Molecular mechanisms of autophagy and its role in cancer development
Mecanismos moleculares de la autofagia y su papel en el cáncer
DOI:
https://doi.org/10.15446/revfacmed.v64n3.54152Palabras clave:
Autophagy, Cancer, Tumorigenesis, Proliferation (en)Autofagia, Cáncer, Tumorigénesis, Proliferación (es)
Descargas
Autophagy is an evolutionary process preserved in eukaryotes, which removes harmful components and maintains cell homeostasis in response to a variety of extracellular stimuli. It is involved in both physiological and pathological conditions, including cancer.
The role of autophagy in the treatment of cancer is described as a “double-edged sword”, which reflects its involvement in tumor suppression, survival and subsequent proliferation of tumor cells. Recent advances are useful for planning appropriate adjustments to inhibit or promote autophagy in order to obtain therapeutic efficacy in cancer patients. The objectives of this review are to clarify the role of autophagy in cancer and to highlight the need for more research in the field.
La autofagia es un proceso conservado evolutivamente en eucariotas que elimina componentes dañinos y mantiene la homeostasis celular en respuesta a una serie de estímulos extracelulares. Está implicada tanto en condiciones fisiológicas como patológicas, incluyendo el cáncer.
El papel de la autofagia en el tratamiento del cáncer se describe como un “arma de doble filo”, un término que refleja su participación en la supresión tumoral, la supervivencia y la proliferación de células tumorales. Los avances recientes ayudan a proyectar los ajustes apropiados en la inhibición o la promoción de la autofagia con el objetivo de conferir eficacia terapéutica en los pacientes con cáncer. Esta revisión tiene como objetivo aclarar los roles de la autofagia en el cáncer y destacar la necesidad de una mayor investigación en el campo.review article
DOI: https://doi.org/10.15446/revfacmed.v64n3.54152
Molecular mechanisms of autophagy and its role
in cancer development
Mecanismos moleculares de la autofagia y su papel en el cáncer
Received: 12/11/2015. Accepted: 11/01/2016.
Kathleen Salazar-Ramírez1 • Jhonny Molinares-Rodríguez1 • Samir Bolívar-González1
1 Universidad del Atlántico - Faculty of Chemistry and Pharmaceutical Sciences - Pharmaceutical Care and Pharmacology Research Group - Barranquilla - Colombia.
Corresponding author: Samir José Bolívar-González. Laboratory of Molecular Pharmacology, Faculty of Chemical and Pharmaceutical Sciences, Universidad de Chile. Santos Dumont 964, piso 5. Phone number: +56 9 79937723. Santiago de Chile. Chile.
Email: samirbolivargonzalez@hotmail.com.
| Abstract |
Autophagy is an evolutionary process preserved in eukaryotes, which removes harmful components and maintains cell homeostasis in response to a variety of extracellular stimuli. It is involved in both physiological and pathological conditions, including cancer.
The role of autophagy in the treatment of cancer is described as a “double-edged sword”, which reflects its involvement in tumor suppression, survival and subsequent proliferation of tumor cells. Recent advances are useful for planning appropriate adjustments to inhibit or promote autophagy in order to obtain therapeutic efficacy in cancer patients. The objectives of this review are to clarify the role of autophagy in cancer and to highlight the need for more research in the field.
Keywords: Autophagy; Cancer; Tumorigenesis; Proliferation (MeSH).
Salazar-Ramírez K, Molinares-Rodríguez J, Bolívar-González S. Molecular mechanisms of autophagy and its role in cancer development. Rev. Fac. Med. 2016;64(3):529-35. English. doi:
https://doi.org/10.15446/revfacmed.v64n3.54152.
| Resumen |
La autofagia es un proceso conservado evolutivamente en eucariotas que elimina componentes dañinos y mantiene la homeostasis celular en respuesta a una serie de estímulos extracelulares. Está implicada tanto en condiciones fisiológicas como patológicas, incluyendo el cáncer.
El papel de la autofagia en el tratamiento del cáncer se describe como un “arma de doble filo”, un término que refleja su participación en la supresión tumoral, la supervivencia y la proliferación de células tumorales. Los avances recientes ayudan a proyectar los ajustes apropiados en la inhibición o la promoción de la autofagia con el objetivo de conferir eficacia terapéutica en los pacientes con cáncer. Esta revisión tiene como objetivo aclarar los roles de la autofagia en el cáncer y destacar la necesidad de una mayor investigación en el campo.
Palabras clave: Autofagia; Cáncer; Tumorigénesis; Proliferación (DeCS).
Salazar-Ramírez K, Molinares-Rodríguez J, Bolívar-González S. [Mecanismos moleculares de la autofagia y su papel en el cáncer]. Rev. Fac. Med. 2016;64(3):529-35. English. doi:
https://doi.org/10.15446/revfacmed.v64n3.54152.
Introduction
Tumorigenesis is a complex multistage process which involves tumor initiation, promotion, progression to malignancy and metastasis. Tumor cells are characterized mainly by the result of uncontrolled proliferation processes, where cell division occurs faster. In addition to proliferation, other affected molecular mechanisms are programmed cell death or apoptosis, and the cell cycle (1).
Autophagy plays an important role, not only in the different stages of tumorigenesis, but also in disease states that lead to a microenvironment that promotes tumorigenesis. The role of this process in pathological states associated with higher risk of cancer, such as chronic liver disease, obesity and inflammatory bowel disease, is increasingly clear (2-4).
Pharmacological management of autophagy, with the intention of preventing a favorable microenvironment for tumor initiation, may require an opposite approach to limit tumor progression once pre-malignant cells are established. In this review, the regulation of autophagy, types of autophagy, autophagy itself and its mechanism as tumor suppressor or inducer are addressed. Finally, autophagy as a therapy against cancer, mainly in tumor cells with competent autophagy and defective autophagy, and induction of cell death by autophagy as a therapeutic strategy are discussed.
Molecular regulation of autophagy
Autophagy is a mechanism essential for maintaining cellular homeostasis in the body in the absence of important nutrients that work as an energy source. This process begins with the retention of cytoplasmic components, such as protein aggregates and damaged or aged organelles, through double-membrane vesicles called autophagosomes. The retentate is then transferred to degradation organelles such as lysosomes or vacuoles for destruction and eventual recycling of resulting macromolecules (5-7).
Although the study of the components of autophagy in mammalian cells was first performed in 1950, currently, it has been proved that there are studies performed in this population, and many others conducted using microorganisms such as yeast, where the existence of about 31 ATG genes has been observed; these genes are closely related to autophagy and the place where they are located in is known as perivacuolar site (PAS). The ongoing study of the nature of autophagy is becoming more important; here, the animal model in yeast is a powerful medium to decipher multiple concerns (8,9).
