Inhibidores de dipeptidil dipeptidasa-IV: de la teoría a la práctica
Dipeptidil dipeptidase-IV inhibidors: from the theory to the practice
DOI:
https://doi.org/10.15446/revfacmed.v63n2.48820Palabras clave:
Diabetes mellitus tipo 2, Inhibidores de dipeptidil-peptidasa IV, Incretinas (es)Diabetes mellitus type 2, Dipeptidyl-peptidase IV inhibitors, Incretins (en)
Esta revisión de los inhibidores de dipeptidil peptidasa-IV busca motivar el uso racional de tal grupo farmacológico en la práctica diaria. Este grupo es una nueva opción terapéutica en monoterapia o terapia combinada para el tratamiento de los pacientes con diabetes mellitus tipo 2. En Colombia, se encuentran disponibles: sitagliptina, vildagliptina, saxagliptina y linagliptina.
Si bien todas las gliptinas tienen el mismo mecanismo de acción—aumentan la vida media del péptido similar al glucagón—, esta revisión presenta las diferencias entre sus propiedades farmacológicas, eventos adversos y perfil de seguridad. Estos medicamentos son de segunda o tercera línea para el tratamiento oral de los pacientes con diabetes mellitus tipo 2, o primera línea en los pacientes intolerantes a la metformina. Además, algunas de las ventajas que tienen son que: generan menor riesgo de hipoglucemia, tienen efecto neutro sobre el peso, son seguros en adultos mayores, disminuyen la variabilidad de la glucemia y, adicionalmente, se pueden utilizar en la enfermedad renal crónica avanzada, con o sin terapia de reemplazo renal, y en la insuficiencia hepática.
This review of dipeptidyl peptidase-IV inhibitors seeks to encourage the rational use of these drugs in daily practice; this group is a new therapeutic option in monotherapy or combination therapy for the treatment of patients with diabetes mellitus type 2. Sitagliptin, vildagliptin, saxagliptin and linagliptin are available in Colombia.
While all gliptins have the same mechanism of action—they increase the average life of glucagon-like peptide—, this review presents the differences among their pharmacological properties, adverse events and safety profile. These drugs are second or third-line for the oral treatment of patients with diabetes mellitus type 2, or first-line in patients intolerant to metformin. They also have some advantages like lower risk of hypoglycemia, neutral effect on weight, safety for the elderly, reduction of glycaemia variability; additionally, they can be used in advanced chronic kidney disease, with or without renal replacement therapy, and in liver failure
DOI: https://doi.org/10.15446/revfacmed.v63n2.48820
ARTÍCULO DE REVISIÓN
Inhibidores de dipeptidil dipeptidasa-IV: de la teoría a la práctica
Dipeptidil dipeptidase-IV inhibidors: from the theory to the practice
Yessica Agudelo-Zapata1, Álvaro Javier Burgos-Cárdenas1, Ángela Johana Díaz-Martínez1, Análida Elizabeth Pinilla-Roa1
1 Departamento de Medicina Interna, Facultad de Medicina. Universidad Nacional de Colombia. Bogotá, D.C.
Correspondencia: Yessica Agudelo-Zapata. Carrera 30 No. 45-03. Facultad de Medicina, Edificio 471. Departamento de Medicina Interna, oficina 510. Universidad Nacional Colombia, Sede Bogotá. Teléfono: +57 3218828745. Correo electrónico: yagudeloz@unal.edu.co.
Recibido: 02/02/2015 Aceptado:05/03/2015
Resumen
Esta revisión de los inhibidores de dipeptidil peptidasa-IV busca motivar el uso racional de tal grupo farmacológico en la práctica diaria. Este grupo es una nueva opción terapéutica en monoterapia o terapia combinada para el tratamiento de los pacientes con diabetes mellitus tipo 2. En Colombia, se encuentran disponibles: sitagliptina, vildagliptina, saxagliptina y linagliptina.
Si bien todas las gliptinas tienen el mismo mecanismo de acción—aumentan la vida media del péptido similar al glucagón—, esta revisión presenta las diferencias entre sus propiedades farmacológicas, eventos adversos y perfil de seguridad. Estos medicamentos son de segunda o tercera línea para el tratamiento oral de los pacientes con diabetes mellitus tipo 2, o primera línea en los pacientes intolerantes a la metformina. Además, algunas de las ventajas que tienen son que: generan menor riesgo de hipoglucemia, tienen efecto neutro sobre el peso, son seguros en adultos mayores, disminuyen la variabilidad de la glucemia y, adicionalmente, se pueden utilizar en la enfermedad renal crónica avanzada, con o sin terapia de reemplazo renal, y en la insuficiencia hepática.
Palabras clave: Diabetes mellitus tipo 2; Inhibidores de dipeptidil-peptidasa IV; Incretinas (DeCS).
Agudelo-Zapata Y, Burgos-Cárdenas AJ, Díaz-Martínez AJ, Pinilla-Roa AE. Inhibidores de dipeptidil dipeptidasa-IV: de la teoría a la práctica. Rev. Fac. Med. 2015;63(2):259-70.doi: https://doi.org/10.15446/revfacmed.v63n2.48820.
Summary
This review of dipeptidyl peptidase-IV inhibitors seeks to encourage the rational use of these drugs in daily practice; this group is a new therapeutic option in monotherapy or combination therapy for the treatment of patients with diabetes mellitus type 2. Sitagliptin, vildagliptin, saxagliptin and linagliptin are available in Colombia.
While all gliptins have the same mechanism of action—they increase the average life of glucagon-like peptide—, this review presents the differences among their pharmacological properties, adverse events and safety profile. These drugs are second or third-line for the oral treatment of patients with diabetes mellitus type 2, or first-line in patients intolerant to metformin. They also have some advantages like lower risk of hypoglycemia, neutral effect on weight, safety for the elderly, reduction of glycaemia variability; additionally, they can be used in advanced chronic kidney disease, with or without renal replacement therapy, and in liver failure.
Keywords: Diabetes mellitus, type 2; Dipeptidyl-peptidase IV inhibitors; Incretins (MeSH).
Agudelo-Zapata Y, Burgos-Cárdenas AJ, Díaz-Martínez AJ, Pinilla-Roa AE. [Dipeptidil dipeptidase-IV inhibidors: from the theory to the practice]. Rev. Fac. Med. 2015;63(2):259-70.Spanish. doi: https://doi.org/10.15446/revfacmed.v63n2.48820.
Introducción
La diabetes mellitus tipo 2 (DM2) es una enfermedad de alta prevalencia mundial, en Colombia llega hasta el 8 % (1,2). Actualmente en el país, los medicamentos para el tratamiento farmacológico de esta enfermedad que se manejan en el Plan Obligatorio de Salud (POS) son: sulfonilureas (glibenclamida), biguanidas (metformina), insulina y sus análogos (3). Mientras que los Inhibidores de Dipeptidil-Peptidasa-IV (IDPP-IV) no se encuentran disponibles en el POS, a pesar de contar con una vasta literatura médica que los respalda desde el 2006; no obstante, el clínico puede prescribirlos realizando los trámites administrativos correspondientes. Situación similar ocurre con los análogos de péptido relacionados al glucagón (GLP-1) que fueron aprobados por la FDA (administración de alimentos y medicamentos) a partir del año 2005 (4).
Ahora bien, la metformina es la piedra angular en el tratamiento oral de los pacientes con DM2; así, a la luz de la evidencia actual este medicamento es la recomendación de primera línea en las guías clínicas (5), debido a su efecto sobre la hemoglobina glicosilada (HbA1c), que es superior al de cualquier medicamento oral incluyendo a los IDPP-IV. Sin embargo, la terapia oral para el tratamiento de la DM2 es limitada y hay pocas opciones cuando se trata de situaciones clínicas, tales como: la intolerancia a la metformina, la enfermedad renal crónica (ERC) avanzada, la insuficiencia hepática severa, rechazo del paciente a la insulina y al uso de agujas, sumado al alto riesgo de hipoglucemia (6).
Los IDPP-IV disponibles en Colombia, en orden de introducción, son: sitagliptina, vildagliptina, saxagliptina y linagliptina para el tratamiento de los pacientes con DM2. El objetivo de esta revisión es brindar información sobre los IDPP-IV disponibles, sus diferencias e indicaciones, para una prescripción racional y adecuada.
Efecto incretina
Las incretinas son hormonas producidas principalmente por el intestino que regulan la acción endocrina del páncreas (7). En los años sesenta se describió el efecto incretina como la acción que tiene la ingesta oral de glucosa sobre la secreción de insulina en el páncreas, mediada por hormonas (4,8). La primera en ser descrita fue el polipéptido insulinotrópico dependiente de glucosa (GIP), producido por las células K del intestino delgado (9). Posteriormente, se descubrió el GLP-1, que tiene mayor efecto sobre la secreción de insulina; esta incretina es sintetizada por las células L que se encuentran en el yeyuno, luego es metabolizada rápidamente por la acción de la enzima Dipeptidil Peptidasa IV (DPP-IV) convirtiéndola en un metabolito de menor potencia (Figuras 1 y 2) (7).
Por consiguiente, el aumento de la concentración de GLP-1 en plasma es el efecto farmacológico de los IDPP-IV, que finalmente se traduce en lo siguiente: a nivel de las células β del páncreas aumenta la síntesis de insulina, estimula el crecimiento de esta célula y evita la apoptosis (10). De forma sistémica la hormona disminuye: el apetito, el vaciamiento gástrico, el peso corporal, la presión arterial y, aunque aún en estudio, favorece el ingreso de glucosa al miocardio afectado por isquemia (7,11). Sin embargo, no se ha demostrado que los IDPP-IV tengan los mismo efectos farmacológicos que el GLP-1 endógeno y sus análogos (Figura 1), por ejemplo en el peso corporal, ya que los IDPP-IV no disminuyen el peso (12).
La DPP-IV o adenosina desaminasa es un miembro de las peptidasas de serina que incluyen las peptidasas de unión a las membranas como la proteína de activación de fibroblastos, enzimas citoplasmáticas como las DPP-VIII y IX; además, hay dipeptidasas no enzimáticas como las DPP-VI y X que están presentes en las membranas neuronales (Figura 2) (13).
La DPP-IV se encuentra expresada en la mayoría de los tejidos humanos, principalmente a nivel endotelial, por lo que se ha relacionado con la salud cardiovascular, aunque aún no se ha descrito con exactitud por qué se relaciona su inhibición con aumento de descompensación de falla cardiaca, en pacientes de riesgo (14); también se encuentra, en una forma soluble en el plasma. A nivel de los linfocitos T, la DPP-IV se convierte en el CD26, por lo cual la inhibición de la enzima se ha relacionado con un aumento teórico de infecciones y de trastornos de autoinmunidad, pero sin una clara función fisiopatológica. Dado el rol de algunas Dipeptidil-Peptidasas (DPP) como supresores tumorales, su inhibición se ha relacionado con el riesgo de cáncer pancreático, de colon, próstata, pero si ninguna evidencia clínica. Finalmente, se indica que la alta selectividad de los inhibidores por la DPP-IV es lo que les permite ser seguros (14).
