Comparison of hemocytes of 5th-instar nymphs of Rhodnius prolixus (Stal) and Rhodnius robustus (Larousse 1927), before and after molting
Comparación de los hemocitos de las ninfas de V instar de Rhodnius prolixus (Stål) and Rhodnius robustus, antes y después de la muda (Larousse 1927)
DOI:
https://doi.org/10.15446/revfacmed.v63n1.44901Palabras clave:
Rhodnius prolixus, hemocytes, molt, Rhodnius robustus (en)Rhodnius prolixus, hemocitos, muda (es)
Background. The immune response of insects involves humoral factors and cellular elements known as hemocytes. There are different reports that explore the response of hemocytes to infections, but the effect that molting has on this response has not been explored so far. We hypothesized that there would be a change in the percentage of hemocytes as a response to the molting process.
Objective. The aim of this work was to compare the hemogram (CBC), the formula, and the differential count of hemocytes in IV instar before molting and in V instar nymphs 24 hours after molting in two species: Rhodnius prolixus and Rhodnius robustus.
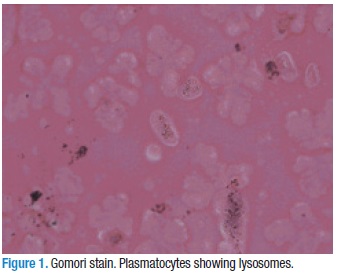
Materials and methods. We assayed different staining methodologies including Giemsa, Alcian Blue pH 2.0, Alcian Blue pH 2.6, Gomori substrate, PAS (Schiff), Sudan Black and Papanicolau with positive controls for each one. In the Gomori staining, we observed lysosomes in the granulocytes and plasmatocytes, but the differentiation was better detected using Giemsa staining.
Results. There were no statistically significant differences between the two species studied in plasmatocytes (p=0,053) or even in granulocytes (p=0,5). However, differences were significant in the prohemocytes (p=0,001) during the molting process in both Rhodnius prolixus and Rhodnius robustus.
Conclusions. Significant differences in prohemocytes between nymphs of IV and V instar were detected. No significant differences in the amount of cells were observed between the two species and the two stages. These findings may be explained due to their role as precursor cell of prohemocytes.
Antecedentes. La respuesta inmune de los insectos involucra factores humorales y elementos celulares llamados hemocitos. Existen varios trabajos explorando la respuesta de los hemocitos frente a infecciones, pero no como respuesta al proceso de muda.
Objetivo. Comparar el hemograma: la fórmula y el recuento diferencial de hemocitos de Rhodnius prolixus y Rhodnius robustus en ninfas de IV estadio antes de la muda y ninfas de V estadio 24 horas después de la muda.
Materiales y métodos. Se ensayaron las coloraciones Giemsa, Alcian Blue pH 2.0, Alcian Blue pH 2.6, sustrato Gomori, PAS (Schiff), Sudán Negro, Papanicolau, con controles positivos para cada una. Con la coloración de Gomori se observaron lisosomas en los granulocitos y en plasmatocitos, pero la diferenciación se logró mejor con la coloración de Giemsa.
Resultados. Los plasmatocitos fueron más abundantes en las ninfas de IV estadio de Rhodnius robustus y en las de V estadio de las dos especies, pero en las ninfas de V estadio de R. prolixus, el recuento de prohemocitos fue mayor; para los oenocitoides, esferulocitos y adipohemocitos el recuento no alcanzó al 1 %. En las dos especies no se encontraron diferencias estadísticamente significativas en plasmatocitos (p=0,05319), ni en granulocitos (p=0,5), entre las ninfas de IV y V estadio, pero fue significativa en prohemocitos (p=0,001).
Conclusiones. Se detectaron diferencias significativas en prohemocitos entre las ninfas de IV y V estadio. No hubo un aumento significativo en el conteo de células en los diferentes estadios.
