Diseño experimental para determinar el efecto del tamaño del pellet de catalizadores bimetálicos Cu-Ni en la reacción de desplazamiento de agua
Experimental design to determine the effect of the pellet size of bimetallic Cu-Ni catalysts in the water gas shift reaction
Desenho experimental para determinar o efeito do tamanho do pellet de catalisadores bimetálicos de Cu-Ni na reacção de deslocamento de agua
DOI:
https://doi.org/10.15446/rev.colomb.quim.v47n1.64699Palabras clave:
Reacción de desplazamiento de agua, pellets de Cu-Ni, diseño experimental. (es)water gas shift reaction, pellets of Cu-Ni, experimental design, carbon monoxide conversion (en)
reação de deslocamento do gás de água, grânulos de Cu-Ni, desenho experimental, conversão de monóxido de carbono (pt)
Se sintetizaron catalizadores bimetálicos de Cu-Ni en relación molar 2-1 en pellets cilíndricos de carbón activado de diferentes diámetros (0,2 cm; 0,3 cm y 0,4 cm) y longitud (0,4 cm y 0,6 cm); se evaluaron en la reacción de desplazamiento de agua a 330 °C y presión atmosférica. Se realizó un diseño experimental para evaluar el efecto de las variables, diámetro y longitud, sobre la conversión del monóxido de carbono. La importancia de los principales factores y sus interacciones se examinaron por medio del análisis de varianza (ANOVA). Los resultados mostraron que tanto el diámetro como la longitud afectan significativamente la conversión del monóxido de carbono. Los resultados de la actividad catalítica, bajo las mejores condiciones de diámetro (0,4 cm) y longitud (0,6 cm) de los pellets, mostraron una conversión del monóxido de carbono del 96%.

Red de Revistas Científicas de América Latina y el Caribe, España y Portugal
Aplicada y Analítica
Diseño experimental para determinar el efecto del tamaño del pellet de catalizadores bimetálicos Cu-Ni en la reacción de desplazamiento de agua
Experimental design to determine the effect of the pellet size of bimetallic Cu-Ni catalysts in the water gas shift reaction
Desenho experimental para determinar o efeito do tamanho do pellet de catalisadores bimetálicos de Cu-Ni na reacção de deslocamento de agua
Diseño experimental para determinar el efecto del tamaño del pellet de catalizadores bimetálicos Cu-Ni en la reacción de desplazamiento de agua
Revista Colombiana de Química, vol. 47, núm. 1, 2018
Universidad Nacional de Colombia
Recepción: 08 Mayo 2017
Aprobación: 29 Septiembre 2017
Resumen: Se sintetizaron catalizadores bimetálicos de Cu-Ni en relación molar 2-1 en pellets cilíndricos de carbón activado de diferentes diámetros (0,2 cm; 0,3 cm y 0,4 cm) y longitud (0,4 cm y 0,6 cm); se evaluaron en la reacción de desplazamiento de agua a 330 °C y presión atmosférica. Se realizó un diseño experimental para evaluar el efecto de las variables, diámetro y longitud, sobre la conversión del monóxido de carbono. La importancia de los principales factores y sus interacciones se examinaron por medio del análisis de varianza (ANOVA). Los resultados mostraron que tanto el diámetro como la longitud afectan significativamente la conversión del monóxido de carbono. Los resultados de la actividad catalítica, bajo las mejores condiciones de diámetro (0,4 cm) y longitud (0,6 cm) de los pellets, mostraron una conversión del monóxido de carbono del 96%.
Palabras clave: Reacción de desplazamiento de agua, pellets de Cu-Ni, diseño experimental, conversión de monóxido de carbono.
Abstract: Bimetallic Cu-Ni catalysts were synthesized with molar ratio 2-1 in cylindrical activated carbon pellets of different diameters (0.2 cm, 0.3 cm, and 0.4 cm) and length (0.4 cm and 0.6 cm). They were evaluated in the water gas shift reaction at 330 °C and atmospheric pressure. An experimental design was developed to evaluate the effect of variables, diameter and length of the pellets, over carbon monoxide conversion. The importance of the main factors and their interactions were examined by analysis of variance (ANOVA). The results showed that both diameter and length of the pellets had significant effect on carbon monoxide conversion. The results of the catalytic activity indicated that up to 96% CO conversion was obtained at the optimum characteristic of the pellets, diameter of 0.2 cm and length of 0.6 cm.