Los IDPP-IV disponibles en el mundo actualmente incluyen sitagliptina, vildagliptina, saxagliptina, linagliptina y alogliptina. Los tres primeros pertenecen al grupo de los péptido-miméticos y los dos últimos al grupo de los derivados de xantinas. A excepción de la vildagliptina, el resto han sido aprobados por la Food and Drug Administration (FDA), para mayores de 18 años pero no en mujeres gestantes o lactantes (4). Otras gliptinas que se encuentran en fase III de ensayos clínicos incluyen dutogliptina y gemigliptina, entre otras (Tabla 1) (15).
En este artículo se presentan sólo los IDPP-IV disponibles en Colombia y en orden de aparición histórica: sitagliptina, vildagliptina, saxagliptina y linagliptina.
Sitagliptina
Es un IDPP-IV altamente selectivo, fue aprobado por la FDA en el año 2006 (4). En Colombia, está disponible la presentación oral sola o combinada con metformina.
Farmacodinamia y farmacocinética
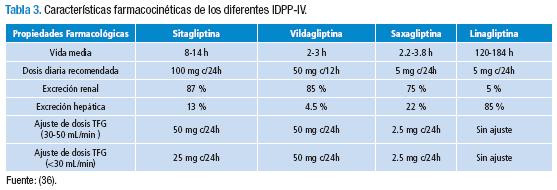
La acción inhibitoria sobre la DPP-IV es dosis dependiente (16). Tiene una biodisponibilidad del 87 %, unión a proteínas del 34-46 %, una vida media de 10-12 horas y la eliminación es principalmente renal (75-80 %) y el resto es metabolizado por los citocromo (CYP): CYP3A4 y CYP2C8; por lo que presenta interacción medicamentosa con algunos fármacos, entre los que se encuentran: macrólidos, anti-retrovirales, antidepresivos, bloqueadores de canales de calcio, esteroides, entre otros (17).
En pacientes con deterioro de la función renal la sitagliptina requiere ajuste de dosis; por tanto, el clínico, debe calcular la Tasa de Filtración Glomerurlar (TFG) por medio de la fórmula de Cockroft y Gault (18). En el caso de TFG de 30-50 mL/min se disminuye 50 % de la dosis y si la TFG es menor de 30 mL/min a un 25 % de la dosis (17,19). En el caso de insuficiencia hepática leve a moderada (Child-Pugh A o B) su farmacocinética no se altera y podría utilizarse con precaución en la insuficiencia severa (17,20).
Efectividad clínica
Se han desarrollado varios estudios para evaluar la efectividad de la sitagliptina en monoterapia o en combinación con otros agentes antidiabéticos (17). Como monoterapia contra placebo, se ha mostrado que disminuye de manera estadísticamente significativa los valores de HbA1c, glucemia basal y postprandial a las 12, 18 y 24 semanas (17,21); en tanto que el mayor beneficio se evidenció en pacientes con HbA1c por encima de 9% (22). Al evaluar la sitagliptina en terapia combinada con metformina vs. metformina sola, se encontró que ambas intervenciones disminuyen los valores de HbA1c, glucosa en ayunas y posprandial; no obstante, el efecto era mayor en los paciente que recibían terapia combinada, de igual manera, en pacientes que recibían pioglitazona al adicionar sitagliptina, se logró llevar a la meta de HbA1c <7% a más sujetos (17).
Asimismo, al comparar la metformina vs la sitagliptina en un meta-análisis, esta última demostró ser inferior en mejorar la sensibilidad a la insulina, pero los dos medicamentos tienen efecto similar en control de peso, disminución de la HbA1c, y la función de la célula β (23). La reducción de HbA1c como monoterapia puede ser de hasta 0.94 % y puede haber una reducción adicional hasta 1.1 % cuando se usa en terapia combinada con insulina (24,25). Además, la sitagliptina ha sido comparada con vildagliptina y algunos estudios sugieren superioridad de ésta última en desenlaces como control glucémico circadiano con disminución significativa de hiperglucemias (26).
Seguridad y efectos adversos
La sitagliptina es segura y bien tolerada con bajo riesgo de hipoglucemia y efecto nulo en el peso (27). Se han reportado pocos efectos adversos como: nasofaringitis, infecciones del tracto respiratorio superior, cefalea, lumbalgia, osteoartritis, dolor en extremidades, aumento del conteo leucocitario, de los niveles de ácido úrico y discreta disminución de la fosfatasa alcalina (17). Sin embargo, esto no ha representado un mayor riesgo de hospitalización por todas las causas en estudios retrospectivos (28–30), aunque aún hay controversia en la posible asociación del uso de sitagliptina con mayor riesgo de hospitalización por falla cardiaca (31).
Entre 2006 y 2009, se reportaron 88 casos de pancreatitis (32), en los que no hay claridad sobre el mecanismo fisiopatológico; sin embargo, un meta-análisis evidenció que el riesgo de pancreatitis es igual al placebo (33). En este sentido, se han reportado reacciones de hipersensibilidad como: angioedema, condiciones cutáneas exfoliativas, anafilaxis y hay un reporte de caso de reacción medicamentosa con eosinofilia y síntomas sistémicos (4,34).
En resumen, la sitagliptina no es superior a la metformina disminuyendo la HbA1c, pero se puede administrar en caso de Enfermedad Renal Crónica (ERC) con ajuste de dosis y, en insuficiencia hepática sin ajuste tiene efecto nulo en el peso, tiene efecto positivo sobre la célula β y es importante tener en cuenta la interacción medicamentosa dado su metabolismo por el CYP450. Se están llevando a cabo estudios para evaluar su seguridad cardiovascular (30,31).
Vildagliptina
La vildagliptina es un IDPP-IV selectivo, fue aprobado por la agencia europea de medicamentos en el año 2008, y pertenece al grupo de las cianopirrodilidionas y los péptido-miméticos (27,35).
Farmacodinamia y farmacocinética
Con respecto a la farmacodinamia, la vidagliptina tiene acción sobre otras enzimas DPP como la VIII, IX y la proteína activadora de fibroblastos (6). Además, produce una inhibición reversible sobre los dominios S1 y S2 de la DPP-IV, formando un enlace covalente sobre el residuo de serina 630 (36).
Se administra por vía oral, en aproximadamente 1.5 horas, tiene una biodisponibilidad del 85%; con dosis de 100 mg a los 45 minutos bloquea el 95% de la actividad de la DPP-IV durante 12 horas (37). El volumen de distribución es aproximadamente de 71 L y se une a proteínas en un 9.3%.
En pacientes, con función renal normal, la vida media es de 2 a 3 horas, pero el efecto del medicamento perdura durante más tiempo, dado el bloqueo competitivo y de la disociación lenta sobre la enzima (38). Adicional a esto, se metaboliza a través de la hidrólisis, con poca participación del CYP-450, lo cual es atractivo para uso en pacientes poli-medicados (39). Hasta el momento no se ha encontrado interacciones medicamentosas con los antidiabéticos tradicionales, antihipertensivos o medicamentos de rango terapéutico estrecho como la warfarina o la digoxina (40). Su excreción se produce, principalmente a nivel renal, con un porcentaje de eliminación hepática del 13% (40).
En cuanto a la farmacocinética en pacientes con ERC, se encontró que la concentración de vildagliptina no aumenta con el deterioro renal, como sí lo hace su metabolito inactivo hidrolizado, el LAY 151. Por lo que se recomienda la utilización de una sola dosis de 50 mg día en pacientes con TFG por debajo de 50 mL/min (41). En un estudio retrospectivo con 15 pacientes sometidos a diálisis peritoneal y hemodiálisis, el uso de vildagliptina estuvo asociado a una reducción, significativamente estadística, de HbA1c y albúmina glicosilada; sin embargo, no se midieron eventos adversos (42). Por otra parte, un meta-análisis de vildagliptina en monoterapia o terapia combinada, en pacientes con ERC, no encontró aumento de los eventos adversos en esta población (43). De hecho se han realizado comparaciones aleatorizadas de vildagliptina contra placebo en pacientes con ERC de diferentes estadios, sin encontrar una asociación entre su uso y un mayor número de reacciones adversas (44,45).
Ahora bien, en insuficiencia hepática de leve a severa no hay alteración de los parámetros farmacocinéticas, ni mayor grado de reacciones adversas; sin embargo, no se ha probado su uso, dado el antecedente de elevación de transaminasas en casos aislados (43,46).
Efectividad clínica
La eficacia clínica de la vildagliptina ha sido demostrada tanto en monoterapia como en terapia combinada con antidiabéticos orales e insulina. Existen tres estudios que evalúan la efectividad de vildagliptina como monoterapia, en ellos se demostró una reducción de la HbA1c de aproximadamente 1 %, sin evidencia de aumento de peso o hipoglucemia. Asimismo, se intentó evaluar la no inferioridad en relación a la metformina, pero no se obtuvieron resultados concluyentes. La reducción de la HbA1c fue del 1% en promedio para vildagliptina y 1.4% para la metformina (47,48). De igual manera, no se ha encontrado que sea inferior a las tiazolidionas o sulfonilureas (49).
En cuanto a la terapia combinada, la adición de vildagliptina al tratamiento de pacientes no controlados en monoterapia —con metformina, glimepiride, pioglitazona o insulina— resultó en una disminución adicional de la HbA1c (49). Estos hallazgos son congruentes con el estudio EDGE, el cual evaluó de forma prospectiva, en más de 45000 pacientes la efectividad de la adición de vildagliptina como segunda línea de tratamiento, se observó disminución de la HbA1c en todas las combinaciones (50).
Seguridad y eventos adversos
Vildagliptina es un medicamento seguro, no hay reportes de eventos adversos graves. Se ha asociado a angioedema en pacientes que consumen inhibidores de la enzima convertidora de angiotensina II (IECA), sin reportes de anafilaxia (27). Existe una alerta de seguridad en pacientes con elevación de las transaminasas, por lo que en insuficiencia hepática su prescripción está en entredicho. En un meta-análisis de estudios clínicos aleatorizados no se encontró asociación de vildagliptina con desenlaces cardiovasculares mayores (27); está pendiente la publicación del estudio Effect of Vildagliptin on Left Ventricular Function in Patients With Type 2 Diabetes and Congestive Heart Failure (VIVID), para valorar los efectos de vildagliptina en pacientes con falla cardiaca (13). De otra parte, este medicamento no ha sido asociado a pancreatitis ni al desarrollo de neoplasias (27).
En síntesis, este IDPP-IV es una opción en pacientes con DM2, tanto en primera como en segunda línea de tratamiento, polimedicados, con variabilidad glucémica y en ERC. Se debe evitar su uso en pacientes con insuficiencia hepática y monitorizar aquellos que consumen IECA.
Saxagliptina
Esta gliptina fue descubierta en el año 2005, y aprobada por la FDA en el año 2009 (51). Al igual que la vildagliptina inhibe DPP-VIII, DPP-IX y la proteína de activación de los fibroblastos (52,53).
Farmacodinamia y farmacocinética
La saxagliptina tiene una biodisponibilidad del 60 %. Esta es metabolizada hepáticamente por el CYP-450 específicamente por la enzima CYP 3A4/5, a un metabolito activo llamado 5 hidroxisaxagliotina, este metabolito es también un inhibidor selectivo, reversible y competitivo de la DDP-4. Después de dos semanas, la inhibición de la DPP-IV por saxagliptina es de aproximadamente 50 a 79%, dependiendo de la dosis (54).