INVESTIGACIÓN ORIGINAL
DOI: https://doi.org/10.15446/revfacmed.v63n1.44901
Comparison of hemocytes of V-instar nymphs of Rhodniusprolixus (Stål) and Rhodnius robustus (Larousse 1927), before and after molting
Comparación de los hemocitos de las ninfas de V instar de Rhodnius prolixus (Stål) and Rhodnius robustus, antes y después de la muda (Larousse 1927)
Elizabeth Ruiz1, Myriam Consuelo López1, Favio Aurelio Rivas1, Ángel Yovanny Sánchez2, Ligia Inés Moncada1
1 Departamento de Salud Pública, Facultad de Medicina, Universidad Nacional de Colombia. Bogotá D. C., Colombia.
2 Departamento de Patología, Facultad de Medicina, Universidad Nacional de Colombia. Bogotá D. C., Colombia.
Correspondence: Ligia Inés Moncada-Álvarez. Departamento de Salud Pública, Facultad de Medicina, Oficina 302, Universidad Nacional de Colombia. Carrera 30 No. 45-03. Teléfono: +57 1 3165000- Bogotá D.C., Colombia. E-mail: limoncadaa@unal.edu.co.
Recibido: 12/08/2014 Aceptado: 17/09/2014
Summary
Background. The immune response of insects involves humoral factors and cellular elements known as hemocytes. There are different reports that explore the response of hemocytes to infections, but the effect that molting has on this response has not been explored so far. We hypothesized that there would be a change in the percentage of hemocytes as a response to the molting process.
Objective. The aim of this work was to compare the hemogram (CBC), the formula, and the differential count of hemocytes in IV instar before molting and in V instar nymphs 24 hours after molting in two species: Rhodnius prolixus and Rhodnius robustus.
Materials and methods. We assayed different staining methodologies including Giemsa, Alcian Blue pH 2.0, Alcian Blue pH 2.6, Gomori substrate, PAS (Schiff), Sudan Black and Papanicolau with positive controls for each one. In the Gomori staining, we observed lysosomes in the granulocytes and plasmatocytes, but the differentiation was better detected using Giemsa staining.
Results. There were no statistically significant differences between the two species studied in plasmatocytes (p=0,053) or even in granulocytes (p=0,5). However, differences were significant in the prohemocytes (p=0,001) during the molting process in both Rhodnius prolixus and Rhodnius robustus.
Conclusions. Significant differences in prohemocytes between nymphs of IV and V instar were detected. No significant differences in the amount of cells were observed between the two species and the two stages. These findings may be explained due to their role as precursor cell of prohemocytes.
Keywords: Rhodnius prolixus, hemocytes, molt, Rhodnius robustus (MeSH).
Ruiz E, López MC, Rivas FA, Sánchez AY, Moncada LI. Comparison of hemocytes of V-instar nymphs of Rhodnius prolixus (Stål) and Rhodnius robustus (Larousse 1927), before and after molting. Rev. Fac. Med.2015;63(1):11-7. doi: https://doi.org/10.15446/revfacmed.v63n1.44901.
Resumen
Antecedentes. La respuesta inmune de los insectos involucra factores humorales y elementos celulares llamados hemocitos. Existen varios trabajos explorando la respuesta de los hemocitos frente a infecciones, pero no como respuesta al proceso de muda.
Objetivo. Comparar el hemograma: la fórmula y el recuento diferencial de hemocitos de Rhodnius prolixus y Rhodnius robustus en ninfas de IV estadio antes de la muda y ninfas de V estadio 24 horas después de la muda.
Materiales y métodos. Se ensayaron las coloraciones Giemsa, Alcian Blue pH 2.0, Alcian Blue pH 2.6, sustrato Gomori, PAS (Schiff), Sudán Negro, Papanicolau, con controles positivos para cada una. Con la coloración de Gomori se observaron lisosomas en los granulocitos y en plasmatocitos, pero la diferenciación se logró mejor con la coloración de Giemsa.
Resultados. Los plasmatocitos fueron más abundantes en las ninfas de IV estadio de Rhodnius robustus y en las de V estadio de las dos especies, pero en las ninfas de V estadio de R. prolixus, el recuento de prohemocitos fue mayor; para los oenocitoides, esferulocitos y adipohemocitos el recuento no alcanzó al 1 %. En las dos especies no se encontraron diferencias estadísticamente significativas en plasmatocitos (p=0,05319), ni en granulocitos (p=0,5), entre las ninfas de IV y V estadio, pero fue significativa en prohemocitos (p=0,001).
Conclusiones. Se detectaron diferencias significativas en prohemocitos entre las ninfas de IV y V estadio. No hubo un aumento significativo en el conteo de células en los diferentes estadios.
Palabras clave: Rhodnius prolixus, hemocitos, muda (DeCS).