Keywords: water gas shift reaction, pellets of Cu-Ni, experimental design, carbon monoxide conversion.
Resumo: Foram sintetizados catalisadores bimetálicos de Cu-Ni em proporção molar 2-1 sobre pastilhas de carvão ativado de diferentes diâmetros (0,2 cm; 0,3 cm e 0,4 cm) e comprimentos (0,4 cm e 0,6 cm) e foram avaliados na reação de mudança de vapor de agua a 330 °C e pressão atmosférica. Um desenho experimental foi desenvolvido para avaliar o efeito de variáveis, diâmetro e comprimento, na conversão de monóxido de carbono. A importância dos principais fatores e as suas interações foram examinados por análise de variância (ANOVA), os resultados mostraram que tanto o diâmetro e o comprimento afetam significativamente a conversão de monóxido de carbono. Os resultados da atividade catalítica com as melhores condições de diâmetro (0,4 cm) e o comprimento (0,6 cm) das pastilhas mostrou uma conversão de monóxido de carbono de 96%.
Palavras-chave: reação de deslocamento do gás de água, grânulos de Cu-Ni, desenho experimental, conversão de monóxido de carbono.
Introducción
La reacción de desplazamiento de agua, Ecuación [1], conocida como water gas shift (WGS) es un proceso industrial que permite la conversión del monóxido de carbono y agua en dióxido de carbono e hidrógeno, además de su uso en la mezcla de gases de gran importancia en la síntesis de amoníaco y metanol (1), en procesos de refinería de hidrocarburos y en aplicaciones en las celdas de combustible, siendo estas últimas ampliamente estudiadas, dado su elevado potencial energético y la baja emisión de gases de efecto invernadero (2).

Sin embargo, uno de los principales inconvenientes de esta reacción es la limitación termodinámica del equilibrio. Dado su carácter exotérmico, ΔH0 = -41,2 kJ·mol-1, la conversión del monóxido de carbono y la producción de hidrógeno se favorecen a bajas temperaturas. La reacción se ha realizado en presencia de catalizadores heterogéneos que han permitido mejorar la conversión del monóxido de carbono y la selectividad hacia el hidrógeno.
Entre los catalizadores comúnmente utilizados se destacan Pt/CeO2 (3), Pt/TiO2 (4), Au/CeO2 (5), Au/TiO2 (6) y Rh/Al2O3 (7) aunque, debido al área superficial relativamente baja (50-400 m2·g-1), los elevados costos, la desactivación a temperaturas altas por sinterización del metal y la rápida desactivación de los mismos, bajo los ciclos de encendido y apagado frecuente (8), se motiva la sustitución de estos catalizadores por otros que contengan metales de menor costo, manteniendo el rendimiento catalítico del sistema.
En comparación con los metales nobles soportados, los catalizadores de Cu y Cu-Ni tales como Cu/ZrO2 (9), Cu-Ni/carbón activado (10), Cu-Ni/Al2O3 (11), Cu-Ni/CeO2 (12), Cu-Ni/SiO2 (13), han sido reportados como una alternativa prometedora en la reacción de desplazamiento de agua. Entre los diferentes soportes, el carbón activado es un material de elevada área superficial, económico, altamente disponible, y con adecuada resistencia mecánica y térmica (14). En un trabajo anterior (10) se reportó la preparación de catalizadores mono y bimetálicos Cu-Ni a partir de pellets cilíndricos (3 mm de diámetro y 4 mm de longitud) de carbón activado y su actividad en la reacción de desplazamiento de agua. Bajo las condiciones experimentales reportadas (mezcla modelo de reacción: 4,5% de CO; 30,5% de H2O, 65% de N2), se encontró que, a temperaturas mayores de 220 °C, la conversión del CO aumenta con la temperatura, siendo el catalizador bimetálico Cu-Ni en relación molar 2-1 el de mayor actividad catalítica. Específicamente, a 330 °C se obtuvo una conversión de CO del 99% y una selectividad del 98% hacia el hidrógeno.