The molecular regulation of autophagy occurs in two different ways: a) through the activation of mTORC1 (mammalian Target of Rapamycin), in response to starvation or exhaustion of energy, and b) through energy detection, regulated by AMP-activated protein kinase (AMPK) (10). Autophagy could also be regulated by the Beclin 1 protein complex, consisting of the Beclin 1 (homolog Atg6) protein, phosphatidyl-inositol 3-kinase (PI3K) class III (PI3KC3/Vps34), p150, and Atg14L or UV radiation resistance-associated gene protein (UVRAG) (11,12). The intrinsic activation of the PI3KC3/Beclin 1 complex leads to the generation of phosphatidylinositol-3-phosphate (PI3P), which is required for the formation of the autophagosome. Moreover, there are several tumor suppressor proteins such as Atg4c, BAX-interacting factor-1 (Bif-1) homolog of phosphatase and tensin (PTEN) and UVRAG, which, besides inhibiting the growth of tumors, have common induction of autophagy (13) (Figure 1).

Figure 1. Molecular regulation of autophagy. Source: Own elaboration based on the data obtained in the study.
Autophagy responds to downregulation by stimuli of growth factors that regulate the phosphatidylinositol-3-kinase pathway (PI3K/AKT), which controls the activation of the mTOR pathway; the latter resides in a multi-protein and macromolecular (mTORC1) complex that is activated by signals associated with nutrients, including amino acids and growth factors, and downregulates autophagy by interacting with the complex Beclin 1.
Autophagy also responds to control due to cellular energy depletion through increased activity of protein kinase activated by AMP (AMPK). In response to elevated levels of 5’-monophosphate adenosine AMP, the inactive AMPK mTORC1 and active Beclin 1 promote Atg9 traffic. Beclin 1 is associated with a macromolecular complex, which includes hVps34, PI3KC3 class III, p150 and UVRAG. The Beclin 1 complex produces phosphatidylinositol-3-phosphate (PI3P), which recruits factors associated with autophagosome formation.
For inhibition of autophagy, the serine/threonine kinase mTOR protein is the most important in human cells; inhibition occurs through maintenance of hyper-phosphorylation of proteins that are needed for initiating the autophagy signaling pathway. mTORC1 promotes protein synthesis, cell division and metabolism in response to the availability of nutrients, growth factors and hormones, while suppressing autophagy. Mutations acquired in different regions of the mTOR C-terminal promote its hyperactivation, benefiting the uncontrolled growth of tumor cells (2).
Types of autophagy
There are three main types of autophagy that work for eukaryotic cells: macroautophagy, microautophagy and chaperone-mediated autophagy (CMA), which are all different in terms of the mechanics of the process (Table 1).
Autophagy and its mechanism as a tumor suppressor
Although autophagy is a survival pathway —used by both normal and tumor cells to survive hunger and stress— paradoxically, its defects are found in many types of human tumors. Allelic loss of gene Beclin 1, essential for autophagy, is common in human breast, ovarian and prostate cancer (16).
In early stages of the tumor, autophagy acts as a tumor suppressor process, since it is responsible for inhibiting the inflammatory events associated with cancer, and it also promotes genomic stability (17). Furthermore, the accumulation of reactive oxygen species (ROS) is one of the main consequences of metabolic stress, which can cause damage to the structure of DNA through the induction of double strand breaks and change in the base sequence of the DNA, leading to activation of proto-oncogenes and, simultaneously, inactivation of tumor suppressor genes (18).
Table 1. Types of autophagy
|
Type |
Mechanism |
|
Macroautophagy |
Dynamic reordering of the membrane: In this type of autophagy, specialized vacuoles serve for transportation purposes; these vacuoles are called autophagosomes and provide protein aggregation, lipids and damaged organelles to lysosomes for degradation. |
|
Microautophagy |
Dynamic reordering of the membrane: Direct introduction of the cytoplasm in the lysosomal surface is made by invagination and protrusion; consequently cells degrade through lysosomal hydrolases once the lysosome is completely closed. |
|
Chaperone-mediated autophagy (CMA) |
Kidnapping of proteins containing KFERQ substrate: This mechanism is mediated by the Hsc70 complex and the cytosolic chaperone protein recognition, which are responsible for the translocation of the substrates deployed across the lysosomal membrane; once inside the lysosome, proteins are degraded by lysosomal hydrolases action. |
Source: Own elaboration based on Yorimitsu & Klionsky (14) y Chen & Klionsky (15).
For several years, ROS have been linked to cancer development in humans; after many studies, it has been concluded that autophagy plays an important role in reducing levels of ROS (19). The existence of high levels of ROS activates autophagy to eliminate these harmful compounds and, in consequence, prevent DNA damage and the development of tumorigenesis (18).
The study by Cao B et al. (20) shows that anti-microbial agents such as clioquinol, can induce tumor cell death; basically, its anti-tumor properties are given by their ability to activate autophagy in cancer cells by increasing the PI3KC3/Beclin 1 complex and disrupting the mTOR signaling pathway.
Autophagy and its mechanism as tumor inducer
One of the most remarkable abilities in the repertoire of tumor cells is the activation of autophagy in response to stress, which allows long-term survival, particularly when apoptosis is defective. Apoptosis might normally eliminate stress resistant tumor cells as a tumor suppressor mechanism; however, tumor cells often evolve to generate defects in this process, allowing activation of autophagy to sustain the survival under nutrient deprivation conditions. Tumor cells can digest themselves gradually under prolonged stress, becoming less than a third of normal size (Figure 2) (21).
Cellular stress activates autophagy in tumor cells, allowing survival by promoting selection of material for cell consumption. As a consequence, small cells that may remain in a dormant state in the presence of stress are generated, but these cells, capable to recover (complete cytogenetic response-CCR) regenerate and restore their cellular proliferation when stress disappears.
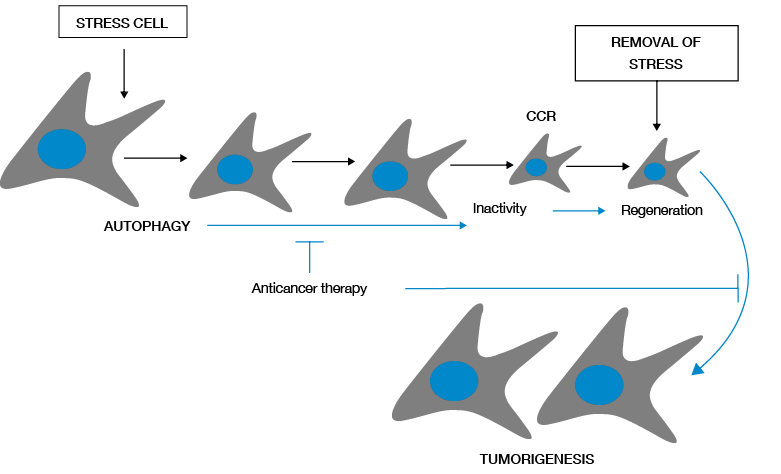
Figure 2. Survival and regeneration mediated by autophagy in tumor cells. Source: Own elaboration based on the data obtained in the study.