Las propiedades farmacocinéticas han sido estudiadas en pacientes con ERC, donde se demostró un perfil de seguridad del medicamento, con un ajuste de dosis a 2.5 mg/día en sujetos con TFG menor de 60 mL/min. Dado el metabolismo hepático, se debe tener en cuenta las interacciones farmacológicas con múltiples medicamentos que son sustrato del CYP-450 como: simvastatina, diltiazem, ketoconazol, rifampicina, entre otros (53); sin embargo, los estudios hasta ahora sugieren que en enfermedad hepática no se requiere ajuste de dosis (55).
Eficacia clínica
Este fármaco en monoterapia no es superior a metformina en cuanto a la disminución de la HbA1c, como se muestra en reportes de ensayos clínicos, donde tiene efecto de reducción hasta del 0.9% de la HbA1c. Por su parte, en combinación con metformina por 24 semanas mostró una disminución hasta del 2.5% de HbA1c, reducción que es superior a la de cada fármaco en monoterapia (54).
Seguridad y eventos adversos
La saxagliptina es generalmente segura y bien tolerada (56), presenta bajo riesgo de hipoglucemia. Incluso algunos estudios han reportado un riesgo igual al placebo (53). Con respecto a otros eventos adversos, como la pancreatitis, un meta-análisis reciente sobre la seguridad reportó igual riesgo que el placebo (57). Sin embargo, el estudio Saxagliptin Assessment of Vascular Outcomes Recorded in Patients with DiabetesMellitus—Thrombolysis inMyocardial Infarction 53 (SAVOR-TIMI 53) demostró una relación con el aumento de hospitalización por falla cardiaca en pacientes con historia previa de ésta, ERC y péptido natriurético elevado, aunque la fisiopatología de esta asociación no está clara (58).
De otro lado, se ha reportado una mayor incidencia de infección urinaria e infecciones del tracto respiratorio, pero hasta el momento no hay datos que demuestren alteración del sistema inmune por ningún IDPP-IV. Otros efectos son la cefalea y las reacciones dermatológicas (53).
En Colombia, este fármaco se introdujo en el 2010. Luego, en el 2012 se realizó un estudio de costo-efectividad financiado por la compañía que lo fabrica, mostrando que en Colombia el uso de saxagliptina es costo efectivo (59).
Finalmente, la saxagliptina no es superior a la metformina en la disminución de la HbA1c, pero al combinarlas es más efectiva. Cabe aclarar que tiene beneficios por la farmacocinética en caso de ERC, disminución en eventos de hipoglucemia, efecto nulo en el peso y efecto positivo sobre la célula β. Además, es importante tener en cuenta el aumento en hospitalizaciones por falla cardiaca en los pacientes con este riesgo y la interacción medicamentosa dado su metabolismo por el CYP-450.
Linagliptina
La linagliptina es el más reciente de los IDPP-IV, fue descubierto en el 2007 (60) y fue aprobada por la FDA en el año 2011 (4).
Famacodinámica y farmacocinética
Se trata de un inhibidor altamente selectivo, competitivo y reversible de la DPP-IV, provee una inhibición hasta del 80 % de la enzima, siendo el IDDP-IV más selectivo. Se encuentra disponible en presentación oral con dosis de 5 mg (en dosis única diaria), en monoterapia o combinada con metformina (61,62).
La linagliptina tiene una biodisponibilidad del 30 %, una vida media de 131 horas, debido a su unión a proteínas plasmáticas. Se elimina principalmente por vía hepática en un 85 %, y en un 5 % por vía renal con un volumen de distribución de 1,110 L. Este medicamento no tiene variación en su farmacocinética en pacientes con ERC (incluso avanzada), por lo que no se requiere ajustar la dosis (63). Además, presenta metabolismo hepático mínimo, es convertida a un metabolito inactivo llamado CD1790. Como este medicamento inhibe la enzima CYP-3A4 del CYP-450, con efecto mínimo a moderado, no tiene interacciones significativas, a excepción de la rifampicina que aumenta el metabolismo de la linagliptina (61).
Efectividad clínica
La linagliptina como monoterapia mostró disminución de la HbA1c en 0.69 %, la glucemia en ayunas en 23.3 mg/dL a los 6 meses y la posprandial de 33.5 mg/dL (62). Cuando se utiliza en terapia combinada con metformina se logra una disminución adicional de 0.64 % de la HbA1c y de la glucemia en ayunas de 21.1 mg/dL (61,62). En combinación con pioglitazona, sulfonilureas e insulina también se obtuvo una disminución de HbA1c >0.5 % (61). Asimismo, el efecto de linagliptina sobre el peso es neutro (61–63).
Seguridad y efectos adversos
La linagliptina tiene un perfil de seguridad similar al placebo y bajo riesgo de hipoglucemia (61–63). Los eventos adversos más frecuentes reportados en 22 estudios aleatorizados fueron: cefalea (3.1 %), nasofaringitis (5,8 %) e infección del tracto respiratorio superior (3.3 %) (61). Menos de 2.5 % de las personas que recibieron al menos una dosis de linagliptina reportaron tos, hiperlipidemia o aumento en el peso (62).
Aunque aún no se disponen estudios de mortalidad o desenlaces cardiovasculares a largo plazo, en los estudios realizados hasta ahora, no se han evidenciado cambios en valores de la presión arterial ni de la frecuencia cardiaca (62). En un meta-análisis el riesgo de eventos cardiovasculares no aumento (62); sin embargo, dado que se requieren más estudios, actualmente se está desarrollando el estudio CAROLINA, que compara de manera prospectiva la seguridad cardiovascular del medicamento comparado con glimepiride y, el estudio CARMELINA, que evalúa desenlaces cardiovasculares y renales con linagliptina en pacientes con DM2 con alto riesgo vascular (61). También se estudió la incidencia de pancreatitis, la cual fue comparable cuando los pacientes fueron tratados con placebo (63).
En resumen, linagliptina es el más reciente de los IDPP-IV, tiene una vida media larga y por su metabolismo de predominio hepático es de elección en pacientes con disminución de la TFG, el medicamento aunque no es superior a metformina, para disminuir la HbA1c mantiene un efecto neutro en el peso, es relativamente seguro aunque están pendientes estudios sobre su riesgo cardiovascular.
IDPP-IV en la práctica clínica
Definitivamente los IDPP-IV están indicados como monoterapia y terapia combinada de segunda y tercera línea en pacientes mayores de 18 años con DM2 de acuerdo a las guías clínicas de la Asociación Americana de Diabetes (ADA) 2015 y la Federación Internacional de Diabetes (IDF) en las guías clínicas del 2015 y 2014, respectivamente (5,64).
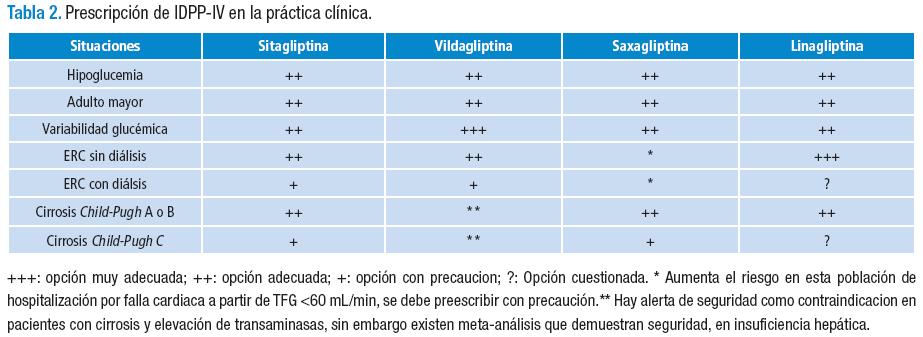
A continuación, se presentan algunos escenarios especiales en los que los IDPP-IV pueden optimizar el control glucémico de los pacientes y, en la Tabla 2 se consolidan los diferentes indicadores clínicos para su prescripción:
Primer caso clínico en hipoglucemia
Paciente de 80 años con DM2, sin ERC, en tratamiento con metformina y glibenclamida, quien refiere síntomas neurovegetativos intermitentes sugestivos de hipoglucemia durante el día, los cuales —durante el seguimiento— se confirmaron como hipoglucemia; por tanto, el médico tratante considera importante continuar con metformina, asociarlo a otro antidiabético oral y suspender glibenclamida por la hipoglucemia.
¿Los IDPP-IV están indicados en este escenario?
El riesgo de hipoglucemia en el tratamiento con IDPP-IV en monoterapia es igual al placebo, dado que las gliptinas incrementan la vida media del GLP-1, de forma proporcional a la ingesta de carbohidratos (65). Por lo tanto, al no haber liberación de GLP-1 durante el ayuno, no hay efecto farmacológico de las gliptinas. En este sentido, este grupo farmacológico puede ser considerado en pacientes con riesgo o evidencia de hipoglucemia, por ejemplo, el adulto mayor. En conclusión, en este escenario se podría utilizar cualquiera de los IDPP-IV disponibles en el país (36).
Segundo caso clínico en variabilidad glucémica
Paciente de 60 años quien consulta por DM2 y glucometrías que varían entre 50 y 400 mg/dL durante el día y HbA1c de 8%. Actualmente está en tratamiento con dosis de metformina (850 mg, cada 8 horas) y glibenclamida (5 mg cada 12 horas), reusándose al tratamiento con insulina. El médico, en acuerdo con el paciente, quiere continuar con un esquema oral, que disminuya la variabilidad en las glucometrías, con optimización del control de la HbA1c.
¿Los IDPP-IV mejoran la variabilidad glucémica, hay diferencia entre ellos?
Aunque el concepto de variabiliadad glucémica es controvertido, se ha indicado por diversos autores que podría impactar en la aparición de complicaciones tanto macrovasculares como microvasculares, a pesar de una HbA1c en metas (36). Los IDDP-IV se han propuesto como una opción por su mecanismo de acción dependiente de la concentración de la glucemia posprandial. Sin embargo, no hay una evidencia fuerte que demuestre que estos medicamentos impactan en desenlaces duros como riesgo de hospitalizaciones o mortalidad cardiovascular o por otras causas, pero sí una evidencia débil que muestra que la vildagliptina es superior a la sitagliptina en disminuir la variabilidad de glucemia diaria y, dicho resultado se ha correlacionado con una disminución del grosor de la íntima media carotidea. Por lo cual, un paciente con estas características se podría beneficiar de un IDPP-IV, debería continuar con la metformina, suspender la glibenclamida e insistir en inicio de la terapia con insulina (36,65,66).
Tercer caso clínico, en ERC sin terapia de reemplazo renal
Paciente obeso con índice de masa corporal de 34 Kg/m2, con nefropatía diabética avanzada TFG 20 mL/min (estadio 4) (18), con promedio de glucometrías de los últimos 15 días de 200 y HbA1c 8.5 %. Este paciente está en tratamiento con insulina análoga de larga acción con dosis de 1 UI/Kg/día. Dada la obesidad y la dosis de insulina, se considera importante adicionar un medicamento oral, pero la metformina está contraindicada por el estadio de ERC.