Ruiz E, López MC, Rivas FA, Sánchez AY, Moncada LI. [Comparación de los hemocitos de las ninfas de V instar de Rhodnius prolixus (Stål) and Rhodnius robustus, antes y después de la muda (Larousse 1927). Rev. Fac. Med. 2015;63(1):11-7. doi: https://doi.org/10.15446/revfacmed.v63n1.44901
Introduction
Invertebrate cellular immune response involves the processes of phagocytosis, encapsulation, and nodulation, which are all mediated through both humoral and cellular mechanisms. These mechanisms are found in the hemolymph of the insects and are known as hemocytes (1). In addition to these functions, they are responsible for clearing apoptotic cells during development (2,3), and are involved in metabolism, synthesis, and nutrient storage (4). In insects, it is known that immune cells are produced in two stages during insect development: during head or dorsal mesoderm embryogenesis and during the larval or nymphal stages (5). They are located in separate anatomical compartments and are differentiated by sequential waves, transcription, and epigenetic factors that determine the fate of different cell types that are conserved through evolution (6).
In the species Rhodnius prolixus, the presence of hemocytes has been associated with infections, but few studies have characterized insect blood cells and the influence of the endocrine system (that is known to be related specifically to the hormone production of 20– hydroxyecdysone). There are some studies concerning the presence of the hemocyte at different stages. In the molting process, the production of new tissue demands synthesis de novo of proteins and productions of different elements, including the hemocytes that contribute to the formation and breakdown of wings in Manduca sexta (7,8). As such, we hypothesized that there are differences in the complete blood count (CBC) of IV and V Rhodnius prolixus nymphs. The aim of this study was to compare the number of hemocytes before and after molting among nymphs from the V instar of Rhodnius prolixus and Rhodnius robustus in order to validate differences in the hemocyte count. We chose this point in time because at that point of the insect development, apoptosis and the de novo synthesis of peptides, which are involved in the organization and production of new substances needed by the insect to develop and mature, are present.
Methodology
Maintenance of triatomine colonies
Insect colonies of Rhodnius prolixus species, from the Colombian region of Casanare, were established in the laboratory of Medical Entomology at the Universidad Nacional de Colombia for approximately ten years. R. robustus, from the Colombian region of Arauca, were used. Temperature conditions ranged from 24 to 28 ° C, and relative humidity (RH) was between 50-60 %. The insects were fed mouse blood.
Staining method selection
In preliminary trials, hemolymph samples were stained with different reagents, including Giemsa stain, Alcian Blue stain pH 2.0, Alcian Blue stain pH 2.6, Gomori-Takamatsu substrate, PAS (Schiff) stain, Sudan Black B, and Papanicolau stain to test which of them displayed a better differentiation of cell characteristics. For each staining, a positive control was performed.
The controls used to test the different staining methods were: periodic acid-Schiff (cut of small intestine), Giemsa (gastric biopsy diagnosis of moderate chronic atrophic antral gastritis and presence of Helicobacter pylori + + + / + + +), Alcian Blue (cut of esophagus diagnosed with Barrett's esophagus), Sudan Black (adipose tissue), Papanicolau (cytology smears). Also, Gomori-Takamatsu staining was used to detect lysosomes in hemocytes and thus relate their presence in the participation of certain lineages of hemocytes in cellular defense processes. Based on the results using the staining methods selected, we then proceeded to obtain samples of hemolymph from V instar nymphs for each insect species and staining method.
Preparation and staining of hemolymph samples
Groups of IV R. robustus and R. prolixus nymphs were fed on mouse blood until they reached repletion in order to facilitate the obtaining of hemolymph sample and also to ensure their passage to the next stage. Each of the nymphs were cut at the infrafemoral level. The abdomen was slightly pressed to obtain an aliquot of 1µL of hemolymph, which was coded with the initials of the species, the sample stage, the type of staining, and the consecutive number. The same procedure was performed with V instar nymphs 24 hours after molting.
Observation, characterization and classification
Hemolymph samples were observed under a light microscope with a magnification of 100X. Characteristics of each cell type were recorded and observations were analyzed, considering the different studies related to hemocyte morphology and, in particular, those published by Jones (9).
Hemocyte measurement
The larger diameter, smaller diameter and the nucleus was measured using a Alphaphot Nikon microscope (Tokio, Japan) with a calibrated rule.
Differential hemocyte count
For counting, fifty V nymphs were cut at the infrafemoral level. The sample of hemolymph was taken. Insects were fed and allowed to molt. Twenty-four hours after molting, we proceeded to carry out a new infrafemoral cut and counting in order to determine the results for each of the species tested. The bounded number of cells counted was 200, because this was the range of the minimum value of hemocytes found.
