satisfactory extraction efficiency with recoveries higher than 89%. The pre-column amino acids derivatization time was significantly reduced using a domestic microwave oven. Besides, exploiting a column-switching system with an OASIS pre-column allowed the efficient and fast cleaning of PTC-AA extract, thereby reducing matrix interferences. Different validation parameters showed that with the developed method rapid and highly reproducible results can be obtained.
The proposed method was successfully applied for the evaluation of amino acids content in Trinitario, Forastero, and Criollo cocoa samples grown in different seasons of the year. All the analyzed samples had a significant content of free amino acids, ranging from 3.87 to 5.97 g kg-1 in the absence of fermentation. In general, amino acid degradation was observed on the first day of fermentation, whereas some samples showed progressive increase as the fermentation process carried on, with a predominance of hydrophobic amino acids such as leucine, phenylalanine, valine, alanine, and isoleucine. Moreover, during the roasting step a fraction of the amino acids, especially the hydrophobic ones, is partially degraded by the Maillard’s reaction to form the compounds associated with the aroma and flavor of cocoa.
References
Referencias
E.M. Afoakwa, A. Paterson, M. Fowler, and A. Ryan, “Flavor formation and character in cocoa and chocolate: A critical review”, Critical Reviews in Food Science and Nutrition, vol. 48, pp. 840–857, 2008. DOI: 10.1080/10408390701719272.
J. Voigt, B. Biehl, H. Heinrichs, S. Kamaruddin, G. Marsoner, and A. Hugi, “In-vitro formation of cocoa specific aroma precursors: aroma-related peptides generated from cocoa seed protein by co-operation of an aspartic endoprotease and a carboxypeptidase”, Food Chemisty, vol. 49, pp. 173-80, 1994. DOI: https://doi.org/10.1016/0308-8146(94)90155-4
J. Voigt and B. Biehl, “Precursors of the cocoa-specific aroma components are derived from the vicilin-class (7S) globulin of the cocoa seeds by proteolytic processing”, Botanical Acta, vol. 108, pp. 283-289, 1995. DOI: 10.1111/j.1438-8677.1995.tb00496.x
H. Puziah, S. Jinap, K.S.M. Sharifah, and A. Asbi, “Changes in free amino acid, peptide-N, sugar and pyrazine concentration during fermentation”, Journal of the Science Food and Agriculture, vol. 78, pp. 535-542, 1998. DOI: https://doi.org/10.1002/(SICI)1097-0010(199812)78:4<535::AID-JSFA151>3.0.CO;2-6.
G.A. Reineccius, D.A. Andersen, T.E. Kavanagh, and P.G. Keeney, “Identification and quantification of the free sugars in cocoa beans”, Journal of Agricultural and Food Chemistry, vol. 20, n° 2, pp. 199-202, 1972. DOI: https://doi.org/10.1021/jf60180a033.
L. Lima, H. Almeida, M. Nout, and M. Zwietering, “Theobroma cacao L: The food of the Gods. Quality determinants of comercial cocoa beans, with particular reference to the impact of fermentation”, Critical Reviews in Food Science and Nutrition, vol. 51, pp. 731-761, 2011. DOI: 10.1080/10408391003799913.
N. Camu, T. De Winter, S. Addo, J. Takrama, H. Bernaert, and L. De Vuyst, “Fermentation of cocoa beans: influence of microbial activities and polyphenol concentration of the flavor of chocolate”, Journal of the Science of Food and Agriculture, vol. 88, pp. 2288-2297, 2008. DOI: 10.1002/jsfa.
J. Serra Bonvehi, “Investigation of aromatic compounds in roasted cocoa power”, European Food Research and Technology, vol. 221, pp. 19-29, 2005. DOI: 10.1007/s00217-005-1147-y.
E. Cros and N. Jeanjean, “Formation de l’arôme cacao. Cacao et chocolat. Production, utilisation, caractéristiques". J. Pontillon. Paris. Ed. Tec & Doc, 1998, pp. 188-206.