En este caso ¿Se pueden utilizar los IDPP-IV?, y ¿Cuáles son más seguros?
Hay evidencia suficiente para utilizar los IDDP-IV en la ERC en cualquiera de sus estadios —inclusive en el estadio 5 con o sin diálisis— (67), pero, con ajuste de la dosis, en el caso de: sitagliptina, vildagliptina y saxagliptina (Tabla 3). En el caso de saxagliptina, los resultados del estudio SAVOR TIMI 53 reportan que hay mayor riesgo de falla cardiaca en los pacientes con ERC (TFG menor de 60 mL/min) cuando son tratados con saxagliptina (58); por lo cual, en este escenario, debe haber precaución en la prescripción. Quizás el paciente se podría beneficiar más de gliptinas como la linagliptina, que no requiere ajuste de la dosis y además se está evaluando su eficacia, en el estudio MARLINA, para disminuir microalbuminuria. O también de la sitagliptina y la vildagliptina que requieren ajuste (4,68).
Cuarto caso clínico en pacientes con ERC y en terapia de reemplazo renal
Paciente en terapia de reemplazo renal a quien se desea tratar con hipoglucemiante oral, al estar contraindicado metformina y glibenclamida.
¿Cuál IDPP-IV estaría indicado?
El uso de IDPP-IV en pacientes en hemodiálisis no es frecuente, no obstante, hay algunos estudios que sugieren seguridad y eficacia similar a la del paciente sin terapia de reemplazo renal (42,67).
Con respecto a la sitagliptina, hay un estudio que demostró una disminución en HbA1c de 0.7 % comparado con 0.2 % en el grupo placebo/glipizida y la incidencia de eventos adversos fue similar en ambos grupos. Con relación a la saxagliptina, esta aumenta el riesgo de hospitalización por falla cardiaca (58), aunque se ha propuesto que requiere ajuste en hemodiálisis siendo necesario administrar una dosis suplementaria posterior a cada sesión, ya que cada 4h de hemodiálisis remueve aproximadamente 23% de la saxagliptina (67).
Por otro lado, en el caso de la vildagliptina se ha realizado un estudio con 15 pacientes en terapia de reemplazo renal, que recibían dosis de 50 mg al día, resultó ser efectiva para la reducción de la HbA1c, sin reporte de eventos adversos (42). Finalmente, la linagliptina tiene un perfil de eliminación ideal para pacientes en terapia de reemplazo renal por su metabolismo hepático, pero la evidencia es limitada (67).
En síntesis, los IDPP-IV deben ser usados con precaución en ERC y diálisis, ya que la evidencia es muy limitada y, por lo tanto, su seguridad y eficacia están aún en estudio.
Quinto caso clínico, paciente con insuficiencia hepática
Pacientes con insuficiencia hepática (cirrosis en cualquier estadio Child-Pugh A, B, C) y DM2; el médico tratante quiere utilizar un mediamento oral para el tratamiento de la DM2.
¿Los IDDP-IV se pueden utilizar? ¿Cuáles son más seguros?
La farmacocinética de los IDDP-IV ha sido estudiada en diferentes estadios de insuficiencia hepática y ninguno de los medicamentos requiere ajuste de dosis (46). En cuanto a su seguridad, la vildagliptina es la única que ha tenido reportes de hepatotoxicidad demostrada por elevación de transaminasas, pero hay meta-análisis de estudios clínicos aleatorizados que no mostraron aumento de reacciones adversas hepáticas (40,43). Ante el antecedente sobre su seguridad, su uso en esta situación ha sido limitado. El resto de las gliptinas no han sido asociadas a reacciones adversas hepáticas. La saxagliptina tiene estudios sobre su seguridad en insuficiencia hepática leve y moderada, en tanto que se puede utilizar con precaución en insuficiencia hepática severa (55).
Por su parte, la sitagliptina tiene estudios en el tratamiento de pacientes con DM2 y hepatitis C, con efectos pleiotrópicos hepatoprotectores en enfermedades hepáticas (69–71). Es segura en insuficiencia hepatica leve y moderada, se podría administrar con precaución en la severa (72).
Finalmente, la linagliptina, el último IDDP-IV disponible en Colombia, a pesar de que tiene un metabolismo de predominio hepático, tiene estudios que demuestran seguridad en insuficiencia hepática leve y moderada, pero no tiene ninguna evidencia en la severa, por lo cual no se debe utilizar en dicho estadio (46,72).
En resumen, un paciente con cirrosis Child-Pugh A o B con DM2 puede ser tratado con los IDPP-IV disponibles en Colombia. Pero, se advierte que para la vildagliptina hay una alerta de seguridad; asimismo, la sitagliptina y la saxagliptina podrían usarse con precaución en pacientes con Child-Pugh C.
Controversia de los IDPP-IV en falla cardiaca
El uso de IDPP-IV en falla cardíaca es controversial. Hay resultados contradictorios entre estudios preclínicos y clínicos. Los estudios preclínicos muestran un rol benéfico en la fisiopatología de la falla cardíaca (73). Esto no ha sido tan claro en la clínica, dado los resultados del estudio SAVOR TIMI 53, que muestra un aumento de hospitalizaciones por falla cardíaca y con meta-análisis han publicado que podría ser un efecto de clase (74,75).
Mientras que el estudio VIVIDD con vidagliptina, hasta el momento ha mostrado un efecto benéfico en los pacientes con falla cardíaca con fracción de eyección disminuida, resultados que aún no están publicados. Están pendientes los resultados de los ensayos TECOS para evaluar sitagliptina y desenlaces cardiovasculares, a su vez, el CAROLINA que evaluará a linagliptina como ya se indicó. Estos ensayos clínicos pueden arrojar luz sobre las posibles ventajas e inconvenientes de la inhibición de las DPP-IV en pacientes con daibetes y antecedentes de enfermedades cardiovasculares.
No obstante, un estudio observacional prospectivo mostró que los análogos de GLP-1 y los IDPP-IV no aumentaron el riesgo de falla cardíaca (76). Dada la controversia en este tema, el uso de estos medicamentos debe hacerse con precaución en este tipo de pacientes.
Conclusiones
Los IDPP-IV, a la luz de la evidencia expuesta, son una opción en monoterapia o en terapia combinada para el tratamiento oral de los pacientes con DM2. Aunque estos medicamentos no han mostrado superioridad a la metformina, sí hay situaciones especiales en las que los IDPP-IV se convierten en la primera opción oral de tratamiento o pueden complementarlo, tales como: la intolerancia a metformina, la ERC desde leve a moderada pero, particularmente en estadios avanzados de enfermemdad renal crónica IV y V (TFG menor de 30 mL/minuto), la insuficiencia hepática leve a moderada, el alto riesgo de hipoglucemia e hipoglucemia recurrente demostrada y la alta variabilidad glucémica.
Hay que tener en cuenta, que la efectividad de estos medicamentos aumenta cuando el paciente tiene pobre control glucémico, ya que cualquier antidiabético muestra un efecto mayor en reducción de HbA1c cuando se parte de valores altos (77).
Finalmente, las diferencias farmacocinéticas entre los IDPP-IV disponibles hacen que según el escenario clínico se prefiera uno sobre otro. Aunque los estudios realizados hasta ahora no reportan eventos adversos significativos, aún están en desarrollo los estudios de seguridad para desenlaces cardiovasculares mayores y mortalidad.
Conflicto de intereses
Ninguno declarado por los autores.
Financiación
Ninguna declarada por los autores.
Agradecimientos
Ninguno declarado por los autores.
Referencias
1. Aschner P. [Epidemiology of diabetes in Colombia]. Av en Diabetol [Internet]. 2010 [cited 2015 apr 09];26:95-100. Spanish. doi: http://doi.org/f2jmjd.
2. Aschner P, Aguilar-Salinas C, Aguirre L, Franco L, Gagliardino JJ, de Lapertosa SG, et al. Diabetes in South and Central America: an update. Diabetes Res Clin Pract [Internet]. 2014 [cited 2015 apr 09];103(2):238-43. doi: http://doi.org/f2qh6q.
3. Ministerio de Salud y Protección Social. Resolución 005521 de 2013. Por la cual se define, aclara y actualiza integralmente el Plan Obligatorio de Salud (POS). Colombia [Internet]; 2013 [cited 2015 apr 09]. p. 1-146. Available from: http://goo.gl/DJQjfZ.
4. Neumiller JJ. Incretin-Based Therapies. Med Clin North Am [Internet]. 2015 [cited 2015 apr 09];99(1):107-29. doi: http://doi.org/2qp.
5. American Diabetes Association. Approaches to Glycemic Treatment. (Sec. 7). In Standards of Medical Care in Diabetes-2015. Diabetes Care [Internet]. 2015 [cited 2015 apr 09];38(Suppl 1):S41-8. doi: http://doi.org/3hr.
6. Scheen AJ. A review of gliptins in 2011. Expert Opin Pharmacotherapy [Internet]. 2012 [cited 2015 apr 09];13(1):81-99. doi: http://doi.org/c3fxf4.
7. Baggio LL, Drucker DJ. Biology of incretins: GLP-1 and GIP. Gastroenterology [Internet]. 2007 [cited 2015 apr 09];132(6):2131-57. doi: http://doi.org/c8rk2h.
8. Elrick H, Stimmler L, Hlad CJ, Arai Y. Plasma insulin response to oral and intravenous glucose administration. J Clin Endocrinol Metab [Internet]. 1964 [cited 2015 apr 09];24(10):1076-82. doi: http://doi.org/fvjwqw.
9. Campbell JE, Drucker DJ. Pharmacology, physiology, and mechanisms of incretin hormone action. Cell Metab [Internet]. 2013 [cited 2015 apr 09];17(6):819-37. doi: http://doi.org/3hs.
10. White JR. Dipeptidyl peptidase-IV inhibitors: Pharmacological profile and clinical use. Clinical Diabetes [Internet]. 2008 [cited 2015 apr 09];26(2):53-7. doi: http://doi.org/d74xkx.
11. Ussher JR, Drucker DJ. Cardiovascular biology of the incretin system. Endocr Rev [Internet]. 2012 [cited 2015 apr 09];33(2):187-215. doi: http://doi.org/2p3.
12. Brown DX, Evans M. Choosing between GLP-1 receptor agonists and DPP-4 inhibitors: A pharmacological perspective. J Nutr Metab [Internet]. 2012 [cited 2015 apr 09]. p. 1-10. doi: http://doi.org/3ht.
13. Mulvihill EE, Drucker DJ. Pharmacology, physiology, and mechanisms of action of dipeptidyl peptidase-4 inhibitors. Endocr Rev [Internet]. 2014 [cited 2015 apr 09];35(6):992-1019. doi: http://doi.org/3hw.
14. Zhao Y, Yang L, Zhou Z. Dipeptidyl peptidase-4 inhibitors: Multitarget drugs, not only antidiabetes drugs. J Diabetes [Internet]. 2014 [cited 2015 apr 09];6(1):21-9. doi: http://doi.org/3hx.