Establishing cell latency as a regenerative capacity is highly dependent on autophagy; in tumor cells with defects in autophagy, achieving latency and cell regeneration is less efficient. Therefore, autophagy confers tumor cells tolerance to cellular stress, limiting damage and maintaining cell viability (Figure 3) (22).
Although autophagy mitigates damage and promotes cellular senescence, inhibiting tumorigenesis by allowing tumor cells to survive cellular stress, remain dormant and regenerate with elimination of stress, it also promotes tumorigenesis.
Other factors that can originate and stimulate tumorigenesis are damaged tumor cells in tumors with defects in autophagy, particularly those with protein aggregates and genomic damage, and the presence of chronic inflammation, which generates a favorable microenvironment that alters cell death.
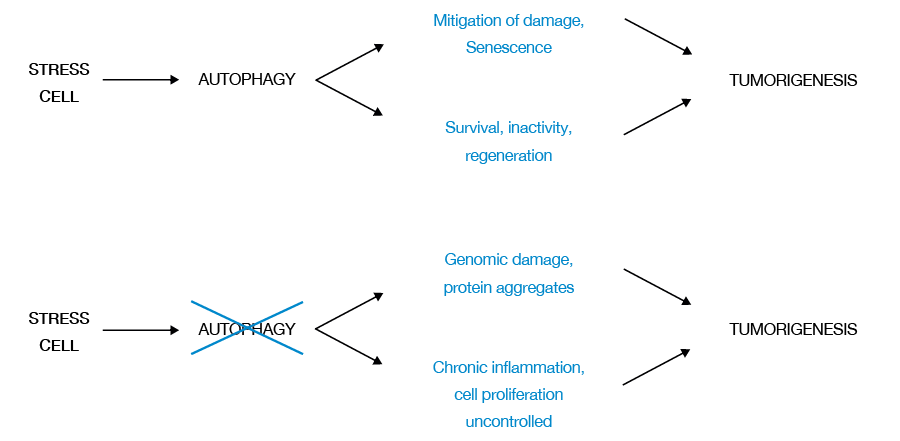
Figure 3. Two-way function of autophagy in tumorigenesis. Source: Own elaboration based on the data obtained in the study.
Targeting autophagy for cancer therapy
The role of autophagy in oncogenesis is variable, since it is a tumor suppressor during the early stages of the tumor and may contribute to their growth during their development (23). Autophagy, as a response to cancer therapy, can promote/suppress tumor development, for this reason, improving cancer therapy is considered an unusual goal.
Tumor cells with competent autophagy
This type of cells can activate autophagy as an adaptive response to therapeutic agents against cancer, therefore, autophagy could act as a resistance or survival mechanism of tumor cells in prolonged treatments. The absence of cancer cells in an essential mechanism for resistance, by specifically inhibiting autophagy, is expected to improve the efficacy of anticancer drugs (24). Cancer cells with defective apoptosis and low metabolic stress have been proved to establish autophagy activation as a survival mechanism; in contrast, tumor cells with competent apoptosis under stress may undergo rapid cell death following activation of apoptosis. Therefore, inhibition of autophagy is expected to be therapeutically more beneficial in the treatment of tumors that have defects in apoptosis (25).
Autophagy inhibition: therapeutic target
against cancer
Inhibition of autophagy as a scenario to sensitize tumor cells to anti-neoplastic treatment has been validated in several studies. Inhibition of autophagy by chloroquine, a lysosomotropic agent which raises pH and interferes with autophagosome degradation within lysosomes, shows an improvement in anti-tumor activity of cyclophosphamide (alkylating agent) in a lymphoma induced model and colorectal cancer (26). Similarly, 3-methyladenine (3-MA), an autophagy inhibitor, allows sensitization in nasopharyngeal carcinoma cells (Hone-1) to treatment with cisplatin and radiotherapy, which relates to the prevention of endoplasmic reticulum stress induced by autophagy in such cells (27).
Several studies support the idea that autophagy, as a physiological process in response to treatments in cancer cells, can help tumors evade drug - induced cytotoxicity —as survival mechanism—.Thus, it was demonstrated in non-small cell lung cancer (NSCLC) that autophagy regulates their resistance to treatment with paclitaxel, mainly by decreasing microRNA-216b (miR-216b); therefore, strategies that increase levels of miR-216b or inhibit cell autophagy can improve the outcome of treatment with paclitaxel against NSCLC (28). Furthermore, it has been shown that chemoresistance of patients with hepatocellular carcinoma (HCC), compared to treatment with cisplatin, is the consequence of activation of autophagy by binding lectin beta-galactoside, and galectin-1 (29).
In order to determine the molecular mechanisms of the chemotherapeutic effect of chloroquine on malignant glioblastomas, recent studies have been devoted to probing the cytotoxicity of chloroquine in combination with temozolomide (TMZ), taking ROS as one of the main causes of dysfunctional mitochondria.
Hori YS et al. found that chloroquine increases cellular ROS and cytotoxicity of TMZ in glioma cells by inhibiting mitochondrial autophagy (30,31). Also, recent studies in patients with positive estrogen receptor (ER (+)) showed a significant increase in sensitivity to apoptosis in breast cancer induced by tamoxifen and fulvestrant after inhibition of autophagy induced by microRNA 214 (miR-214). These results support the regulation of autophagy as a new therapeutic strategy for overcoming endocrine resistance in breast cancers ER (+) (32).
Autophagy is commonly regulated in tumor and normal cells exposed to cancer therapies, but the greater dependence of tumor cells —compared with normal cells— on the cytoprotective effects of autophagy offers a new therapeutic opportunity. In fact, autophagy is induced as a strategy for survival in human tumor cells treated with histone deacetylase inhibitors (HDAC) (33), arsenic trioxide (34), tumor necrosis factor alpha (TNF α) (35), gamma interferon (IFN-γ) (36), rapamycin (37) and hormone therapy antiestrogen (38), suggesting that inhibition of autophagy could reduce resistance of cancer cells in these therapies.