Total hemocyte count
Twenty IV and V instar nymphs from the two triatomine species were randomly selected and fed up until repletion. We carried out an infrafemoral cut to each insect and 0.5 µL of hemolymph was diluted in 10 µL of EDTA anticoagulant to obtain a total dilution of 1:20. The mixture was placed in a Neubauer chamber, and we proceded to cell-counting.
Results
Ethical considerations approval was obtained from the Ethics Committee of the Faculty of Medicine at the National University of Colombia.
When the Gomori staining was applied, we observed lysosomes in granulocytes and plasmatocytes. In prohemocytes, the lysosomes were not observed (Figure 1). With Schiff, Alcian Blue and Sudan Black staining, we could not observe a good differentiation of the hemocytes. With Giemsa, the most relevant features stood out so that we could classify hemocytes into six types: prohemocytes, plasmatocytes and granulocytes, which were the three most frequent types of cells (Figure 2, A-F). Oenocytoids, spherulocytes and adipohemocytes were also observed. Figure 2 demonstrates the morphological characteristics of the three major lineages of hemocytes described.
Prohemocytes
Prohemocytes are characterized as basophil cells, as has already been mentioned. The core was prominent, the shape was round, and the average major diameter was 4.04 (range 2-7 microns). The average minor diameter was 3.45 microns (range 2 - 6μm), the ratio of the nucleus to the cytoplasm was about 1:1.5, which corroborates that the core occupies a large percentage of the cytoplasm. Several cells that were in the early stage of dividing late in both instars IV and V were observed. Several prohemocytes were also detected with chromatin division.
Plasmatocytes
The cells showed variable shape, but mostly ovoid with a central nucleus. The cytoplasm was vacuolated with the presence of thin pseudopodia (Figures 2C and 2D). The phagocytic capacity was better observed in Gomori staining, highlighting the lysosomes, which is reflected in the appearance of the black color. Due to the presence of lysosomes, the tendency was observed in some cases to be grouped. The mean largest diameter of cytoplasm was 8.85 µ (range 4-21 µ), the minor diameter was 6.73 µ (range, 3 to 20 µ), and the ratio varied between 1.2 and 4 µ.
Granulocytes
Granulocytes and plasmatocytes observed in the hemolymph of R. prolixus and R. robustus were observed to have various sizes (10). Their main feature was the presence of large granules; the shape varied from round to oval. (Figure 2 E-F). Sometimes these granules did not stain with Giemsa dye, and they had an appearance ranging from yellowish to deep brown cast.
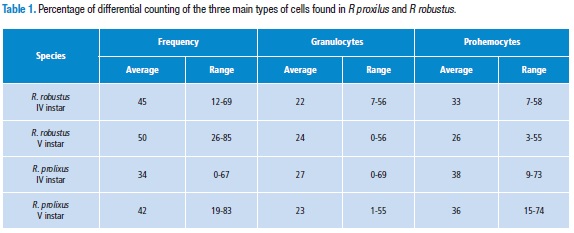
The plasmatocytes were the most abundant cells in nymphs of Rhodnius robustus (IV and V instars). However, V instar Rhodnius prolixus nymphs presented a higher number of prohemocytes than plasmatocytes. Plasmatocytes counts and granulocytes in some individuals showed a wide range; in some cases almost exclusively counts of one cell type (Table 1) were found. The other cells, oenocytoids, spherulocytes and adipohemocytes did not reach even 1% when averaging the count of 50 individuals.
The average differential cell count in the nymphs of instar IV before and V after 24 hours of molting was generally higher for plasmatocytes, followed by granulocytes and prohemocytes. Although three other types of cells were observed, as reported earlier, no cell type reached 1.0 % in the overall count.
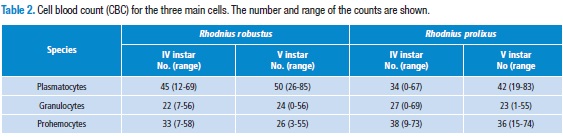
In the species Rhodnius robustus, no statistically significant differences between the plasmatocytes of IV and V instar were detected (p=0.06; 95 %CI=28.918-39.802). The same is true for granulocytes (p=0.5; 95 %CI=19.295-25.385). In contrast prohemocytes showed a significant difference (p=0.001; 95 %CI=29.22 to 36.12). In Table 2, the CBC number and range of the three main cells are shown. In R. prolixus, we observed similar results: plasmatocytes (p=0.05; 95%CI=29.918-39.802), granulocytes (p=0.5; 95 %CI=22.232-32.048), and the difference was significant in prohemocytes (p=0.001; CI=33.94- 42.62). (Table 2).