15. López-Jaramillo P, Velandia C, Castillo G, Sánchez T, Álvarez J. [Incretin analogs and inhibitors of DPP-4: which is their role in the primary prevention of cardiovascular diseases?] Rev Colomb Cardiol [Internet]. 2013 [cited 2015 apr 09];20(5):287-99. Spanish. Available from: http://goo.gl/0mCNym.
16. Bergman AJ, Stevens C, Zhou Y, Yi B, Laethem M, de Smet M, et al. Pharmacokinetic and Pharmacodynamic Properties of Multiple Oral Doses of Sitagliptin, a Dipeptidyl Peptidase-IV Inhibitor: A Double-Blind, Randomized, Placebo-Controlled Study in Healthy Male Volunteers. Clin Ther [Internet]. 2006 [cited 2015 apr 09];28(1):55-72. doi: http://doi.org/fccw5w.
17. Garg K, Tripathi CD, Kumar S. Clinical Review of Sitagliptin: A DPP-4 Inhibitor. J Assoc Physicians India [Internet]. 2013 [cited 2015 apr 09];61(9):645-9. doi: http://goo.gl/vLV1WE.
18. Kidney Disease: Improving Global Outcomes.(KDIGO). KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int Suppl [Internet]. 2013 [cited 2015 apr 09];3(1):1-150. Available from: http://goo.gl/O8HB48.
19. Keller F, Hartmann B, Czock D. Time of effect duration and administration interval for sitagliptin in patients with kidney failure. Eur J Drug Metab Pharmacokinet [Internet]. 2014 [cited 2015 apr 09];39(2):77-85. doi: http://doi.org/3jr.
20. Bohórquez HE, Beltrán-Gálvis OA. Modelos de pronóstico de enfermedad hepática Child & Meld. Rev Col Gastroenterol [Internet]. 2004 [cited 2015 apr 09];19(2):109-14. Available from: http://goo.gl/x97GUe.
21. Maeda H, Kubota A, Tanaka Y, Terauchi Y, Matsuba I, Group A-KS. The safety, efficacy and predictors for HbA1c reduction of sitagliptin in the treatment of Japanese type 2 diabetes. Diabetes Res Clin Pract [Internet]. 2012 [cited 2015 apr 10];95(1):e20-2. http://doi.org/dxzsg2.
22. Aschner P, Kipnes MS, Lunceford JK, Sanchez M, Mickel C, Williams-Herman DE, et al. Effect of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy on glycemic control in patients with type 2 diabetes. Diabetes Care [Internet]. 2006 [cited 2015 apr 10];29(12):2632-7. doi: http://doi.org/c24p3d.
23. Du Q, Wu B, Wang Y-J, Yang S, Zhao Y-Y, Liang YY. Comparative effects of sitagliptin and metformin in patients with type 2 diabetes mellitus: a meta-analysis. Curr Med Res Opin [Internet]. 2013 [cited 2015 apr 10];29(11):1487-94. doi: http://doi.org/3jk.
24. Takai M, Ishikawa M, Maeda H, Kanamori A, Kubota A, Amemiya H, et al. Safety and efficacy of adding sitagliptin to insulin in patients with type 2 diabetes: the ASSIST-K study. Diabetes Res Clin Pract [Internet]. 2014 [cited 2015 apr 10];103(3):e30-3. doi: http://doi.org/f2rcrm.
25. Ahrén B. Clinical results of treating type 2 diabetic patients with sitagliptin, vildagliptin or saxagliptin-diabetes control and potential adverse events. Best Pract Res Clin Endocrinol Metab [Internet]. 2009 [cited 2015 apr 10];23(4):487-98. doi: http://doi.org/c97kng.
26. Guerci B, Monnier L, Serusclat P, Petit C, Valensi P, Huet D, et al. Continuous glucose profiles with vildagliptin versus sitagliptin in add-on to metformin: results from the randomized Optima study. Diabetes Metab [Internet]. 2012 [cited 2015 apr 10];38(4):359-66. doi: http://doi.org/3jm.
27. Karagiannis T, Boura P, Tsapas A. Safety of dipeptidyl peptidase 4 inhibitors: a perspective review. Ther Adv drug Saf [Internet]. 2014 [cited 2015 apr 10];5(3):138-46. doi: http://doi.org/3jn.
28. Eurich DT, Simpson S, Senthilselvan A, Asche CV, Sandhu-Minhas JK, McAlister FA. Comparative safety and effectiveness of sitagliptin in patients with type 2 diabetes: retrospective population based cohort study. BMJ [Internet]. 2013 [cited 2015 apr 10];346:f2267. doi: http://doi.org/3jp.
29. Zhan M, Xu T, Wu F, Tang Y. Sitagliptin in the treatment of type 2 diabetes: a meta-analysis. J Evid Based Med [Internet]. 2012 [cited 2015 apr 10];5(3):154-65. doi: http://doi.org/3jq.
30. Weir DL, McAlister FA, Senthilselvan A, Minhas-Sandhu JK, Eurich DT. Sitagliptin use in patients with diabetes and heart failure: a population-based retrospective cohort study. JACC Heart Fail [Internet]. 2014 [cited 2015 apr 10];177(6):86-90. doi: http://doi.org/3js.
31. Wang KL, Liu CJ, Chao TF, Huang CM, Wu CH, Chen SJ, et al. Sitagliptin and the risk of hospitalization for heart failure: a population-based study. Int J Cardiol [Internet]. 2014 [cited 2015 apr 10];177(1):86-90. doi: http://doi.org/3jt.
32. Montilla S, Marchesini G, Sammarco A, Trotta MP, Siviero PD, Tomino C, et al. Drug utilization, safety, and effectiveness of exenatide, sitagliptin, and vildagliptin for type 2 diabetes in the real world: Data from the Italian AIFA Anti-diabetics Monitoring Registry. Nutr Metab Cardiovasc Dis [Internet]. 2014 [cited 2015 apr 10];24(12):1346-53. doi: http://doi.org/3jv.
33. Monami M, Dicembrini I, Mannucci E. Dipeptidyl peptidase-4 inhibitors and pancreatitis risk: a meta-analysis of randomized clinical trials. Diabetes Obes Metab [Internet]. 2014 [cited 2015 apr 10];16(1):48-56. doi: http://doi.org/3jw.
34. Sin C, Mahé E, Sigal ML. Drug reaction with eosinophilia and systemic symptoms (DRESS) in a patient taking sitagliptin. Diabetes Metab [Internet]. 2012 [cited 2015 apr 10];38(6):571-3. doi: http://doi.org/3jx.
35. Villhauer EB, Brinkman JA, Naderi GB, Burkey BF, Dunning BE, Prasad K, et al. 1-[[(3-hydroxy-1-adamantyl)amino]acetyl]-2-cyano-(S)-pyrrolidine: a potent, selective, and orally bioavailable dipeptidyl peptidase IV inhibitor with antihyperglycemic properties. J Med Chem [Internet]. 2003 [cited 2015 apr 10];46(13):2774-89. doi: http://doi.org/frnnpd.
36. Ceriello A, Sportiello L, Rafaniello C, Rossi F. DPP-4 inhibitors: pharmacological differences and their clinical implications. Expert Opin Drug Saf [Internet]. 2014 [cited 2015 apr 10];13(Suppl 1):S57-68. doi: http://doi.org/3jz.
37. He YL, Valencia J, Zhang Y, Schwartz SL, Ligueros-Saylan M, Foley J, et al. Hormonal and metabolic effects of morning or evening dosing of the dipeptidyl peptidase IV inhibitor vildagliptin in patients with type 2 diabetes. Br J Clin Pharmacol [Internet]. 2010 [cited 2015 apr 10];70(1):34-42. doi: http://doi.org/cgpzqn.
38. Forst T, Bramlage P. Vildagliptin, a DPP-4 inhibitor for the twice-daily treatment of type 2 diabetes mellitus with or without metformin. Expert Opin Pharmacother [Internet]. 2014 [cited 2015 apr 10];15(9):1299-313. doi: http://doi.org/3j2.
39. He H, Tran P, Yin H, Smith H, Batard Y, Wang L, et al. Absorption, metabolism, and excretion of [14C]vildagliptin, a novel dipeptidyl peptidase 4 inhibitor, in humans. Drug Metab Dispos [Internet]. 2009 [cited 2015 apr 10];37(3):536-44. doi: http://doi.org/dsxbns.
40. He YL. Clinical pharmacokinetics and pharmacodynamics of vildagliptin. Clin Pharmacokinet [Internet]. 2012 [cited 2015 apr 10];51(3):147-62. doi: http://doi.org/3j4.
41. He YL, Kulmatycki K, Zhang Y, Zhou W, Reynolds C, Ligueros-Saylan M, et al. Pharmacokinetics of vildagliptin in patients with varying degrees of renal impairment. Int J Clin Pharmacol Ther [Internet]. 2013 [cited 2015 apr 10];51(9):693-703. doi: http://doi.org/3j5.
42. Ito H, Mifune M, Matsuyama E, Furusho M, Omoto T, Shinozaki M, et al. Vildagliptin is Effective for Glycemic Control in Diabetic Patients Undergoing either Hemodialysis or Peritoneal Dialysis. Diabetes Ther [Internet]. 2013 [cited 2015 apr 10];4(2):321-9. doi: http://doi.org/3j7.
43. Ligueros-Saylan M, Foley JE, Schweizer A, Couturier A, Kothny W. An assessment of adverse effects of vildagliptin versus comparators on the liver, the pancreas, the immune system, the skin and in patients with impaired renal function from a large pooled database of Phase II and III clinical trials. Diabetes Obes Metab [Internet]. 2010 [cited 2015 apr 10];12(6):495-509. doi: http://doi.org/frkxsj.
44. Lukashevich V, Schweizer A, Shao Q, Groop P-H, Kothny W. Safety and efficacy of vildagliptin versus placebo in patients with type 2 diabetes and moderate or severe renal impairment: a prospective 24-week randomized placebo-controlled trial. Diabetes Obes Metab [Internet]. 2011 [cited 2015 apr 10];13(10):947-54. doi: http://doi.org/cp5c42.
45. Kothny W, Shao Q, Groop PH, Lukashevich V. One-year safety, tolerability and efficacy of vildagliptin in patients with type 2 diabetes and moderate or severe renal impairment. Diabetes Obes Metab [Internet]. 2012 [cited 2015 apr 10];14(11):1032-9. doi: http://doi.org/3j8.
46. Scheen AJ. Pharmacokinetics in patients with chronic liver disease and hepatic safety of incretin-based therapies for the management of type 2 diabetes mellitus. Clin Pharmacokinet [Internet]. 2014 [cited 2015 apr 10];53(9):773-85. doi: http://doi.org/3j9.
47. Schweizer A, Couturier A, Foley JE, Dejager S. Comparison between vildagliptin and metformin to sustain reductions in HbA(1c) over 1 year in drug-naïve patients with Type 2 diabetes. Diabet Med [Internet]. 2007 [cited 2015 apr 10];24(9):955-61. doi: http://doi.org/fhfc85.
48. Dejager S, Schweizer A, Foley JE. Evidence to support the use of vildagliptin monotherapy in the treatment of type 2 diabetes mellitus. Vasc Health Risk Manag [Internet]. 2012 [cited 2015 apr 10];8(1):339-48. doi: http://doi.org/3kb.