Another strategy for inhibition of autophagy includes the use of siRNAs (small interfering RNA), which target autophagy essential genes and sensitize cancer cells to the induction of cell death by radiation cells (39), and a wide range of chemotherapeutic agents, including cyclophosphamide and N-(4-hydroxyphenyl) retinamide (40).
Tumor cells with defective autophagy
These tumors probably adapt to a state of defective autophagy over time and acquire compensatory mechanisms of cell survival. Hence, cancer cells with defects in autophagy are not expected to rely on this mechanism for cyto-protection during chemotherapy and radiotherapy; inhibition of autophagy cannot increase cytotoxicity by anti-neoplastic or irradiation drugs (41).
Moreover, tumor cells with defective autophagy probably have high susceptibility to metabolic stress, high levels of DNA damage and propensity to genomic instability, which are properties with different implications for responsiveness to anti-neoplastic treatments (42-43). Although, it is still poorly documented, tumor cells with defective autophagy may be particularly sensitive to metabolic stress induction regimens, such as anti-angiogenic pharmaceutical drugs, growth factors receptor inhibitors, glucose deprivation and agents that induce DNA damage, including platinum compounds and topoisomerase inhibitors (44).
Induction of autophagy: therapeutic target against cancer
Since defects in apoptosis are often observed in many tumor cells and may increase their resistance to several conventional therapies for carcinogenesis, targeting alternative pathways to cell death is an attractive strategy to improve anti-tumor therapy (45). Consequently, induction of autophagic cell death can serve as a novel therapeutic strategy to eliminate the development of various cancers, especially those with high thresholds of deficient apoptosis (46).
Several studies have reported that different agents, including arsenic trioxide (46) and the vitamin D analog EB1089 (47), induce autophagic cell death in tumor cells in vitro; unfortunately, in these cases, autophagic cell death was determined based on morphological characteristics, so the studies may not represent a true autophagic cell death (48,49). However, other reports have shown specific examples of autophagic cell death in response to certain agents.
Some tumor cells, especially those lacking essential modulators of apoptosis such as BAX, BAK or caspases, exhibit cell death autophagy in vitro when treated with certain chemotherapy drugs, such as etoposide, fenretinide and dexamethasone (50-52).
Also, other studies have shown that polyphenols activate autophagy, controlling cell regulator mechanisms; these results provide strong support to the idea that plant polyphenols are really useful in treating diseases such as cancer, where autophagy plays an important role (53).
Induction of autophagy, in response to nutrient starvation, is responsible for the beneficial effects on longevity in the presence of caloric restriction; at least in Caenorhabditis elegans, its activation slows aging and prolongs useful life (54).
It is tempting to speculate that periodic induction of autophagy may also be responsible for a preventive effect against the development of cancer processes in the presence of caloric restriction; if this were true, the pharmacological induction of autophagy could be used for chemoprevention of cancer. Future studies are needed to clarify whether the induction of autophagic cell death in cancer has a relevant clinical utility.
Conclusions
Autophagy can act in two ways during cancer development: as a mechanism of tumor suppression or as an adaptive response to stress to maintain cell survival. Nonetheless, the molecular mechanisms underlying the regulation of autophagy and the role of this process in tumor cells is not fully understood yet. For this reason, pharmacological modulation of autophagy may have significant clinical potential as a new therapeutic strategy for the eradication of cancer.
Induction of autophagy may be useful for cancer chemoprevention in normal cells or for triggering an alternative cell death mechanism in certain tumor cells, especially those with compromised apoptotic functions. Furthermore, deletion of autophagic pathways can be combined with conventional antitumor regimens to achieve greater efficiency, thereby, avoiding drug resistance in tumor cells, which represents a valuable therapeutic strategy for radio and chemo-sensitization.
On the other hand, additional questions and concerns arise, as it is known that autophagy inhibits oxidative stress, inflammation and genome instability, favoring tumor suppression in some models; it is still to determine whether these events contribute to the suppression of human cancer. If so, the essential autophagy gene should be represented among genes with recurrent mutations in the development of human cancers. Based on currently available data, this does not seem to be the case, but there is a possibility that the loss of tumor suppression by autophagy in cancer occurs indirectly.
The use of autophagy against cancer offers new opportunities for drug development, as more potent and specific inhibitors of this process are clearly needed to ensure the efficacy and safety of anti-tumor treatment. Future efforts should focus on the modulation of autophagy for maximum therapeutic benefit, as well as in elucidating the genetic and physiological conditions that determine the function of pro-survival or pro-death autophagy.
A major limitation of the research to date is that all models of cancer have addressed the role of autophagy only in tumors, leaving aside the direct comparison with this deficiency in normal tissues. Since, there is evidence that autophagy is important for some normal tissues, a critical question is whether the systemic inactivation of this process is selective enough to harm cancer growth without affecting normal tissues with harmful consequences.
Ultimately, the pharmacological manipulation of autophagy for prevention and treatment of cancer depends on the ability to correctly recognize the functional status of autophagy in tumors and the availability of specific modulators.
Conflict of interests
None stated by the authors.
Funding
None stated by the authors.
Acknowledgements
The authors express their gratitude to the Faculty of Chemistry and Pharmaceutical Sciences of Universidad del Atlántico for allowing the use of their facilities to conduct this research.
References
1.Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100(1):57-70. http://doi.org/bm35gq.
2.White E. The role for autophagy in cancer. J. Clin. Invest. 2015;125(1):42-6. http://doi.org/bj9g.
3.White E, Mehnert JM, Chan CS. Autophagy, Metabolism, and Cancer. Clin. Cancer Res. 2015;21(22):5037-46. http://doi.org/bj9h.
4.Chen HY, White E. Role of autophagy in cancer prevention. Cancer Prev. Res. (Phila). 2011;4(7):973-983. http://doi.org/bf9z39.
5.Deretic V. Autophagy as an immune defense mechanism. Curr. Opin. Immunol. 2006;18(4):375-82. http://doi.org/d9wkvq.
6.Deretic V. Autophagy in innate and adaptive immunity. Trends Immunol. 2005;26(10):523-8. http://doi.org/d9g974.
7.Hussey S, Travassos LH, Jones NL. Autophagy as an emerging dimension to adaptive and innate immunity. Semin. Immunol. 2009;21(4):233-41. http://doi.org/bw9s44.
8.Nakatogawa H, Suzuki K, Kamada Y, Ohsumi Y. Dynamics and diversity in autophagy mechanisms: lessons from yeast. Nat. Rev. Mol. Cell Biol. 2009;10(7):458-67. http://doi.org/ccbhzx.
9.Yang Z, Klionsky DJ. An Overview of the Molecular Mechanism of Autophagy. Curr. Top. Microbiol. Immunol. 2009;335:1-32.
http://doi.org/bg4j72.