Moreover, the counts of the cells in 0.5 µL of hemolymph in 20 individuals of IV and V instar of Rhodnius prolixus and Rhodnius robustus are summarized in Table 3. These differences were not statistically significant (p=0,38. Student t-test).
The comparison of the differential counts between IV instar of the two species was observed. There was no statistically significant difference between the three main groups of cells: prohemocytes, p= 0.1320; plasmatocytes, p= 0.1741; and granulocytes, p= 0.1810. The same was true in the V instar: prohemocytes, p= 0.1741; plasmatocytes, p= 0.3572, granulocytes, p= 0.1320.
Discussion
In this study, six types of hemocytes were identified, based on size, morphology, presence of granules, and dye-staining properties. The count number of the most abundant cells observed —plasmatocytes and granulocytes— showed no significant differences between the IV and V instars in the two species. In contrast, prohemocyte counts were statistically different in both species between the two instars. This fact could be explained because prohemocytes are the blood precursor cells and, due to molting process, synthesis and differentiation of the other types of hemocytes are produced.
As reported by De Azambuja et al (11), several staining methods have been conducted in order to establish which of them could have a better performance for cell counting. The best staining methods were Giemsa and Gomori. However, the differentiation that occurs between basophilic and acidophilic parts using Giemsa facilitates differentiation between them due to the positive results obtained with the Gomori substrate and the Papanicolau stain. The Gomori staining had the advantage that lysosomes were observed (8) and hemocytes of Manduca sexta were identified through Percoll gradient separation. However, the authors could not perform a separation of different types of cells (granulocytes and plasmatocytes) that were found in all layers of density.
The presence of lysosomes with Gomori stain, in both plasmatocytes and granulocytes confirm the phagocytic availability of these cells, in spite of the fact that granulocytes are considered the professional phagocytic cells (12). Nevertheless, many authors consider that the only phagocytic cells are plasmatocytes, Guilianini et al (13) Ling & Yu (14) consider that granulocyte phagocytosis is very important, as well as the oenocytoids, that could eat bacteria. Thus, the presence of lysosomes in granulocytes suggests their phagocytic capacity, although, in humans, there are many cells with lysosomes that cannot be considered quintessential phagocytes. The characteristics of the cytoplasms and nuclei of cells, ascertained using Giemsa staining, allowed for a better classification.
Measurements of prohemocytes and granulocytes stained with Giemsa are in accordance with those reported by Jones (9), with regard to shape and size. In addition, as the same author informed, with Giemsa stain, none of cells presented a strong acidophilic appearance. In granulocytes, as was described by Jones (9), the round inclusion did not stain and sometimes had a yellowish cast. As previously reported by the same authors, the variability in the size of plasmatocytes is extensive. The tendency of plasmatocytes to cluster in the hemolymph of the larvae of Saccostrea glomerata was observed in this study, as reported by Aladaileh et al (15). The mean largest diameter of cytoplasm was 8.85 µ (range 4-21 µ) and the smallest diameter was 6.73 µ (3-20 µ). The range was between 1.2 and 4 µ.
Nevertheless, Barraco & Loch (16) reported similar differential counts of prohemocytes, plasmatocytes and granulocytes in the two nymphal stages. Here, we observed similar counts of plasmatocytes and granulocytes and a significant differences in counts of prohemocytes. In contrast Stoepler et al (17) found no difference between the counts of hemocytes of young and mature larvae of Euclea delphinii and Lithacodes fasciola, possibly because, 24 hours after molting, there are many processes that require the participation of hemocytes. It is important to note that, as already described by Jones, there are many cells classified as prohemocytes, but the shape of the nucleus is very different and it may seem that they were in a cleavage process. It is also important to recognize that the differentiation and production of hemocytes is regulated to some extent by transcription factors and specifically the endocrine steroid hormone ecdysone (18) and because prohemocytes are the precursor cells of different lineages of blood cells in insects. This finding is consistent as they occur in greater numbers in passing from one nymphal stage to another.