49. Keating GM. Vildagliptin: a review of its use in type 2 diabetes mellitus. Drugs [Internet]. 2014 [cited 2015 apr 10];74(5):587-610. doi: http://doi.org/3kc.
50. Mathieu C, Barnett AH, Brath H, Conget I, de Castro JJ, Göke R, et al. Effectiveness and tolerability of second-line therapy with vildagliptin vs. other oral agents in type 2 diabetes: a real-life worldwide observational study (EDGE). Int J Clin Pract [Internet]. 2013 [cited 2015 apr 10];67(10):947-56. doi: http://doi.org/f23ghf.
51. Traynor K. FDA approves saxagliptin for type 2 diabetes. Am J Heal Syst Pharm [Internet]. 2009 [cited 2015 apr 10];66(17):1513. doi: http://doi.org/bm8jfh.
52. Augeri DJ, Robl JA, Betebenner DA, Magnin DR, Khanna A, Robertson JG, et al. Discovery and preclinical profile of Saxagliptin (BMS-477118): a highly potent, long-acting, orally active dipeptidyl peptidase IV inhibitor for the treatment of type 2 diabetes. J Med Chem [Internet]. 2005 [cited 2015 apr 10];48(15):5025-37. doi: http://doi.org/dqnrv5.
53. Ali S, Fonseca V. Saxagliptin overview: special focus on safety and adverse effects. Expert Opin Drug Saf [Internet]. 2013 [cited 2015 apr 10];12(1):103-9. doi: http://doi.org/3kd.
54. Kania DS, Gonzalvo JD, Weber ZA. Saxagliptin: a clinical review in the treatment of type 2 diabetes mellitus. Clin Ther [Internet]. 2011 [cited 2015 apr 10];33(8):1005-22. doi: http://doi.org/fwjv4z.
55. Boulton DW, Li L, Frevert EU, Tang A, Castaneda L, Vachharajani NN, et al. Influence of renal or hepatic impairment on the pharmacokinetics of saxagliptin. Clin Pharmacokinet [Internet]. 2011 [cited 2015 apr 10];50(4):253-65. doi: http://doi.org/bgw67w.
56. Davidson JA. Tolerability of saxagliptin in patients with inadequately controlled type 2 diabetes: results from 6 phase III studies. J Manag Care Pharm [Internet]. 2014 [cited 2015 apr 13];20(2):120-9. Available from: http://goo.gl/DvLHGK.
57. Raz I, Bhatt DL, Hirshberg B, Mosenzon O, Scirica BM, Umez-Eronini A, et al. Incidence of pancreatitis and pancreatic cancer in a randomized controlled multicenter trial (SAVOR-TIMI 53) of the dipeptidyl peptidase-4 inhibitor saxagliptin. Diabetes Care [Internet]. 2014 [cited 2015 apr 13];37(9):2435-41. doi: http://doi.org/3mp.
58. Scirica BM, Braunwald E, Raz I, Cavender MA, Morrow DA, Jarolim P, et al. Heart Failure, Saxagliptin and Diabetes Mellitus: Observations from the SAVOR-TIMI 53 Randomized Trial [cited 2015 apr 13]. Circulation. 2014 ;130(18):1579-88. doi: http://doi.org/3mq.
59. Elgart JF, Gonzalez L, Caporale JE, Valencia JE, Gagliardino JJ. [Economic evaluation of type 2 diabetes treatment with saxagliptin in Colombia]. Medwave [Internet]. 2012 [cited 2015 apr 13];12(02):e5306-e5306. Spanish. doi: http://doi.org/fzrjcg.
60. Eckhardt M, Langkopf E, Mark M, Tadayyon M, Thomas L, Nar H, et al. 8-(3-(R)-aminopiperidin-1-yl)-7-but-2-ynyl-3-methyl-1-(4-methyl-quinazolin-2-ylmethyl)-3,7-dihydropurine-2,6-dione (BI 1356), a highly potent, selective, long-acting, and orally bioavailable DPP-4 inhibitor for the treatment of type 2 diabetes. J Med Chem [Internet]. 2007 [cited 2015 apr 13];50(26):6450-3. doi: http://doi.org/cp2dtg.
61. McKeage K. Linagliptin: an update of its use in patients with type 2 diabetes mellitus. Drugs [Internet]. 2014 [cited 2015 apr 13];74(16):1927-46. doi: http://doi.org/3mr.
62. McGill JB. Linagliptin for type 2 diabetes mellitus: a review of the pivotal clinical trials. Ther Adv Endocrinol Metab [Internet]. 2012 [cited 2015 apr 13];3(4):113-24. doi: http://doi.org/3ms.
63. Barnett AH. Linagliptin for the treatment of type 2 diabetes mellitus: a drug safety evaluation. Expert Opin Drug Saf [Internet]. 2015 [cited 2015 apr 13];14(1):149-59. doi: http://doi.org/3mt.
64. Clinical Guidelines Task Force. Global guideline for type 2 diabetes. Diabetes Res Clin Pract [Internet]. 2014 [cited 2015 apr 13];104(1):1-52. doi: http://doi.org/f2rbnm.
65. Ampudia-Blasco FJ, Ceriello A. [Importance of daily glycemic variability in achieving glycemic targets in type 2 diabetes: role of DPP-4 inhibitors]. Med Clin (Barc) [Internet]. 2010 [cited 2015 apr 13];135 Suppl(Supl 1):33-9. Spanish. doi: http://doi.org/fprk2b.
66. Marfella R, Barbieri M, Grella R, Rizzo MR, Nicoletti GF, Paolisso G. Effects of vildagliptin twice daily vs. Sitagliptin once daily on 24-hour acute glucose fluctuations. J Diabetes Complications [Internet]. 2010 [cited 2015 apr 13];24(2):79-83. doi: http://doi.org/cnpsvb.
67. Flynn C, Bakris GL. Noninsulin glucose-lowering agents for the treatment of patients on dialysis. Nat Rev Nephrol [Internet]. 2013 [cited 2015 apr 13];9(3):147-53. doi: http://doi.org/3mv.
68. European Medicines Agency. Efficacy, Safety & Modification of Albuminuria in type 2 diabetes subjects with Renal disease with LINAgliptin. EU Clinical Trials Register [Internet]. 2012 [cited 2015 mar 29]. Available from: http://goo.gl/0oskqw.
69. Itou M, Kawaguchi T, Taniguchi E, Sata M. Dipeptidyl peptidase-4: a key player in chronic liver disease. World J Gastroenterol [Internet]. 2013 [cited 2015 apr 13];19(15):2298-306. doi: http://doi.org/3mw.
70. Arase Y, Suzuki F, Kobayashi M, Suzuki Y, Kawamura Y, Matsumoto N, et al. Efficacy and safety in sitagliptin therapy for diabetes complicated by chronic liver disease caused by hepatitis C virus. Hepatol Res [Internet]. 2011 [cited 2015 apr 13];41(6):524-9. doi: http://doi.org/b6ftqv.
71. Yanai H. Dipeptidyl peptidase-4 inhibitor sitagliptin significantly reduced hepatitis C virus replication in a diabetic patient with chronic hepatitis C virus infection. Hepatobiliary Pancreat Dis Int [Internet]. 2014 [cited 2015 apr 13];13(5):556. doi: http://doi.org/3m2.
72. Scheen AJ. Pharmacokinetic and toxicological considerations for the treatment of diabetes in patients with liver disease. Expert Opin Drug Metab Toxicol [Internet]. 2014 [cited 2015 apr 13];10(6):839-57. doi: http://doi.org/3m3.
73. Salles TA, dos Santos L, Barauna VG, Girardi AC. Potential Role of Dipeptidyl Peptidase IV in the Pathophysiology of Heart Failure. Int J Mol Sci [Internet]. 2015 [cited 2015 apr 13];16(2):4226-49. doi: http://doi.org/3m4.
74. Monami M, Dicembrini I, Mannucci E. Dipeptidyl peptidase-4 inhibitors and heart failure: a meta-analysis of randomized clinical trials. Nutr Metab Cardiovasc Dis [Internet]. 2014 [cited 2015 apr 13];24(7):689-97. doi: http://doi.org/3m5.
75. Savarese G, Perrone-Filardi P, D'Amore C, Vitale C, Trimarco B, Pani L, et al. Cardiovascular effects of dipeptidyl peptidase-4 inhibitors in diabetic patients: A meta-analysis. Int J Cardiol [Internet]. 2014 [cited 2015 apr 13];181:239-44. doi: http://doi.org/3m6.
76. Yu OH, Filion KB, Azoulay L, Patenaude V, Majdan A, Suissa S. Incretin-Based Drugs and the Risk of Congestive Heart Failure. Diabetes Care [Internet]. 2015 [cited 2015 apr 13];38(2):277-84. doi: http://doi.org/3m7.
77. Bloomgarden ZT, Dodis R, Viscoli CM, Holmboe ES, Inzucchi SE. Lower baseline glycemia reduces apparent oral agent glucose-lowering efficacy: A meta-regression analysis. Diabetes Care [Internet]. 2006 [cited 2015 apr 13];29(9):2137-9. doi: http://doi.org/bh82wv.
Referencias
Aschner P. [Epidemiology of diabetes in Colombia]. Av en Diabetol [Internet]. 2010 [cited 2015 apr 09];26:95-100. Spanish. doi: http://doi.org/f2jmjd.
Aschner P, Aguilar-Salinas C, Aguirre L, Franco L, Gagliardino JJ, de Lapertosa SG, et al. Diabetes in South and Central America: an update. Diabetes Res Clin Pract [Internet]. 2014 [cited 2015 apr 09];103(2):238-43. doi: http://doi.org/f2qh6q.
Ministerio de Salud y Protección Social. Resolución 005521 de 2013. Por la cual se define, aclara y actualiza integralmente el Plan Obligatorio de Salud (POS). Colombia [Internet]; 2013 [cited 2015 apr 09]. p. 1-146. Available from: http://goo.gl/DJQjfZ.
Neumiller JJ. Incretin-Based Therapies. Med Clin North Am [Internet]. 2015 [cited 2015 apr 09];99(1):107-29. doi: http://doi.org/2qp.
American Diabetes Association. Approaches to Glycemic Treatment. (Sec. 7). In Standards of Medical Care in Diabetes-2015. Diabetes Care [Internet]. 2015 [cited 2015 apr 09];38(Suppl 1):S41-8. doi: http://doi.org/3hr.
Scheen AJ. A review of gliptins in 2011. Expert Opin Pharmacotherapy [Internet]. 2012 [cited 2015 apr 09];13(1):81-99. doi: http://doi.org/c3fxf4.
Baggio LL, Drucker DJ. Biology of incretins: GLP-1 and GIP. Gastroenterology [Internet]. 2007 [cited 2015 apr 09];132(6):2131-57. doi: http://doi.org/c8rk2h.
Elrick H, Stimmler L, Hlad CJ, Arai Y. Plasma insulin response to oral and intravenous glucose administration. J Clin Endocrinol Metab [Internet]. 1964 [cited 2015 apr 09];24(10):1076-82. doi: http://doi.org/fvjwqw.