T.A. Rohan, “Precursors of chocolate aroma: a comparative study of fermented and unfermented cocoa beans”, Journal of Food Science, vol. 29, n° 4, pp. 456-459, 1964. DOI: https://doi.org/10.1111/j.1365-2621.1964.tb01760.x.
P.M. Kirchhoff, B. Biehl, and G. Crone, “Peculiarity of the accumulation of free amino acids during cocoa fermentation”, Food Chemistry, vol. 31, pp. 295-311, 1989.
P.M. Kirchhoff, B. Biehl, H. Ziegeler-Berghausen, M. Hammoor, and R. Lieberei, "Kinetics of the formation of free amino acids in cocoa seeds during fermentation”, Food Chemistry, vol. 34, pp. 161-179, 1989.
E. Portillo, L. Graziani de Fariñasand E. Bentancourt, “Chemical analysis of Criollo Porcelana Cocoa (Theobroma cocoa L.) in the South of Maracaibo Lake”, Revista Facultad de Agronomía Universidad del Zulia, Venezuela, vol. 24, pp. 522-546, 2007.
E. Portillo, D. Fuenmayor, B. Rodríguez, and R. Díaz, “Effect of the fermentation on the content of purines in Criollo and Hybrid cocoa (Theobroma cocoa L) in the south of Maracaibo´s lake”, Revista Facultad de Agronomía Universidad del Zulia, Venezuela, vol. 28, n°1, pp. 228-237, 2011.
L. Graziani de Fariñas, L. Ortiz de Bertorelli, M. Lemus, and P. Parra, “Effect of blending two types of cocoa seeds on chemical properties during fermentation”, Agronomía Tropical, vol. 52, n° 3, pp. 325-342, 2002.
L. Graziani de Fariñas, L. Ortiz de Bertorelli, and P. Parra, “Chemical characteristics of seeds of different types of cocoa from the locality of Cumboto, Aragua”, Agronomía Tropical, vol. 53, n°2, pp. 133-144, 2003.
L. Ortiz de Bertorelli, G. Camacho, and L. Graziani de Fariñas, “Effect of sun drying on the quality of fermented cocoa beans”, Agronomía Tropical, vol. 54, n°1, pp. 31-43, 2004.
L. Ortiz de Bertorelli, L. Graziani de Fariñas, and R.L. Gervaine, “Evaluation of some chemical characteristics of cocoa beans in fermentation”, Agronomía Tropical, vol. 51, n°1, pp. 73-79, 2009.
L. Ortiz de Bertorelli, L. Graziani de Fariñas, and R.L. Gervaine, “Some factors affecting characteristics of fermented and sun dried cocoa grain”, Agronomía Tropical, vol. 59, n°2, pp. 119-127, 2009.
A. Zambrano, et al., “Chemical evaluation of aroma and flavor of precursors of the Criollo Merideño cocoa during fermentation in two edaphoclimatic conditions”, Agronomía Tropical, vol. 60, n° 2, pp. 211-219, 2010.
M.R. Brunetto, et al., “Determination of theobromine, theophylline and caffeine in cocoa samples by high-performance liquid chromatographic method with on-line simple cleanup in a switching-column system”, Food Chemistry, vol. 100, pp. 459-467, 2007. DOI: 10.1016/j.foodchem.2005.10.007.
C. Álvarez, E. Pérez and M.C. “Physical-chemical characterization of fermented, dried and roasted cocoa beans cultivated in the region of Cuyagua, Aragua State”, Agronomía Tropical, vol. 57, n°4, pp. 249-256, 2007.
International Office of Cocoa, Chocolate and Sugar Confectionary (IOCCC). 1996. “Determination of the Fat Content of Cocoa Powder by Soxhlet Extraction”, IOCCC. Analytical Method N° 37.
B.A. Bidlingmeyer, S.A. Cohen, and T.L. Tarvin, “Rapid analysis of amino acids using pre-column derivatization”, Journal of Chromatography, vol. 336, pp. 93-104, 1984. DOI: https://doi.org/10.1016/S0378-4347(00)85133-6.