According to our results, no significant difference was found in the total number of hemocytes present in the hemolymph of the two species of Rhodnius studied. This data is consistent with that published by Barraco & Loch (16), because there is an increased production of hemocytes to molt to the next nymphal stage, although this variation was not statistically significant. De Azambuja et al (11) suggested the hypothesis that differences in the total count and differential hemocyte counts (CBC) are due to nutritional factors or developmental conditions. In this study, we found that the development conditions only affected plasmatocytes and prohemocytes, which would confirm the thesis of these authors. However, they also found in some individuals that both the total cell count and differential cell count was highly variable, suggesting that there are also other factors affecting the population.
This paper can at least rule out the possibility of parasitic infection, because the insects that were taken on the experiments were from a colony maintained in the laboratory for many years with no infection guaranteed at least from parasites. Because we sampled the same individual before and after molt, Theopold et al (19) found in Lepidoptera that in the healing process two kind of cells were involved: granulocytes and plasmatocytes. However, in our work we did not find differences between these two type of cells before and after molting process. In contrast to prohemocytes, the number of these cells were higher before molt in both species. Probably this difference is due to the molting process but we cannot discard the probability that the healing process could be involved in this difference.
In conclusion, we report that plasmatocytes are the predominant cells in the hemolymph of Rhodnius prolixus and R. robustus, before and after molting. The total number of hemocytes and the percentages of plasmatocytes and granulocytes were similar in IV and V instars for the two species of Rhodnius. However, the count had statistical significance for prohemocytes, between stages in the two species studied.
Conflict of interest
None declared by the authors.
Financing
The financial support for the project was obtained from the DIB, (grant: number. 8548). Colciencias support was awarded to Elizabeth Ruiz (grant for young researcher (grant number 463-2008).
Acknowledgments
We are grateful to Andrea Marcela Conde, and the staff of Medical Entomology, Parasitology and Pathology laboratories, Faculty of Medicine, Universidad Nacional de Colombia.
Referencias
1. Williams MJ. Drosophila Hemopoiesis and Cellular Immunity. J Immunol. 2007;178:4711-6. doi: http://doi.org/zf9.
2. Silverstein SC. Phagocitosis of microbes: insights and prospects. Trends cell Biol. 1995;5(3):141-2. doi: http://doi.org/fmktfc.
3. Kurtz J. Phagocitosis by invertebrate hemocytes: causes of individual variation in Panorpia vulgaris scorpion flies. Micros Res Tech. 2002;57(6):456-68. doi: http://doi.org/dcws99.
4. Mandato CA, Diehl-Jones WL, Moore SJ, Downer RGH. The effects of eicosanoid biosynthesis inhibitors on prophenoloxidase activation, phagocytosis and cell spreading in Galleria melonella. J Insect Physiol. 1997;43(1):1-8. doi: http://doi.org/dt2xjk.
5.Holz A, Bossinger B, Strasser T, Janning W, Klapper R. The two origins of hemocytes in Drosophila. Development. 2003;130(20):4955-62. doi: http://doi.org/brfkz7.
6.Honti V, Csordas G, Hurucz E, Markus R, Ando I. The cell-mediated immunity of Drosophila melanogaster: hemocyte lineages, immune compartments microanatomy and regulation. Develop Comp Immunol. 2014;42(1):47-56. doi: http://doi.org/zgb.
7.Nardi JB, Miklasz SD. Hemocytes contribute to both the formation and breakdown of the basal lamina in developing wings of Manduca sexta. Tissue and Cell. 1989;21(4):559-67. doi: http://doi.org/cfvwkj.
8.Beetz S, Brinkmann M, Trenczeck T. Differences between larval and pupa hemocytes of the tobacco hornworm, Manduca sexta, determined by monoclonal antibodies and density centrifugation. J Ins Pathol. 2004;50(9):805-19. doi: http://doi.org/d7hbmt.
9.Jones JC. The hemocytes of Rhodnius prolixus Stål. Biol Bol. 1965;129(2):282-94. doi: http://doi.org/cf436q.
10.Amaral IMR, Neto JFM, Pereira JB, Franco MR, Beletti ME, Kerr WE, Ueira-Vieira C. Circulating hemocytes fron larvae of Melipona scutellaris (Hymenoptera,Apidae,Meliponini): Cell types and their role in phagocytosis. Micron. 2010; 41(2):123-9. doi: http://doi.org/d6n2jq.