Campbell JE, Drucker DJ. Pharmacology, physiology, and mechanisms of incretin hormone action. Cell Metab [Internet]. 2013 [cited 2015 apr 09];17(6):819-37. doi: http://doi.org/3hs.
White JR. Dipeptidyl peptidase-IV inhibitors: Pharmacological profile and clinical use. Clinical Diabetes [Internet]. 2008 [cited 2015 apr 09];26(2):53-7. doi: http://doi.org/d74xkx.
Ussher JR, Drucker DJ. Cardiovascular biology of the incretin system. Endocr Rev [Internet]. 2012 [cited 2015 apr 09];33(2):187-215. doi: http://doi.org/2p3.
Brown DX, Evans M. Choosing between GLP-1 receptor agonists and DPP-4 inhibitors: A pharmacological perspective. J Nutr Metab [Internet]. 2012 [cited 2015 apr 09]. p. 1-10. doi: http://doi.org/3ht.
Mulvihill EE, Drucker DJ. Pharmacology, physiology, and mechanisms of action of dipeptidyl peptidase-4 inhibitors. Endocr Rev [Internet]. 2014 [cited 2015 apr 09];35(6):992-1019. doi: http://doi.org/3hw.
Zhao Y, Yang L, Zhou Z. Dipeptidyl peptidase-4 inhibitors: Multitarget drugs, not only antidiabetes drugs. J Diabetes [Internet]. 2014 [cited 2015 apr 09];6(1):21-9. doi: http://doi.org/3hx.
López-Jaramillo P, Velandia C, Castillo G, Sánchez T, Álvarez J. [Incretin analogs and inhibitors of DPP-4: which is their role in the primary prevention of cardiovascular diseases?] Rev Colomb Cardiol [Internet]. 2013 [cited 2015 apr 09];20(5):287-99. Spanish. Available from: http://goo.gl/0mCNym.
Bergman AJ, Stevens C, Zhou Y, Yi B, Laethem M, de Smet M, et al. Pharmacokinetic and Pharmacodynamic Properties of Multiple Oral Doses of Sitagliptin, a Dipeptidyl Peptidase-IV Inhibitor: A Double-Blind, Randomized, Placebo-Controlled Study in Healthy Male Volunteers. Clin Ther [Internet]. 2006 [cited 2015 apr 09];28(1):55-72. doi: http://doi.org/fccw5w.
Garg K, Tripathi CD, Kumar S. Clinical Review of Sitagliptin: A DPP-4 Inhibitor. J Assoc Physicians India [Internet]. 2013 [cited 2015 apr 09];61(9):645-9. doi: http://goo.gl/vLV1WE.
Kidney Disease: Improving Global Outcomes.(KDIGO). KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney Int Suppl [Internet]. 2013 [cited 2015 apr 09];3(1):1-150. Available from: http://goo.gl/O8HB48.
Keller F, Hartmann B, Czock D. Time of effect duration and administration interval for sitagliptin in patients with kidney failure. Eur J Drug Metab Pharmacokinet [Internet]. 2014 [cited 2015 apr 09];39(2):77-85. doi: http://doi.org/3jr.
Bohórquez HE, Beltrán-Gálvis OA. Modelos de pronóstico de enfermedad hepática Child & Meld. Rev Col Gastroenterol [Internet]. 2004 [cited 2015 apr 09];19(2):109-14. Available from: http://goo.gl/x97GUe.
Maeda H, Kubota A, Tanaka Y, Terauchi Y, Matsuba I, Group A-KS. The safety, efficacy and predictors for HbA1c reduction of sitagliptin in the treatment of Japanese type 2 diabetes. Diabetes Res Clin Pract [Internet]. 2012 [cited 2015 apr 10];95(1):e20-2. http://doi.org/dxzsg2.
Aschner P, Kipnes MS, Lunceford JK, Sanchez M, Mickel C, Williams-Herman DE, et al. Effect of the dipeptidyl peptidase-4 inhibitor sitagliptin as monotherapy on glycemic control in patients with type 2 diabetes. Diabetes Care [Internet]. 2006 [cited 2015 apr 10];29(12):2632-7. doi: http://doi.org/c24p3d.
Du Q, Wu B, Wang Y-J, Yang S, Zhao Y-Y, Liang YY. Comparative effects of sitagliptin and metformin in patients with type 2 diabetes mellitus: a meta-analysis. Curr Med Res Opin [Internet]. 2013 [cited 2015 apr 10];29(11):1487-94. doi: http://doi.org/3jk.
Takai M, Ishikawa M, Maeda H, Kanamori A, Kubota A, Amemiya H, et al. Safety and efficacy of adding sitagliptin to insulin in patients with type 2 diabetes: the ASSIST-K study. Diabetes Res Clin Pract [Internet]. 2014 [cited 2015 apr 10];103(3):e30-3. doi: http://doi.org/f2rcrm.
Ahrén B. Clinical results of treating type 2 diabetic patients with sitagliptin, vildagliptin or saxagliptin-diabetes control and potential adverse events. Best Pract Res Clin Endocrinol Metab [Internet]. 2009 [cited 2015 apr 10];23(4):487-98. doi: http://doi.org/c97kng.
Guerci B, Monnier L, Serusclat P, Petit C, Valensi P, Huet D, et al. Continuous glucose profiles with vildagliptin versus sitagliptin in add-on to metformin: results from the randomized Optima study. Diabetes Metab [Internet]. 2012 [cited 2015 apr 10];38(4):359-66. doi: http://doi.org/3jm.
Karagiannis T, Boura P, Tsapas A. Safety of dipeptidyl peptidase 4 inhibitors: a perspective review. Ther Adv drug Saf [Internet]. 2014 [cited 2015 apr 10];5(3):138-46. doi: http://doi.org/3jn.
Eurich DT, Simpson S, Senthilselvan A, Asche CV, Sandhu-Minhas JK, McAlister FA. Comparative safety and effectiveness of sitagliptin in patients with type 2 diabetes: retrospective population based cohort study. BMJ [Internet]. 2013 [cited 2015 apr 10];346:f2267. doi: http://doi.org/3jp.
Zhan M, Xu T, Wu F, Tang Y. Sitagliptin in the treatment of type 2 diabetes: a meta-analysis. J Evid Based Med [Internet]. 2012 [cited 2015 apr 10];5(3):154-65. doi: http://doi.org/3jq.
Weir DL, McAlister FA, Senthilselvan A, Minhas-Sandhu JK, Eurich DT. Sitagliptin use in patients with diabetes and heart failure: a population-based retrospective cohort study. JACC Heart Fail [Internet]. 2014 [cited 2015 apr 10];177(6):86-90. doi: http://doi.org/3js.
Wang KL, Liu CJ, Chao TF, Huang CM, Wu CH, Chen SJ, et al. Sitagliptin and the risk of hospitalization for heart failure: a population-based study. Int J Cardiol [Internet]. 2014 [cited 2015 apr 10];177(1):86-90. doi: http://doi.org/3jt.
Montilla S, Marchesini G, Sammarco A, Trotta MP, Siviero PD, Tomino C, et al. Drug utilization, safety, and effectiveness of exenatide, sitagliptin, and vildagliptin for type 2 diabetes in the real world: Data from the Italian AIFA Anti-diabetics Monitoring Registry. Nutr Metab Cardiovasc Dis [Internet]. 2014 [cited 2015 apr 10];24(12):1346-53. doi: http://doi.org/3jv.
Monami M, Dicembrini I, Mannucci E. Dipeptidyl peptidase-4 inhibitors and pancreatitis risk: a meta-analysis of randomized clinical trials. Diabetes Obes Metab [Internet]. 2014 [cited 2015 apr 10];16(1):48-56. doi: http://doi.org/3jw.
Sin C, Mahé E, Sigal ML. Drug reaction with eosinophilia and systemic symptoms (DRESS) in a patient taking sitagliptin. Diabetes Metab [Internet]. 2012 [cited 2015 apr 10];38(6):571-3. doi: http://doi.org/3jx.
Villhauer EB, Brinkman JA, Naderi GB, Burkey BF, Dunning BE, Prasad K, et al. 1-[[(3-hydroxy-1-adamantyl)amino]acetyl]-2-cyano-(S)-pyrrolidine: a potent, selective, and orally bioavailable dipeptidyl peptidase IV inhibitor with antihyperglycemic properties. J Med Chem [Internet]. 2003 [cited 2015 apr 10];46(13):2774-89. doi: http://doi.org/frnnpd.
Ceriello A, Sportiello L, Rafaniello C, Rossi F. DPP-4 inhibitors: pharmacological differences and their clinical implications. Expert Opin Drug Saf [Internet]. 2014 [cited 2015 apr 10];13(Suppl 1):S57-68. doi: http://doi.org/3jz.
He YL, Valencia J, Zhang Y, Schwartz SL, Ligueros-Saylan M, Foley J, et al. Hormonal and metabolic effects of morning or evening dosing of the dipeptidyl peptidase IV inhibitor vildagliptin in patients with type 2 diabetes. Br J Clin Pharmacol [Internet]. 2010 [cited 2015 apr 10];70(1):34-42. doi: http://doi.org/cgpzqn.
Forst T, Bramlage P. Vildagliptin, a DPP-4 inhibitor for the twice-daily treatment of type 2 diabetes mellitus with or without metformin. Expert Opin Pharmacother [Internet]. 2014 [cited 2015 apr 10];15(9):1299-313. doi: http://doi.org/3j2.
He H, Tran P, Yin H, Smith H, Batard Y, Wang L, et al. Absorption, metabolism, and excretion of [14C]vildagliptin, a novel dipeptidyl peptidase 4 inhibitor, in humans. Drug Metab Dispos [Internet]. 2009 [cited 2015 apr 10];37(3):536-44. doi: http://doi.org/dsxbns.
He YL. Clinical pharmacokinetics and pharmacodynamics of vildagliptin. Clin Pharmacokinet [Internet]. 2012 [cited 2015 apr 10];51(3):147-62. doi: http://doi.org/3j4.
He YL, Kulmatycki K, Zhang Y, Zhou W, Reynolds C, Ligueros-Saylan M, et al. Pharmacokinetics of vildagliptin in patients with varying degrees of renal impairment. Int J Clin Pharmacol Ther [Internet]. 2013 [cited 2015 apr 10];51(9):693-703. doi: http://doi.org/3j5.
Ito H, Mifune M, Matsuyama E, Furusho M, Omoto T, Shinozaki M, et al. Vildagliptin is Effective for Glycemic Control in Diabetic Patients Undergoing either Hemodialysis or Peritoneal Dialysis. Diabetes Ther [Internet]. 2013 [cited 2015 apr 10];4(2):321-9. doi: http://doi.org/3j7.
Ligueros-Saylan M, Foley JE, Schweizer A, Couturier A, Kothny W. An assessment of adverse effects of vildagliptin versus comparators on the liver, the pancreas, the immune system, the skin and in patients with impaired renal function from a large pooled database of Phase II and III clinical trials. Diabetes Obes Metab [Internet]. 2010 [cited 2015 apr 10];12(6):495-509. doi: http://doi.org/frkxsj.