Los análisis de difracción de rayos X del catalizador mostraron líneas de difracción con posiciones intermedias a las señales de Cu y Ni puros, lo cual evidenció la formación de la solución sólida Cu-Ni. Adicionalmente, el aumento del parámetro de red con el aumento de la concentración de Cu se ajustó a la Ley de Vegard, confirmando la formación de una solución sólida entre los dos metales, relacionada con la mayor actividad catalítica de este catalizador, en comparación con los materiales preparados con otras relaciones de Cu-Ni.
Estudios adicionales de espectroscopía fotoelectrónica de Rayos X (XPS) mostraron que el catalizador bimetálico Cu-Ni, en relación molar 2-1, exhibió señales de Cu2p3/2, Cu2p1/2, Ni 2p3/2 y Ni2p1/2 de 933,2; 953,1; 858,5 y 876,6; respectivamente. Estas se encuentran a una mayor energía de enlace que las respectivas señales de Cu2p3/2 (932,8 eV), Cu2p1/2 (952,8 eV), Ni 2p3/2 856,7 eV) y Ni2p1/2 (874,6 eV) de los catalizadores monometálicos de Cu y Ni, y se ha reportado como una evidencia de la formación de la solución sólida Cu-Ni. Adicionalmente, este mismo catalizador, mantuvo una conversión de CO estable (alrededor del 50%) durante 20 h de análisis continuo bajo condiciones reales de mezcla de reacción (9% CO; 30,5% H2O; 11% CO2; 50% H2; 1,5% N2) a 330 ºC.
Para catalizadores peletizados se ha determinado que la actividad catalítica está controlada por el diámetro y longitud de los mismos, los cuales afectan el volumen de los espacios vacíos que se forman durante el empaquetamiento. Del mismo modo, está directamente relacionado con la caída de presión y las limitaciones difusionales externas e internas del lecho catalítico (15).
En este artículo se reportan los resultados de un diseño experimental para estudiar el efecto de la longitud y el diámetro de catalizadores pelletizados Cu-Ni sobre la conversión de CO en la reacción de desplazamiento de agua. Para investigar las condiciones más adecuadas de diámetro y longitud de los catalizadores y establecer la conversión máxima de CO, se ajustó un modelo polinominal de segundo orden, se verificaron todos sus supuestos y se obtuvo la superficie de respuesta con la que se identificó la influencia de dichas variables en la actividad catalítica del sistema en términos de la conversión del CO.
Ahora bien, los resultados de diseño experimental se pueden analizar a partir de un enfoque paramétrico, conocido como análisis ANOVA, donde se debe cumplir el supuesto de normalidad, varianza constante e independencia en el término de error. También se pueden analizar mediante enfoques no paramétricos, por ejemplo el método de Kruscal-Wallis, cuando no es posible validar el supuesto de normalidad del término de error (16). Debido a lo anterior, se optó por el primer enfoque ya que con el modelo bajo consideración se validaron todos los supuestos del modelo.
Materiales y métodos
Diseño experimental y análisis estadístico
La determinación de las condiciones que maximizarían la conversión de CO, en el rango de trabajo de las variables estudiadas, se llevó a cabo en dos etapas: 1) la formulación del diseño de experimentos, y 2) el ajuste de un modelo polinomial a los datos experimentales para determinar el valor de los factores (diámetro y longitud) que satisficieran las condiciones más adecuadas.
Diseño de experimentos
El diseño de experimentos tiene por objeto reducir el número de pruebas experimentales, incluyendo diversas combinaciones que permitan estimar los efectos sinérgicos y antagonistas de la interacción entre el diámetro y la longitud del catalizador sobre la conversión del monóxido de carbono. Las variables del proceso fueron codificadas como +1, -1 y 0, las cuales corresponden a un valor alto, bajo y un punto central, respectivamente. El programa R (Project for statistical computing) versión 3.2.5 de análisis estadístico (17), se utilizó para generar el diseño experimental y analizar los datos obtenidos. Los experimentos se realizaron de acuerdo con un diseño de dos factores AxB con seis réplicas (experimentos). El número total de experimentos, los niveles examinados y los rangos experimentales de cada variable independiente se presentan en la Tabla 1 .