11.De Azambuja P, Garcia ES, Ratcliffe NA. Aspects of Classification of Hemiptera Hemocytes from six Triatomine Species. Mem Inst Oswaldo Cruz. 1991; 86(1):1-10. doi: http://doi.org/czjx42.
12.Kwon H, Bang K, Cho S. Characterization of the hemocytes in larvae of Protaetia brevitarseis seulensis: involvement of granulocyte-mediated phagocytosis. Plos One. 2014;9(8):1-12. doi: http://doi.org/zgc.
13.Giulianini PG, Bertolo F, Battistella S, Amirante GA. Ultrastructure of the hemocytes of Cetonischema aeruginosa larvae (Coleoptera: Scarabidae) involvement of both granulocytes and oenocytoids in vivo phagocytosis. Tissue Cell. 2003;35(4):243-51. doi: http://doi.org/dm5rws.
14.Ling F, Yu XQ. Hemocytes from the tobacco hornworm Manduca sexta have distinct function in phagocytosis of foreign particles and self dead cells. Dev Comp Immunol. 2005;30(3):301-09. doi: http://doi.org/cbsprm.
15.Aladaileh S, Nair SV, Birch D, Raftos DA. Sydney rock oyster (Saccostrea glomerata) hemocytes: Morphology and function. J Invertebr Pathol. 2007;96(1):48-63. doi: http://doi.org/brt3j7.
16.Barraco MA, Loch CT. Ultrastuctural Studies of the Hemocytes of Panstrongylus megistus (Hemiptera:Reduviidae). Mem Inst Oswaldo Cruz. 1989;84:171-88. doi: http://doi.org/c7xwtp.
17.Stoepler TM, Castillo JC, Lili JT, Eleftherianos I. Hemocyte density increases with developmental stage in an immune-challenged forest caterpillar. PlosOne 2013;8(8):1-8. doi: http://doi.org/zgd.
18.Sorrentino RP, Carton Y, Govind S. Cellular Immune Response to parasite Infection in the Drosophila Lymph Gland is Developmentally Regulated. Develop Biol. 2002;243(1):65-80. doi: http://doi.org/dgk6nn.
19.Theopold U, Schmidt O, Söderhäll K, Dushay M. Coagulation in arthropods: defence, wound closure and healing. Trends in Immunol. 2004;25(6):289-94. doi: http://doi.org/fnxjz2.
Referencias
Williams MJ. Drosophila Hemopoiesis and Cellular Immunity. J Immunol. 2007;178:4711-6. doi: http://doi.org/zf9.
Silverstein SC. Phagocitosis of microbes: insights and prospects. Trends cell Biol. 1995;5(3):141-2. doi: http://doi.org/fmktfc.
Kurtz J. Phagocitosis by invertebrate hemocytes: causes of individual variation in Panorpia vulgaris scorpion flies. Micros Res Tech. 2002;57(6):456-68. doi: http://doi.org/dcws99.
Mandato CA, Diehl-Jones WL, Moore SJ, Downer RGH. The effects of eicosanoid biosynthesis inhibitors on prophenoloxidase activation, phagocytosis and cell spreading in Galleria melonella. J Insect Physiol. 1997;43(1):1-8. doi: http://doi.org/dt2xjk.
Holz A, Bossinger B, Strasser T, Janning W, Klapper R. The two origins of hemocytes in Drosophila. Development. 2003;130(20):4955-62. doi: http://doi.org/brfkz7.
Honti V, Csordas G, Hurucz E, Markus R, Ando I. The cell-mediated immunity of Drosophila melanogaster: hemocyte lineages, immune compartments microanatomy and regulation. Develop Comp Immunol. 2014;42(1):47-56. doi: http://doi.org/zgb.
Nardi JB, Miklasz SD. Hemocytes contribute to both the formation and breakdown of the basal lamina in developing wings of Manduca sexta. Tissue and Cell. 1989;21(4):559-67. doi: http://doi.org/cfvwkj.
Beetz S, Brinkmann M, Trenczeck T. Differences between larval and pupa hemocytes of the tobacco hornworm, Manduca sexta, determined by monoclonal antibodies and density centrifugation. J Ins Pathol. 2004;50(9):805-19. doi: http://doi.org/d7hbmt.
Jones JC. The hemocytes of Rhodnius prolixus Stål. Biol Bol. 1965;129(2):282-94. doi: http://doi.org/cf436q.