10.Ryter SW, Choi AM. Autophagy in lung disease pathogenesis and therapeutics. Redox Biology 2015;4:215-225. http://doi.org/bj9j.
11.He C, Levine B. The Beclin 1 interactome. Curr. Opin. Cell Biol. 2010;22(2):140-149. http://doi.org/c3jb3j.
12.Itakura E, Kishi C, Inoue K, Mizushima N. Beclin 1 forms two distinct phosphatidylinositol 3-kinase complexes with mammalian Atg14 and UVRAG. Mol. Biol. Cell. 2008;19(12):5360-5372. http://doi.org/b5tc58.
13.Mizushima N, Komatsu M. Autophagy: renovation of cells and tissues. Cell. 2011;147(4):728-741. http://doi.org/czfssg.
14.Yorimitsu T, Klionsky DJ. Autophagy: molecular machinery for self-eating. Cell Death Differ. 2005;12(Suppl 2):1542-1552. http://doi.org/cqg68q.
15.Chen Y, Klionsky DJ. The regulation of autophagy - unanswered questions. J. Cell. Sci. 2011;124(Pt 2):161-170. http://doi.org/cds32h.
16.Aita VM, Liang XH, Murty VV, Pincus DL, Yu W, Cayanis E, et al. Cloning and genomic organization of beclin 1, a candidate tumor suppressor gene on chromosome 17q21. Genomics. 1999;59(1):59-65. http://doi.org/dcx7cc.
17.Wu WK, Coffelt SB, Cho CH, Wang XJ, Lee CW, Chan FK, et al. The autophagic paradox in cancer therapy. Oncogene. 2012;31(8):939-53. http://doi.org/cndfzf.
18.Wiseman H, Halliwell B. Damage to DNA by reactive oxygen and nitrogen species: role in inflammatory disease and progression to cancer. Biochem. J. 1996;313(Pt 1):17-29. http://doi.org/bj9k.
19.Rouschop KM, Ramaekers CH, Schaaf MB, Keulers TG, Savelkouls KG, Lambin P, et al. Autophagy is required during cycling hypoxia to lower production of reactive oxygen species. Radiother. Oncol. 2009;92(3):411-6. http://doi.org/dhmhdg.
20.Cao B, Li J, Zhou X, Juan J, Han K, Zhang Z, et al. Clioquinol induces pro-death autophagy in leukemia and myeloma cells by disrupting the mTOR signaling pathway. Sci. Rep. 2014;4:5749. http://doi.org/bj9m.
21.White E, Karp C, Strohecker AM, Guo Y, Mathew R. Role of autophagy in suppression of inflammation and cancer. Curr. Opin. Cell Biol. 2010;22(2):212-7. http://doi.org/bxbs25.
22.Yang ZJ, Chee CE, Huang S, Sinicrope F. Autophagy modulation for cancer therapy. Cancer Biol. Ther. 2011;11(2):169-76. http://doi.org/cbjmwn.
23.Maiuri MC, Tasdemir E, Criollo A, Morselli E, Vicencio JM, Carnuccio R, et al. Control of autophagy by oncogenes and tumor suppressor genes. Cell Death Differ. 2009;16(1):87-93. http://doi.org/b3m4qx.
24.Abedin MJ, Wang D, McDonnell MA, Lehmann U, Kelekar A. Autophagy delays apoptotic death in breast cancer cells following DNA damage. Cell Death Differ. 2007;14(3):500-10. http://doi.org/dwqztk.
25.Maiuri MC, Zalckvar E, Kimchi A, Kroemer G. Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat. Rev. Mol. Cell. Biol. 2007;8(9):741-52. http://doi.org/ccrfnx.
26.Amaravadi RK, Yu D, Lum JJ, Bui T, Christophorou MA, Evan GI, et al. Autophagy inhibition enhances therapy-induced apoptosis in a Myc-induced model of lymphoma. J. Clin. Invest. 2007;117:326-336. http://doi.org/bbfk6b.
27.Song L, Ma L, Chen G, Huang Y, Sun X, Jiang C, et al. [Autophagy inhibitor 3-methyladenine enhances the sensitivity of nasopharyngeal carcinoma cells to chemotherapy and radiotherapy]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2016;41(1):9-18. Chinese. http://doi.org/bj9q.
28.Chen K, Shi W. Autophagy regulates resistance of non-small cell lung cancer cells to paclitaxel. Tumour Biol. 2016:1-5. http://doi.org/bj9r.
29.Su YC, Davuluri GV, Chen CH, Shiau DC, Chen CC, Chen CL, et al. Galectin-1-induced autophagy facilitates cisplatin resistance of hepatocellular carcinoma. PLoS One. 2016;11(2):e0148408. http://doi.org/bj9s.
30.Yan Y, Xu Z, Dai S, Qian L, Sun L, Gong Z. Targeting autophagy to sensitive glioma to temozolomide treatment. J. Exp. Clin. Cancer Res. 2016;35:23. http://doi.org/bj9t
31.Hori YS, Hosoda R, Akiyama Y, Sebori R, Wanibuchi M, Mikami T, et al. Chloroquine potentiates temozolomide cytotoxicity by inhibiting mitochondrial autophagy in glioma cells. J. Neurooncol. 2015;122(1):11-20. http://doi.org/bj9v.
32.Yu X, Luo A, Liu Y, Wang S, Li y, Shi W, et al. MiR-214 increases the sensitivity of breast cancer cells to tamoxifen and fulvestrant through inhibition of autophagy. Mol. Cancer. 2015;14(1):208. http://doi.org/bj9w.
33.Carew JS, Nawrocki ST, Kahue CN, Zhang H, Yang C, Chung L, et al. Targeting autophagy augments the anticancer activity of the histone deacetylase inhibitor SAHA to overcome Bcr-Abl-mediated drug resistance. Blood. 2007;110(1):313-22. http://doi.org/b65ggd.
34.Smith DM, Patel S, Raffoul F, Haller E, Mills GB, Nanjundan M. Arsenic trioxide induces a beclin-1-independent autophagic pathway via modulation of SnoN/SkiL expression in ovarian carcinoma cells. Cell Death Differ. 2010;17(12):1867-81. http://doi.org/b384pd.
35.Moussay E, Kaoma T, Baginska J, Muller A, Van Moer K, Nicot N, et al. The acquisition of resistance to TNFα in breast cancer cells is associated with constitutive activation of autophagy as revealed by a transcriptome analysis using a custom microarray. Autophagy. 2011;7(7):760-70. http://doi.org/dpkg5m.