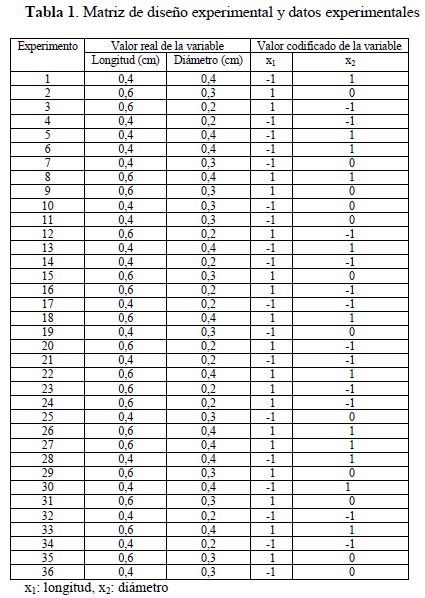
Matriz de diseño experimental y datos experimentales
x1: longitud, x2: diámetroAjuste del modelo
Un modelo polinomial permite determinar el valor más adecuado de las variables del sistema con las cuales se maximiza la respuesta del mismo (18). En este trabajo se utilizó un modelo cuadrático (Ecuación [2]) de regresión polinomial para modelar la conversión del monóxido de carbono como una función matemática y analizar la interacción entre las variables independientes (diámetro y longitud de los pellets) del diseño experimental.

Donde y es la respuesta que corresponde a la conversión del monóxido de carbono, β0 es el intercepto, βi es el coeficiente lineal, βii es el coeficiente cuadrático de cada variable independiente, βij es el coeficiente de interacción entre las variables, i y j son los índices para las variables x1 (longitud) y x2 (diámetro) y ε es el error aleatorio que muestra las diferentes fuentes de variabilidad (19), con los supuestos usuales de normalidad, independencia y homogeneidad de varianza. La adecuación del modelo de regresión de segundo orden se evaluó teniendo en cuenta: el coeficiente de determinación (R2), la significancia del modelo de regresión mediante el valor de la prueba F, el análisis de varianza ANOVA, el coeficiente de variación CV, la desviación media absoluta (AAD) (20) y la verificación de todos los supuestos del modelo. Adicionalmente, se presentan los gráficos de la superficie de respuesta y de contorno, para percibir la naturaleza de la superficie de respuesta.
Preparación de los pellets de Cu-Ni:2-1/AC
Los pellets se prepararon de acuerdo a la metodología previamente reportada (21) que consistió en el siguiente procedimiento: se mezcló carbón activado comercial (AC) Merck (90% de pureza, tamaño de partícula <100 μm) con una solución etanólica de carboximetilcelulosa (10% p/p) como aglutinante. La solución se agitó y se calentó suavemente durante 24 h hasta la evaporación del etanol. El sólido restante se humedeció con suficiente agua para formar una pasta, que se presionó en un sistema hidráulico uniaxial a una tonelada métrica durante 15 min. El carbón compactado se extruyó luego en forma cilíndrica y se cortó en pellets de varios diámetros (0,2 cm; 0,3 cm y 0,4 cm) y longitudes (0,4 cm y 0,6 cm). Después de secar a 80 °C durante 12 h, los pellets se pirolizaron durante 1 h a 700 °C con 25 mL·min-1 de N2 a una velocidad de calentamiento de 20 °C·min-1. Los catalizadores bimetálicos Cu-Ni en relación molar 2-1 se prepararon mediante impregnación húmeda de los pellets obtenidos.
Por otro lado, la metodología utilizada para la impregnación de los pellets se basa en el trabajo de Arbeláez y colaboradores (22). Se utilizó Cu(NO3)2.3H2O (Carlo Erba, España, 99,5%) y Ni(NO3)2.6H2O (Merck, Estados Unidos, 99%) como precursores metálicos con una carga nominal de óxidos metálicos (CuO + NiO) del 20% p/p. Los precursores disueltos en hidróxido de amonio fueron mezclados durante 15 min y adicionados a los pellets de carbón activado. Se agitó durante 12 h, luego se rotoevaporó a 25 rpm y 180 mm Hg de vacío, los pellets impregnados se secaron a 90 °C durante 12 h y se pirolizaron en N2 (25 mL·min-1) a 0,5 °C·min-1 hasta 500 °C, temperatura que se mantuvo durante 3 h. Todas las muestras se almacenaron en un disecador bajo atmósfera inerte antes de usarlos.