M. Hariharan, S. Naga, and T. VanNoord, “Systematic approach to the development of plasma amino acid analysis by high-performance liquid chromatography with ultraviolet detection with precolumn derivatization using phenyl isothiocyanate”, Journal of Chromatography, vol. 621, pp. 15-22, 1993. DOI: https://doi.org/10.1016/0378-4347(93)80071-B
L. Zhang, et al., “One single amino acid for estimation the content of total free amino acids in Qingkailing injection using high-performance liquid chromatography-diode array detection” Journal of Analytical Methods in Chemistry, 2014. DOI: http://dx.doi.org/10.1155/2014/951075.
L. Chen, D. Song, Y. Tian, L. Ding, A. Yu, and H. Zhang. Application of on-line microwave sample-preparation techniques. Trends Anal. Chem. Vol. 27, pp. 151–159, 2008. DOI: 10.1016/j.trac.2008.01.003.
Oasis Applications Notebook. Waters Corporation, 2003. https://www.waters.com/waters/library.htm?lid=1528415&cid=511436&locale=en_US.
S. Jinap, H.N. Lioe, I. Yusep, S. Nazamid, and B. Jamilah, “Role of carboxypeptidases to the free amino acid composition, methylpyrazine formation and sensory characteristic of underfermented cocoa beans”, International Food Research Journal, vol. 17, pp. 763-774, 2010.
G. Bytof, B. Biehl, H. Heinrichs, and J. Voigt, “Specificity and stability of the carboxypeptidase activity in ripe, ungerminated seeds of Theobroma cacao L”, Food Chemistry, vol. 54, n°1, pp. 15-21, 1995. DOI: https://doi.org/10.1016/0308-8146(95)92657-6.
A. Oyekale, M. Bolaji, and O. Olowa, “The effect of climate change in cocoa production and vulnerability assessment in Nigeria”, Agricultural Journal, vol. 4, n°2, pp. 77-85, 2009. DOI: aj.2009.77.85.
T.A. Rohan and T. Stewart, “The precursors of chocolate aroma: changes in the free amino acids during the roasting of cocoa beans”, Journal of Food Science, vol. 31, n°2, pp. 202-205, 1966. DOI: https://doi.org/10.1111/j.1365-2621.1966.tb00479.x.
T.A. Rohan and T. Stewart, “The precursors of chocolate aroma: production of free amino acids during fermentation cocoa beans”, Journal of Food Science, vol. 32, pp. 395-398, 1967. DOI: https://doi.org/10.1111/j.1365-2621.1967.tb09693.x.
T.A. Rohan and T. Stewart, “The Precursors of Chocolate Aroma: changes in the sugars during the roasting of cocoa beans”, Journal of Food Science, vol. 31, n°2, pp. 206-209, 1966. DOI: https://doi.org/10.1111/j.1365-2621.1966.tb00480.x.
M.R. Brunetto, et al., “Headspace gas chromatography-mass spectrometry determination of alkylpyrazines in cocoa liquor samples”, Food Chemistry, vol. 112, pp. 253-257, 2009. DOI: 10.1016/j.foodchem.2008.05.082.
E. Portillo, et al., “Aroma formation of Criollo cocoa (Theobroma cacao L.) in function of the post-harvest treatment in Venezuela”, Revista Agrícola de la Universidad de Oriente, Venezuela, vol. 9, n°2, pp. 458-468, 2009.
S. Jinap, W. I. Wan Rosli, A.R. Russly, and L.M. Nordin, “Effect of roasting time and temperature on volatile component profile during nib roasting of cocoa beans (Theobroma cacao)”, Journal of the Science of Food and Agriculture, vol. 77, pp. 441-448, 1998. DOI: https://doi.org/10.1002/(SICI)1097-0010(199808)77:4<441::AID-JSFA46>3.0.CO;2-%23.
Cómo citar
IEEE
ACM
ACS
APA
ABNT
Chicago
Harvard
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. Sandy Rodrigues Dias, Luiza Adami, Nádia Nara Batista, Ana Paula Pereira Bressani, Silvia Juliana Martinez, Disney Ribeiro Dias, Rosane Freitas Schwan. (2026). Non-Saccharomyces starter cultures modulate the chemical and sensory profile of chocolates from Brazilian cocoa hybrid mixtures. Food Research International, 231, p.118695. https://doi.org/10.1016/j.foodres.2026.118695.