36.Ní Cheallaigh C, Keane J, Lavelle EC, Hope JC, Harris J. Autophagy in the immune response to tuberculosis: clinical perspectives. Clin. Exp. Immunol. 2011;164(3):291-300. http://doi.org/czf38x.
37.Fan QW, Cheng C, Hackett C, Feldman M, Houseman BT, Nicolaides T, et al. Akt and autophagy cooperate to promote survival of drug-resistant glioma. Sci. Signal. 2010;3(147):ra81. http://doi.org/d7wpc6.
38.Qadir MA, Kwok B, Dragowska WH, To KH, Le D, Bally MB, et al. Macroautophagy inhibition sensitizes tamoxifen-resistant breast cancer cells and enhances mitochondrial depolarization. Breast Cancer Res. Treat. 2008;112(3):389-403. http://doi.org/cq77bc.
39.Apel A, Herr I, Schwarz H, Rodemann HP, Mayer A. Blocked autophagy sensitizes resistant carcinoma cells to radiation therapy. Cancer Res. 2008;68(5):1485-94. http://doi.org/bgvbv8.
40.Tiwari M, Bajpai VK, Sahasrabuddhe AA, Kumar A, Sinha RA, Behari S, et al. Inhibition of N-(4-hydroxyphenyl) retinamide-induced autophagy at a lower dose enhances cell death in malignant glioma cells. Carcinogenesis. 2008;29:600-609. http://doi.org/bdz37n.
41.Karantza-Wadsworth V, Patel S, Kravchuk O, Chen G, Mathew R, Jin S, et al. Autophagy mitigates metabolic stress and genome damage in mammary tumorigenesis. Genes Dev. 2007;21:1621-35. http://doi.org/ffxwn5.
42.Mathew R, Kongara S, Beaudoin B, Karp CM, Bray K, Degenhardt K, et al. Autophagy suppresses tumor progression by limiting chromosomal instability. Genes Dev. 2007;21(11):1367-81. http://doi.org/cgvp5x.
43.Chen N, Karantza-Wadsworth V. Role and regulation of autophagy in cancer. Biochim Biophys. Acta. 2009;1793(9):1516-23. http://doi.org/b94m99.
44.Schleicher SM, Moretti L, Varki V, Lu B. Progress in the unraveling of the endoplasmic reticulum stress/autophagy pathway and cancer: implications for future therapeutic approaches. Drug Resist. Updat. 2010;13(3):79-86. http://doi.org/b7z92b.
45.Gozuacik D, Kimchi A. Autophagy and cell death. Curr. Top. Dev. Biol. 2007;78:217-45. http://doi.org/cwrfcr.
46.Kanzawa T, Kondo Y, Ito H, Kondo S, Germano I. Induction of autophagic cell death in malignant glioma cells by arsenic trioxide. Cancer Res. 2003;63(9):2103-8.
47.Høyer-Hansen M, Bastholm L, Mathiasen IS, Elling F, Jäättelä M. Vitamin D analog EB1089 triggers dramatic lysosomal changes and Beclin 1-mediated autophagic cell death. Cell Death Differ. 2005;12(10):1297-309. http://doi.org/bb89wt.
48.Kroemer G, Levine B. Autophagic cell death: the story of a misnomer. Nat. Rev. Mol. Cell Biol. 2008;9(12):1004-10. http://doi.org/fj7c8w.
49.Shimizu S, Kanaseki T, Mizushima N, Mizuta T, Arakawa-Kobayashi S, Thompson CB, et al. Role of Bcl-2 family proteins in a non-apoptotic programmed cell death dependent on autophagy genes. Nat. Cell Biol. 2004;6(12):1221-8. http://doi.org/bqdc7z.
50.Fazi B, Bursch W, Fimia GM, Nardacci R, Piacentini M, Di Sano F, et al. Fenretinide induces autophagic cell death in caspase-defective breast cancer cells. Autophagy. 2008;4(4):435-41. http://doi.org/bj9x.
51.Grandér D, Kharaziha P, Laane E, Pokrovskaja K, Panaretakis T. Autophagy as the main means of cytotoxicity by glucocorticoids in hematological malignancies. Autophagy. 2009;5(8):1198-200. http://doi.org/dkqzfg.
52.Laane E, Tamm KP, Buentke E, Ito K, Kharaziha P, Oscarsson J, et al. Cell death induced by dexamethasone in lymphoid leukemia is mediated through initiation of autophagy. Cell Death Differ. 2009;16(7):1018-29. http://doi.org/fbbq4b.
53.Rigacci S, Miceli C, Nediani C, Berti A, Cascella R, Pantano D, et al. Oleuropein aglycone induces autophagy via the AMPK/mTOR signalling pathway: a mechanistic insight. Oncotarget. 2015;6(34):35344-57. http://doi.org/bj9z.
54.Tóth ML, Sigmond T, Borsos E, Barna J, Erdélyi P, Takács-Vellai K et al. Longevity pathways converge on autophagy genes to regulate life span in Caenorhabditis elegans. Autophagy. 2008;4:330-8. http://doi.org/bj92.

César Alexander Eslava Franco
“Mapas anatómicos”
Universidad Nacional de Colombia
Referencias
Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100(1):57-70. http://doi. org/bm35gq.
White E. The role for autophagy in cancer. J. Clin. Invest. 2015;125(1):42-6. http://doi. org/bj9g.
White E, Mehnert JM, Chan CS. Autophagy, Metabolism, and Cancer. Clin. Cancer Res. 2015;21(22):5037-46. http://doi. org/bj9h.
Chen HY, White E. Role of autophagy in cancer prevention. Cancer Prev. Res. (Phila). 2011;4(7):973-983. http://doi. org/bf9z39.
Deretic V. Autophagy as an immune defense mechanism. Curr. Opin. Immunol. 2006;18(4):375-82. http://doi. org/d9wkvq.
Deretic V. Autophagy in innate and adaptive immunity. Trends Immunol. 2005;26(10):523-8. http://doi. org/d9g974.
Hussey S, Travassos LH, Jones NL. Autophagy as an emerging dimension to adaptive and innate immunity. Semin. Immunol. 2009;21(4):233-41. http://doi. org/bw9s44.
Nakatogawa H, Suzuki K, Kamada Y, Ohsumi Y. Dynamics and diversity in autophagy mechanisms: lessons from yeast. Nat. Rev. Mol. Cell Biol. 2009;10(7):458-67. http://doi. org/ccbhzx.
Yang Z, Klionsky DJ. An Overview of the Molecular Mechanism of Autophagy. Curr. Top. Microbiol. Immunol. 2009;335:1-32.
http://doi. org/bg4j72.