Caracterización de los catalizadores
Los pellets se caracterizaron por análisis químico elemental y área superficial, para comprobar que las diferencias en actividad se debieran a la diferencia de tamaño y no al contenido de los metales.
Análisis químico
Esta técnica se empleó para determinar el contenido de Cu y Ni en los catalizadores preparados. Se usó un espectrómetro Thermo Electron Corporation, modelo S4, utilizando rodio como fuente de radiación operando a 40 kV. Antes del análisis las muestras (0,05 g) se sometieron a digestión en 7,5 mL de HCl y 2,5 mL de HNO3, aforándose con agua destilada en un balón de 100 mL.
Área superficial
El área superficial específica se determinó mediante adsorción de N2 usando un Autochem II 2920 (Micromeritics). Antes del análisis, las muestras se sometieron a desgasificación a 250 °C durante 0,5 h en una corriente de N2/He con una velocidad de calentamiento de 0,5 °C·min-1.
Evaluación de la actividad catalítica
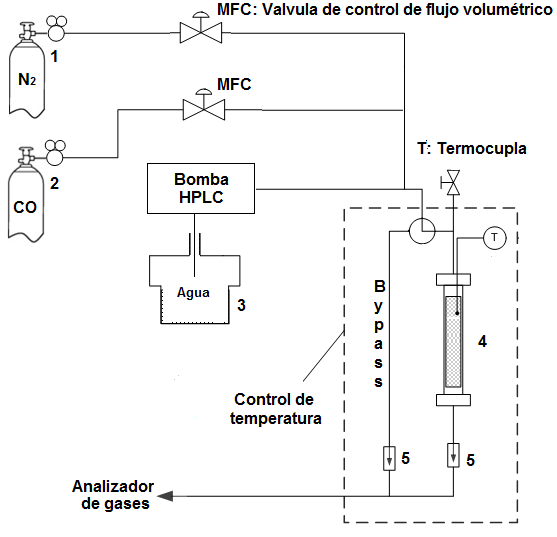
Los ensayos de la actividad se llevaron a cabo utilizando el sistema de reacción mostrado en la Figura 1. Para esto se ubicaron alrededor de 1,5 cm3 (aproximadamente 250 mg) de catalizador pelletizado en un reactor tubular de flujo continuo de acero inoxidable (0,75 cm diámetro interno), operado a presión atmosférica con variación de la temperatura entre 180 y 350 °C, usando una mezcla modelo (4,5% CO; 30,5% H2O; 65% N2). El agua se introdujo al sistema en forma gaseosa utilizando una bomba de HPLC; el agua se vaporizó antes de entrar en el reactor. Antes de las mediciones catalíticas, las muestras se redujeron in situ, con un flujo de 120 mL·min-1 de H2/N2 (10% H2) a 10 ºC·min-1 hasta 400 ºC; esta temperatura se mantuvo durante 2 h.
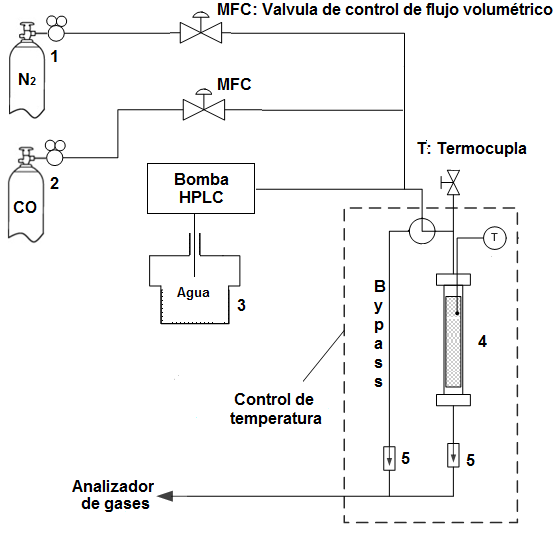
Figura 1
Montaje experimental para la evaluación de la actividad catalítica. Cilindro de N2, (2) cilindro de CO, (3) recipiente con agua (4), reactor tubular de lecho fijo, (5) válvula cheque. MFC válvula de control de flujo volumétrico. Condiciones de análisis: 1 bar, volumen de catalizador: 1,5 cm3; 120 mL·min-1
Los contenidos de CO, CO2 y agua se cuantificaron en línea con un analizador de gases ABB y la actividad se expresó en términos de la conversión del monóxido de carbono (Ecuación [3])

Donde COin y COout son las concentraciones de dióxido de carbono a la entrada y a la salida del reactor, respectivamente
Resultados y discusión
Análisis de Varianza y prueba estadística
En el proceso se detectaron dos observaciones atípicas (observaciones 13 y 14) las cuales influyen de manera importante en la violación del supuesto de homogeneidad de varianza y normalidad. En la literatura se sugiere el uso de métodos de estimación ponderada, una alternativa para no eliminar estas observaciones y no optar por métodos menos robustos (no paramétricos) (23). En este análisis se usó una ponderación de 10-2 para las dos observaciones atípicas y de una unidad para el resto. Los resultados del análisis de varianza, incluyendo sólo los términos significativos, se muestran en la Tabla 2.