2. María Eugenia García, Rommel Aaron Pazmiño Vallejo, Dayana Belén Troya Carrera, Juan Gaibor Chávez. (2023). Estudio de cinética de fermentación alcohólica de cacao en cajón tipo madera. Bionatura, 8(4), p.1. https://doi.org/10.21931/RB/2023.08.04.6.
3. César R. Balcázar-Zumaeta, Efraín M. Castro-Alayo, Ilse S. Cayo-Colca, Guillermo Idrogo-Vásquez, Lucas D. Muñoz-Astecker. (2023). Metabolomics during the spontaneous fermentation in cocoa (Theobroma cacao L.): An exploraty review. Food Research International, 163, p.112190. https://doi.org/10.1016/j.foodres.2022.112190.
4. Jitka KLIKAROVÁ, Lenka ČESLOVÁ, Jan FISCHER. (2021). Rapid analysis of phenyl isothiocyanate derivatives of amino acids present in Czech meads. Journal of Chromatography A, 1644, p.462134. https://doi.org/10.1016/j.chroma.2021.462134.
5. Fredijs Dimins, Ingmars Cinkmanis, Vitalijs Radenkovs, Ingrida Augspole, Anda Valdovska. (2022). Analysis of 18 Free Amino Acids in Honeybee and Bumblebee Honey from Eastern and Northern Europe and Central Asia Using HPLC-ESI-TQ-MS/MS Approach Bypassing Derivatization Step. Foods, 11(18), p.2744. https://doi.org/10.3390/foods11182744.
6. César R. Balcázar-Zumaeta, Editha Fernández-Romero, Alessandra Santos Lopes, Nelson Rosa Ferreira, Gilson Celso Albuquerque Chagas-Júnior, Ives Yoplac, Heydi A. López-Trigoso, Mery L. Tuesta-Occ, Italo Maldonado-Ramirez, Jorge L. Maicelo-Quintana, Ilse S. Cayo-Colca, Efrain M. Castro-Alayo. (2024). Amino acid profile behavior during the fermentation of Criollo cocoa beans. Food Chemistry: X, 22, p.101486. https://doi.org/10.1016/j.fochx.2024.101486.
7. Quintino Reis de Araujo, Guilherme Amorim Homem de Abreu Loureiro, Cid Edson Mendonça Póvoas, Douglas Steinmacher, Stephane Sacramento de Almeida, Dario Ahnert, Virupax Chanabasappa Baligar. (2021). Effect of Different Edaphic Crop Conditions on the Free Amino Acid Profile of PH-16 Dry Cacao Beans. Agronomy, 11(8), p.1637. https://doi.org/10.3390/agronomy11081637.
8. Jonathan Rios-Jara, Danny Lévano-Rodríguez . (2022). Importancia de los dispositivos usados en la fermentación de Cacao (Theobroma cacao L.). Revista agrotecnológica amazónica, 2(1) https://doi.org/10.51252/raa.v2i1.281.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2020 Revista Colombiana de Química

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
Los autores/as conservarán sus derechos de autor y garantizarán a la revista el derecho de primera publicación de su obra, el cuál estará simultáneamente sujeto a la Licencia de reconocimiento de Creative Commons (CC. Atribución 4.0) que permite a terceros compartir la obra siempre que se indique su autor y su primera publicación en esta revista.
Los autores/as podrán adoptar otros acuerdos de licencia no exclusiva de distribución de la versión de la obra publicada (p. ej.: depositarla en un archivo telemático institucional o publicarla en un volumen monográfico) siempre que se indique la publicación inicial en esta revista.
Se permite y recomienda a los autores/as difundir su obra a través de Internet (p. ej.: en archivos telemáticos institucionales o en su página web) antes y durante el proceso de envío, lo cual puede producir intercambios interesantes y aumentar las citas de la obra publicada. (Véase El efecto del acceso abierto).