Ryter SW, Choi AM. Autophagy in lung disease pathogenesis and therapeutics. Redox Biology 2015;4:215-225. http://doi. org/bj9j.
He C, Levine B. The Beclin 1 interactome. Curr. Opin. Cell Biol. 2010;22(2):140-149. http://doi. org/c3jb3j.
Itakura E, Kishi C, Inoue K, Mizushima N. Beclin 1 forms two distinct phosphatidylinositol 3-kinase complexes with mammalian Atg14 and UVRAG. Mol. Biol. Cell. 2008;19(12):5360-5372. http://doi. org/b5tc58.
Mizushima N, Komatsu M. Autophagy: renovation of cells and tissues. Cell. 2011;147(4):728-741. http://doi. org/czfssg.
Yorimitsu T, Klionsky DJ. Autophagy: molecular machinery for self-eating. Cell Death Differ. 2005;12(Suppl 2):1542-1552. http://doi. org/cqg68q.
Chen Y, Klionsky DJ. The regulation of autophagy - unanswered questions. J. Cell. Sci. 2011;124(Pt 2):161-170. http://doi. org/cds32h.
Aita VM, Liang XH, Murty VV, Pincus DL, Yu W, Cayanis E, et al. Cloning and genomic organization of beclin 1, a candidate tumor suppressor gene on chromosome 17q21. Genomics. 1999;59(1):59-65. http://doi. org/dcx7cc.
Wu WK, Coffelt SB, Cho CH, Wang XJ, Lee CW, Chan FK, et al. The autophagic paradox in cancer therapy. Oncogene. 2012;31(8):939-53. http://doi. org/cndfzf.
Wiseman H, Halliwell B. Damage to DNA by reactive oxygen and nitrogen species: role in inflammatory disease and progression to cancer. Biochem. J. 1996;313(Pt 1):17-29. http://doi. org/bj9k.
Rouschop KM, Ramaekers CH, Schaaf MB, Keulers TG, Savelkouls KG, Lambin P, et al. Autophagy is required during cycling hypoxia to lower production of reactive oxygen species. Radiother. Oncol. 2009;92(3):411-6. http://doi. org/dhmhdg.
Cao B, Li J, Zhou X, Juan J, Han K, Zhang Z, et al. Clioquinol induces pro-death autophagy in leukemia and myeloma cells by disrupting the mTOR signaling pathway. Sci. Rep. 2014;4:5749. http://doi. org/bj9m.
White E, Karp C, Strohecker AM, Guo Y, Mathew R. Role of autophagy in suppression of inflammation and cancer. Curr. Opin. Cell Biol. 2010;22(2):212-7. http://doi. org/bxbs25.
Yang ZJ, Chee CE, Huang S, Sinicrope F. Autophagy modulation for cancer therapy. Cancer Biol. Ther. 2011;11(2):169-76. http://doi. org/cbjmwn.
Maiuri MC, Tasdemir E, Criollo A, Morselli E, Vicencio JM, Carnuccio R, et al. Control of autophagy by oncogenes and tumor suppressor genes. Cell Death Differ. 2009;16(1):87-93. http://doi. org/b3m4qx.
Abedin MJ, Wang D, McDonnell MA, Lehmann U, Kelekar A. Autophagy delays apoptotic death in breast cancer cells following DNA damage. Cell Death Differ. 2007;14(3):500-10. http://doi. org/dwqztk.
Maiuri MC, Zalckvar E, Kimchi A, Kroemer G. Self-eating and self-killing: crosstalk between autophagy and apoptosis. Nat. Rev. Mol. Cell. Biol. 2007;8(9):741-52. http://doi. org/ccrfnx.
Amaravadi RK, Yu D, Lum JJ, Bui T, Christophorou MA, Evan GI, et al. Autophagy inhibition enhances therapy-induced apoptosis in a Myc-induced model of lymphoma. J. Clin. Invest. 2007;117:326-336. http://doi. org/bbfk6b.
Song L, Ma L, Chen G, Huang Y, Sun X, Jiang C, et al. [Autophagy inhibitor 3-methyladenine enhances the sensitivity of nasopharyngeal carcinoma cells to chemotherapy and radiotherapy]. Zhong Nan Da Xue Xue Bao Yi Xue Ban. 2016;41(1):9-18. Chinese. http://doi. org/bj9q.
Chen K, Shi W. Autophagy regulates resistance of non-small cell lung cancer cells to paclitaxel. Tumour Biol. 2016:1-5. http://doi. org/bj9r.
Su YC, Davuluri GV, Chen CH, Shiau DC, Chen CC, Chen CL, et al. Galectin-1-induced autophagy facilitates cisplatin resistance of hepatocellular carcinoma. PLoS One. 2016;11(2):e0148408. http://doi. org/bj9s.
Yan Y, Xu Z, Dai S, Qian L, Sun L, Gong Z. Targeting autophagy to sensitive glioma to temozolomide treatment. J. Exp. Clin. Cancer Res. 2016;35:23. http://doi. org/bj9t
Hori YS, Hosoda R, Akiyama Y, Sebori R, Wanibuchi M, Mikami T, et al. Chloroquine potentiates temozolomide cytotoxicity by inhibiting mitochondrial autophagy in glioma cells. J. Neurooncol. 2015;122(1):11-20. http://doi. org/bj9v.
Yu X, Luo A, Liu Y, Wang S, Li y, Shi W, et al. MiR-214 increases the sensitivity of breast cancer cells to tamoxifen and fulvestrant through inhibition of autophagy. Mol. Cancer. 2015;14(1):208. http://doi. org/bj9w.
Carew JS, Nawrocki ST, Kahue CN, Zhang H, Yang C, Chung L, et al. Targeting autophagy augments the anticancer activity of the histone deacetylase inhibitor SAHA to overcome Bcr-Abl-mediated drug resistance. Blood. 2007;110(1):313-22. http://doi. org/b65ggd.
Smith DM, Patel S, Raffoul F, Haller E, Mills GB, Nanjundan M. Arsenic trioxide induces a beclin-1-independent autophagic pathway via modulation of SnoN/SkiL expression in ovarian carcinoma cells. Cell Death Differ. 2010;17(12):1867-81. http://doi. org/b384pd.
Moussay E, Kaoma T, Baginska J, Muller A, Van Moer K, Nicot N, et al. The acquisition of resistance to TNFα in breast cancer cells is associated with constitutive activation of autophagy as revealed by a transcriptome analysis using a custom microarray. Autophagy. 2011;7(7):760-70. http://doi. org/dpkg5m.