Amaral IMR, Neto JFM, Pereira JB, Franco MR, Beletti ME, Kerr WE, Ueira-Vieira C. Circulating hemocytes fron larvae of Melipona scutellaris (Hymenoptera,Apidae,Meliponini): Cell types and their role in phagocytosis. Micron. 2010; 41(2):123-9. doi: http://doi.org/d6n2jq.
De Azambuja P, Garcia ES, Ratcliffe NA. Aspects of Classification of Hemiptera Hemocytes from six Triatomine Species. Mem Inst Oswaldo Cruz. 1991; 86(1):1-10. doi: http://doi.org/czjx42.
Kwon H, Bang K, Cho S. Characterization of the hemocytes in larvae of Protaetia brevitarseis seulensis: involvement of granulocyte-mediated phagocytosis. Plos One. 2014;9(8):1-12. doi: http://doi.org/zgc.
Giulianini PG, Bertolo F, Battistella S, Amirante GA. Ultrastructure of the hemocytes of Cetonischema aeruginosa larvae (Coleoptera: Scarabidae) involvement of both granulocytes and oenocytoids in vivo phagocytosis. Tissue Cell. 2003;35(4):243-51. doi: http://doi.org/dm5rws.
Ling F, Yu XQ. Hemocytes from the tobacco hornworm Manduca sexta have distinct function in phagocytosis of foreign particles and self dead cells. Dev Comp Immunol. 2005;30(3):301-09. doi: http://doi.org/cbsprm.
Aladaileh S, Nair SV, Birch D, Raftos DA. Sydney rock oyster (Saccostrea glomerata) hemocytes: Morphology and function. J Invertebr Pathol. 2007;96(1):48-63. doi: http://doi.org/brt3j7.
Barraco MA, Loch CT. Ultrastuctural Studies of the Hemocytes of Panstrongylus megistus (Hemiptera:Reduviidae). Mem Inst Oswaldo Cruz. 1989;84:171-88. doi: http://doi.org/c7xwtp.
Stoepler TM, Castillo JC, Lili JT, Eleftherianos I. Hemocyte density increases with developmental stage in an immune-challenged forest caterpillar. PlosOne 2013;8(8):1-8. doi: http://doi.org/zgd.
Sorrentino RP, Carton Y, Govind S. Cellular Immune Response to parasite Infection in the Drosophila Lymph Gland is Developmentally Regulated. Develop Biol. 2002;243(1):65-80. doi: http://doi.org/dgk6nn.
Theopold U, Schmidt O, Söderhäll K, Dushay M. Coagulation in arthropods: defence, wound closure and healing. Trends in Immunol. 2004;25(6):289-94. doi: http://doi.org/fnxjz2.
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. José A. De Fuentes-Vicente, Ana E. Gutiérrez-Cabrera. (2022). Encyclopedia of Infection and Immunity. , p.953. https://doi.org/10.1016/B978-0-12-818731-9.00010-0.
2. Nicolás Salcedo-Porras, Carl Lowenberger. (2019). The innate immune system of kissing bugs, vectors of chagas disease. Developmental & Comparative Immunology, 98, p.119. https://doi.org/10.1016/j.dci.2019.04.007.
3. P. Azambuja, E.S. Garcia, P.J. Waniek, C.S. Vieira, M.B. Figueiredo, M.S. Gonzalez, C.B. Mello, D.P. Castro, N.A. Ratcliffe. (2017). Rhodnius prolixus: from physiology by Wigglesworth to recent studies of immune system modulation by Trypanosoma cruzi and Trypanosoma rangeli. Journal of Insect Physiology, 97, p.45. https://doi.org/10.1016/j.jinsphys.2016.11.006.
4. Nicolás Salcedo-Porras, Carl Lowenberger. (2021). Triatominae - The Biology of Chagas Disease Vectors. Entomology in Focus. 5, p.307. https://doi.org/10.1007/978-3-030-64548-9_14.
5. Victor Irungu Mwangi, Erika Gomez Martinez, Rejane Lima Leda, Maria Eugenia S.L.A. Catunda, Andiana de Souza Dias, Yuliana Padron Antonio, Maria das Graças V.B. Guerra. (2023). Resisting an invasion: A review of the triatomine vector (Kissing bug) defense strategies against a Trypanosoma sp infection. Acta Tropica, 238, p.106745. https://doi.org/10.1016/j.actatropica.2022.106745.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2015 Revista de la Facultad de Medicina

Esta obra está bajo una licencia Creative Commons Reconocimiento 3.0 Unported.
-