Lukashevich V, Schweizer A, Shao Q, Groop P-H, Kothny W. Safety and efficacy of vildagliptin versus placebo in patients with type 2 diabetes and moderate or severe renal impairment: a prospective 24-week randomized placebo-controlled trial. Diabetes Obes Metab [Internet]. 2011 [cited 2015 apr 10];13(10):947-54. doi: http://doi.org/cp5c42.
Kothny W, Shao Q, Groop PH, Lukashevich V. One-year safety, tolerability and efficacy of vildagliptin in patients with type 2 diabetes and moderate or severe renal impairment. Diabetes Obes Metab [Internet]. 2012 [cited 2015 apr 10];14(11):1032-9. doi: http://doi.org/3j8.
Scheen AJ. Pharmacokinetics in patients with chronic liver disease and hepatic safety of incretin-based therapies for the management of type 2 diabetes mellitus. Clin Pharmacokinet [Internet]. 2014 [cited 2015 apr 10];53(9):773-85. doi: http://doi.org/3j9.
Schweizer A, Couturier A, Foley JE, Dejager S. Comparison between vildagliptin and metformin to sustain reductions in HbA(1c) over 1 year in drug-naïve patients with Type 2 diabetes. Diabet Med [Internet]. 2007 [cited 2015 apr 10];24(9):955-61. doi: http://doi.org/fhfc85.
Dejager S, Schweizer A, Foley JE. Evidence to support the use of vildagliptin monotherapy in the treatment of type 2 diabetes mellitus. Vasc Health Risk Manag [Internet]. 2012 [cited 2015 apr 10];8(1):339-48. doi: http://doi.org/3kb.
Keating GM. Vildagliptin: a review of its use in type 2 diabetes mellitus. Drugs [Internet]. 2014 [cited 2015 apr 10];74(5):587-610. doi: http://doi.org/3kc.
Mathieu C, Barnett AH, Brath H, Conget I, de Castro JJ, Göke R, et al. Effectiveness and tolerability of second-line therapy with vildagliptin vs. other oral agents in type 2 diabetes: a real-life worldwide observational study (EDGE). Int J Clin Pract [Internet]. 2013 [cited 2015 apr 10];67(10):947-56. doi: http://doi.org/f23ghf.
Traynor K. FDA approves saxagliptin for type 2 diabetes. Am J Heal Syst Pharm [Internet]. 2009 [cited 2015 apr 10];66(17):1513. doi: http://doi.org/bm8jfh.
Augeri DJ, Robl JA, Betebenner DA, Magnin DR, Khanna A, Robertson JG, et al. Discovery and preclinical profile of Saxagliptin (BMS-477118): a highly potent, long-acting, orally active dipeptidyl peptidase IV inhibitor for the treatment of type 2 diabetes. J Med Chem [Internet]. 2005 [cited 2015 apr 10];48(15):5025-37. doi: http://doi.org/dqnrv5.
Ali S, Fonseca V. Saxagliptin overview: special focus on safety and adverse effects. Expert Opin Drug Saf [Internet]. 2013 [cited 2015 apr 10];12(1):103-9. doi: http://doi.org/3kd.
Kania DS, Gonzalvo JD, Weber ZA. Saxagliptin: a clinical review in the treatment of type 2 diabetes mellitus. Clin Ther [Internet]. 2011 [cited 2015 apr 10];33(8):1005-22. doi: http://doi.org/fwjv4z.
Boulton DW, Li L, Frevert EU, Tang A, Castaneda L, Vachharajani NN, et al. Influence of renal or hepatic impairment on the pharmacokinetics of saxagliptin. Clin Pharmacokinet [Internet]. 2011 [cited 2015 apr 10];50(4):253-65. doi: http://doi.org/bgw67w.
Davidson JA. Tolerability of saxagliptin in patients with inadequately controlled type 2 diabetes: results from 6 phase III studies. J Manag Care Pharm [Internet]. 2014 [cited 2015 apr 13];20(2):120-9. Available from: http://goo.gl/DvLHGK.
Raz I, Bhatt DL, Hirshberg B, Mosenzon O, Scirica BM, Umez-Eronini A, et al. Incidence of pancreatitis and pancreatic cancer in a randomized controlled multicenter trial (SAVOR-TIMI 53) of the dipeptidyl peptidase-4 inhibitor saxagliptin. Diabetes Care [Internet]. 2014 [cited 2015 apr 13];37(9):2435-41. doi: http://doi.org/3mp.
Scirica BM, Braunwald E, Raz I, Cavender MA, Morrow DA, Jarolim P, et al. Heart Failure, Saxagliptin and Diabetes Mellitus: Observations from the SAVOR-TIMI 53 Randomized Trial [cited 2015 apr 13]. Circulation. 2014 ;130(18):1579-88. doi: http://doi.org/3mq.
Elgart JF, Gonzalez L, Caporale JE, Valencia JE, Gagliardino JJ. [Economic evaluation of type 2 diabetes treatment with saxagliptin in Colombia]. Medwave [Internet]. 2012 [cited 2015 apr 13];12(02):e5306-e5306. Spanish. doi: http://doi.org/fzrjcg.
Eckhardt M, Langkopf E, Mark M, Tadayyon M, Thomas L, Nar H, et al. 8-(3-(R)-aminopiperidin-1-yl)-7-but-2-ynyl-3-methyl-1-(4-methyl-quinazolin-2-ylmethyl)-3,7-dihydropurine-2,6-dione (BI 1356), a highly potent, selective, long-acting, and orally bioavailable DPP-4 inhibitor for the treatment of type 2 diabetes. J Med Chem [Internet]. 2007 [cited 2015 apr 13];50(26):6450-3. doi: http://doi.org/cp2dtg.
McKeage K. Linagliptin: an update of its use in patients with type 2 diabetes mellitus. Drugs [Internet]. 2014 [cited 2015 apr 13];74(16):1927-46. doi: http://doi.org/3mr.
McGill JB. Linagliptin for type 2 diabetes mellitus: a review of the pivotal clinical trials. Ther Adv Endocrinol Metab [Internet]. 2012 [cited 2015 apr 13];3(4):113-24. doi: http://doi.org/3ms.
Barnett AH. Linagliptin for the treatment of type 2 diabetes mellitus: a drug safety evaluation. Expert Opin Drug Saf [Internet]. 2015 [cited 2015 apr 13];14(1):149-59. doi: http://doi.org/3mt.
Clinical Guidelines Task Force. Global guideline for type 2 diabetes. Diabetes Res Clin Pract [Internet]. 2014 [cited 2015 apr 13];104(1):1-52. doi: http://doi.org/f2rbnm.
Ampudia-Blasco FJ, Ceriello A. [Importance of daily glycemic variability in achieving glycemic targets in type 2 diabetes: role of DPP-4 inhibitors]. Med Clin (Barc) [Internet]. 2010 [cited 2015 apr 13];135 Suppl(Supl 1):33-9. Spanish. doi: http://doi.org/fprk2b.
Marfella R, Barbieri M, Grella R, Rizzo MR, Nicoletti GF, Paolisso G. Effects of vildagliptin twice daily vs. Sitagliptin once daily on 24-hour acute glucose fluctuations. J Diabetes Complications [Internet]. 2010 [cited 2015 apr 13];24(2):79-83. doi: http://doi.org/cnpsvb.
Flynn C, Bakris GL. Noninsulin glucose-lowering agents for the treatment of patients on dialysis. Nat Rev Nephrol [Internet]. 2013 [cited 2015 apr 13];9(3):147-53. doi: http://doi.org/3mv.
European Medicines Agency. Efficacy, Safety & Modification of Albuminuria in type 2 diabetes subjects with Renal disease with LINAgliptin. EU Clinical Trials Register [Internet]. 2012 [cited 2015 mar 29]. Available from: http://goo.gl/0oskqw.
Itou M, Kawaguchi T, Taniguchi E, Sata M. Dipeptidyl peptidase-4: a key player in chronic liver disease. World J Gastroenterol [Internet]. 2013 [cited 2015 apr 13];19(15):2298-306. doi: http://doi.org/3mw.
Arase Y, Suzuki F, Kobayashi M, Suzuki Y, Kawamura Y, Matsumoto N, et al. Efficacy and safety in sitagliptin therapy for diabetes complicated by chronic liver disease caused by hepatitis C virus. Hepatol Res [Internet]. 2011 [cited 2015 apr 13];41(6):524-9. doi: http://doi.org/b6ftqv.
Yanai H. Dipeptidyl peptidase-4 inhibitor sitagliptin significantly reduced hepatitis C virus replication in a diabetic patient with chronic hepatitis C virus infection. Hepatobiliary Pancreat Dis Int [Internet]. 2014 [cited 2015 apr 13];13(5):556. doi: http://doi.org/3m2.
Scheen AJ. Pharmacokinetic and toxicological considerations for the treatment of diabetes in patients with liver disease. Expert Opin Drug Metab Toxicol [Internet]. 2014 [cited 2015 apr 13];10(6):839-57. doi: http://doi.org/3m3.
Salles TA, dos Santos L, Barauna VG, Girardi AC. Potential Role of Dipeptidyl Peptidase IV in the Pathophysiology of Heart Failure. Int J Mol Sci [Internet]. 2015 [cited 2015 apr 13];16(2):4226-49. doi: http://doi.org/3m4.
Monami M, Dicembrini I, Mannucci E. Dipeptidyl peptidase-4 inhibitors and heart failure: a meta-analysis of randomized clinical trials. Nutr Metab Cardiovasc Dis [Internet]. 2014 [cited 2015 apr 13];24(7):689-97. doi: http://doi.org/3m5.
Savarese G, Perrone-Filardi P, D'Amore C, Vitale C, Trimarco B, Pani L, et al. Cardiovascular effects of dipeptidyl peptidase-4 inhibitors in diabetic patients: A meta-analysis. Int J Cardiol [Internet]. 2014 [cited 2015 apr 13];181:239-44. doi: http://doi.org/3m6.
Yu OH, Filion KB, Azoulay L, Patenaude V, Majdan A, Suissa S. Incretin-Based Drugs and the Risk of Congestive Heart Failure. Diabetes Care [Internet]. 2015 [cited 2015 apr 13];38(2):277-84. doi: http://doi.org/3m7.
Bloomgarden ZT, Dodis R, Viscoli CM, Holmboe ES, Inzucchi SE. Lower baseline glycemia reduces apparent oral agent glucose-lowering efficacy: A meta-regression analysis. Diabetes Care [Internet]. 2006 [cited 2015 apr 13];29(9):2137-9. doi: http://doi.org/bh82wv.
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. Santiago Saldarriaga Betancur. (2018). Enfoque terapéutico de la diabetes mellitus tipo 2 en adultos. Más allá de una meta glucémica. Medicina UPB, 37(1), p.36. https://doi.org/10.18566/medupb.v37n1.a05.
2. Análida Elizabeth Pinilla-Roa, María Del Pilar Barrera-Perdomo. (2018). Prevención en diabetes mellitus y riesgo cardiovascular: enfoque médico y nutricional. Revista de la Facultad de Medicina, 66(3), p.459. https://doi.org/10.15446/revfacmed.v66n3.60060.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2015 Revista de la Facultad de Medicina

Esta obra está bajo una licencia Creative Commons Reconocimiento 3.0 Unported.
-