Ní Cheallaigh C, Keane J, Lavelle EC, Hope JC, Harris J. Autophagy in the immune response to tuberculosis: clinical perspectives. Clin. Exp. Immunol. 2011;164(3):291-300. http://doi. org/czf38x.
Fan QW, Cheng C, Hackett C, Feldman M, Houseman BT, Nicolaides T, et al. Akt and autophagy cooperate to promote survival of drug-resistant glioma. Sci. Signal. 2010;3(147):ra81. http://doi. org/d7wpc6.
Qadir MA, Kwok B, Dragowska WH, To KH, Le D, Bally MB, et al. Macroautophagy inhibition sensitizes tamoxifen-resistant breast cancer cells and enhances mitochondrial depolarization. Breast Cancer Res. Treat. 2008;112(3):389-403. http://doi. org/cq77bc.
Apel A, Herr I, Schwarz H, Rodemann HP, Mayer A. Blocked autophagy sensitizes resistant carcinoma cells to radiation therapy. Cancer Res. 2008;68(5):1485-94. http://doi. org/bgvbv8.
Tiwari M, Bajpai VK, Sahasrabuddhe AA, Kumar A, Sinha RA, Behari S, et al. Inhibition of N-(4-hydroxyphenyl) retinamide-induced autophagy at a lower dose enhances cell death in malignant glioma cells. Carcinogenesis. 2008;29:600-609. http://doi. org/bdz37n.
Karantza-Wadsworth V, Patel S, Kravchuk O, Chen G, Mathew R, Jin S, et al. Autophagy mitigates metabolic stress and genome damage in mammary tumorigenesis. Genes Dev. 2007;21:1621-35. http://doi. org/ffxwn5.
Mathew R, Kongara S, Beaudoin B, Karp CM, Bray K, Degenhardt K, et al. Autophagy suppresses tumor progression by limiting chromosomal instability. Genes Dev. 2007;21(11):1367-81. http://doi. org/cgvp5x.
Chen N, Karantza-Wadsworth V. Role and regulation of autophagy in cancer. Biochim Biophys. Acta. 2009;1793(9):1516-23. http://doi. org/b94m99.
Schleicher SM, Moretti L, Varki V, Lu B. Progress in the unraveling of the endoplasmic reticulum stress/autophagy pathway and cancer: implications for future therapeutic approaches. Drug Resist. Updat. 2010;13(3):79-86. http://doi. org/b7z92b.
Gozuacik D, Kimchi A. Autophagy and cell death. Curr. Top. Dev. Biol. 2007;78:217-45. http://doi. org/cwrfcr.
Kanzawa T, Kondo Y, Ito H, Kondo S, Germano I. Induction of autophagic cell death in malignant glioma cells by arsenic trioxide. Cancer Res. 2003;63(9):2103-8.
Høyer-Hansen M, Bastholm L, Mathiasen IS, Elling F, Jäättelä M. Vitamin D analog EB1089 triggers dramatic lysosomal changes and Beclin 1-mediated autophagic cell death. Cell Death Differ. 2005;12(10):1297-309. http://doi. org/bb89wt.
Kroemer G, Levine B. Autophagic cell death: the story of a misnomer. Nat. Rev. Mol. Cell Biol. 2008;9(12):1004-10. http://doi. org/fj7c8w.
Shimizu S, Kanaseki T, Mizushima N, Mizuta T, Arakawa-Kobayashi S, Thompson CB, et al. Role of Bcl-2 family proteins in a non-apoptotic programmed cell death dependent on autophagy genes. Nat. Cell Biol. 2004;6(12):1221-8. http://doi. org/bqdc7z.
Fazi B, Bursch W, Fimia GM, Nardacci R, Piacentini M, Di Sano F, et al. Fenretinide induces autophagic cell death in caspase-defective breast cancer cells. Autophagy. 2008;4(4):435-41. http://doi. org/bj9x.
Grandér D, Kharaziha P, Laane E, Pokrovskaja K, Panaretakis T. Autophagy as the main means of cytotoxicity by glucocorticoids in hematological malignancies. Autophagy. 2009;5(8):1198-200. http://doi. org/dkqzfg.
Laane E, Tamm KP, Buentke E, Ito K, Kharaziha P, Oscarsson J, et al. Cell death induced by dexamethasone in lymphoid leukemia is mediated through initiation of autophagy. Cell Death Differ. 2009;16(7):1018-29. http://doi. org/fbbq4b.
Rigacci S, Miceli C, Nediani C, Berti A, Cascella R, Pantano D, et al. Oleuropein aglycone induces autophagy via the AMPK/mTOR signalling pathway: a mechanistic insight. Oncotarget. 2015;6(34):35344-57. http://doi. org/bj9z.
Tóth ML, Sigmond T, Borsos E, Barna J, Erdélyi P, Takács-Vellai K et al. Longevity pathways converge on autophagy genes to regulate life span in Caenorhabditis elegans. Autophagy. 2008;4:330-8. http://doi. org/bj92.
Cómo citar
APA
ACM
ACS
ABNT
Chicago
Harvard
IEEE
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. Adrian Zając, Joanna Sumorek-Wiadro, Ewa Langner, Iwona Wertel, Aleksandra Maciejczyk, Bożena Pawlikowska-Pawlęga, Jarosław Pawelec, Magdalena Wasiak, Monika Hułas-Stasiak, Dorota Bądziul, Wojciech Rzeski, Michał Reichert, Joanna Jakubowicz-Gil. (2021). Involvement of PI3K Pathway in Glioma Cell Resistance to Temozolomide Treatment. International Journal of Molecular Sciences, 22(10), p.5155. https://doi.org/10.3390/ijms22105155.
2. Juan Pablo Ortiz-Aguire, Esteban Alejandro Velandi-Vargas, Oscar Mauricio Rodríguez-Bohorquez, Diego Amaya-Ramírez, David Bernal-Estévez, Carlos Alberto Parra-López. (2021). Personalized neoantigen-based cancer immunotherapy. A literature review. Revista de la Facultad de Medicina, 69(3), p.e81633. https://doi.org/10.15446/revfacmed.v69n3.81633.
3. Duygu Ağagündüz, Teslime Özge Şahin, Birsen Yılmaz, Kübra Damla Ekenci, Şehriban Duyar Özer, Raffaele Capasso, Abraham Wall Medrano. (2022). Cruciferous Vegetables and Their Bioactive Metabolites: from Prevention to Novel Therapies of Colorectal Cancer. Evidence-Based Complementary and Alternative Medicine, 2022, p.1. https://doi.org/10.1155/2022/1534083.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2017 Revista de la Facultad de Medicina

Esta obra está bajo una licencia Creative Commons Reconocimiento 3.0 Unported.
-