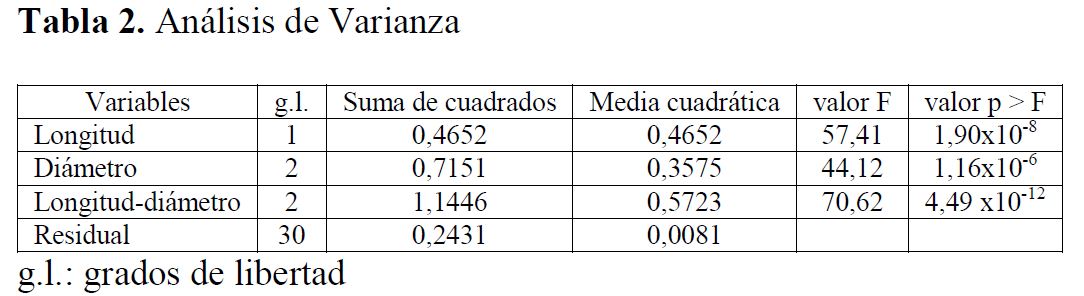
Análisis de Varianza
g.l.: grados de libertadEn la Tabla 2 se presentan los valores del estadístico F (cociente entre las medias cuadráticas del respectivo factor y el residual), el cual se usa para probar la significancia de las variables individuales y sus interacciones sobre la variable de respuesta. Se verificaron todos los supuestos del modelo, independencia, normalidad y homogeneidad de varianza. Se observa que, para la longitud, el diámetro y la combinación entre longitud y diámetro, los valores del estadístico F fueron 57,41; 44,12 y 70,62; respectivamente. Teniendo en cuenta los valores p de la Tabla 2, se concluye que tanto la longitud como el diámetro de los pellets son altamente significativos sobre la conversión del monóxido de carbono. El efecto combinado entre el diámetro y la longitud arrojó un valor F de 70,62 (el valor p más bajo), lo cual indica que la interacción entre estas dos variables es altamente significativa.
Un modelo es considerado significativo cuando el valor p es menor que 0,05 y los valores p que exceden 0,100 no juegan un papel importante en el modelo. Por tanto, en el caso de la conversión de monóxido de carbono, la longitud y el diámetro de los pellets son estadísticamente significativos. A partir del modelo anterior se consideró un modelo de regresión cuadrático en el diámetro y lineal en los factores, este modelo se presenta en la siguiente sección.
Modelo estadístico
El análisis de varianza realizado previo al modelo, permite predecir que la ecuación de regresión de los datos experimentales debe incluir tanto el efecto lineal del diámetro y la longitud, y un efecto cuadrático en el diámetro, así como el efecto combinado de cada una de estas variables. El modelo de regresión resultante, que predice la conversión (en escala logarítmica), a partir de una dependencia lineal tanto de la longitud x1, como del diámetro x2, una relación cuadrática del diámetro y la interacción entre las dos variables se presenta en la Ecuación [4].

Todos los parámetros asociados al modelo ajustado de [4] fueron significativos. El diámetro y la longitud de los pellets con coeficientes 0,14033 y 0,11762, respectivamente, tienen un efecto positivo sobre el logaritmo de la conversión del monóxido de carbono. Del modelo se puede inferir que los pellets con mayor diámetro y longitud son los más activos en la reacción. Igualmente, se observa que la interacción entre estas dos variables tiene un efecto negativo (-0,202) sobre la conversión del monóxido de carbono, la cual decrece cuando disminuye tanto el diámetro como la longitud, parámetros relacionados con la porosidad y el tiempo de retención de los reactivos en el lecho catalítico.
Para un lecho catalítico empacado, la adsorción se debe evaluar en función del volumen, porque los huecos donde no se produce la adsorción se forman entre las partículas como resultado del empaquetamiento, siendo mayor a menor diámetro y longitud, afectando la densidad aparente del sistema catalítico. La adecuación del modelo ajustado se corroboró con el valor del coeficiente de correlación R2 = 0,941, una medida de la variación alrededor de la media explicada por el modelo considerado, asegurando un buen ajuste a los datos experimentales.
El modelo matemático para la conversión del monóxido de carbono se utilizó para construir la superficie de respuesta (Figura 2) y las gráficas de contorno (Figura 3) las cuales son útiles para visualizar la dependencia y la interacción de cada una de las variables.
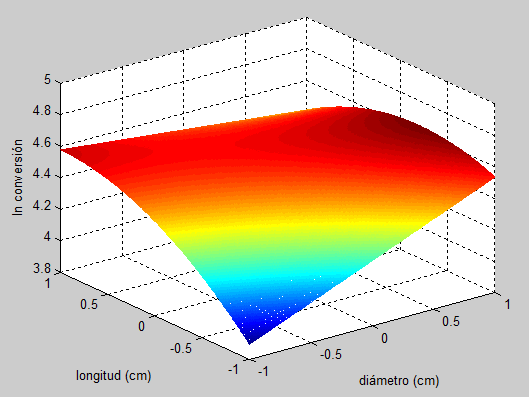
Figura 2.
Superficie de respuesta del modelo
La Figura 2 muestra que la región de mínima conversión de monóxido de carbono se localizó en la región de mínimo diámetro y longitud de los pellets, también se observa que el efecto de un aumento en el diámetro es mayor que un aumento en la longitud. Este efecto está relacionado con la disminución de la densidad aparente y el aumento de la porosidad del lecho. Los resultados muestran que los pellets de 0,4 cm de diámetro y 0,6 cm de longitud son los más activos en la reacción de desplazamiento de agua dando una conversión de 96%. Adicionalmente, el gráfico de contornos (Figura 3) reveló que existen interacciones significativas entre el diámetro y la longitud. Sin embargo, no se observa ni un punto de máxima, ni de mínima en la conversión del monóxido de carbono, aunque se comprueba que un aumento de la longitud y del diámetro mejora la conversión del monóxido de carbono.
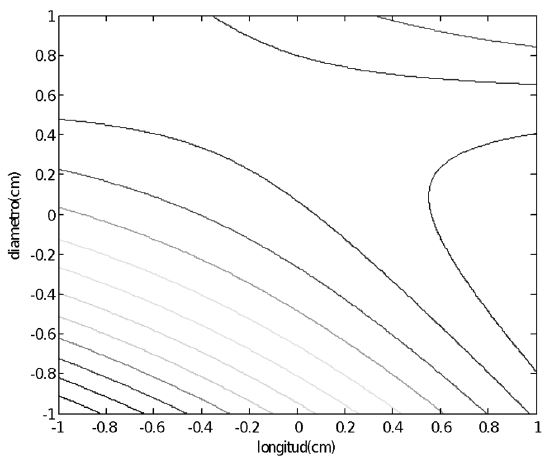
Figura 3.
Gráfico de contornos
Caracterización de los catalizadores
La Tabla 3 muestra los resultados del área superficial de los pellets evaluados. Estos resultados muestran que el área superficial de los pellets fue aproximadamente igual, variando entre 402 y 442 m2·min-1, por lo cual la diferencia en actividad catalítica de los pellets no estaría asociada al área superficial de los mismos. Comparada con el área superficial del carbón activado (891 m2·min-1), la de los pellets disminuyó entre 55 y 50%. Esta diferencia en área superficial sería consecuencia de la incorporación de los metales y las altas temperaturas de calcinación y reducción (600°C) a las que se someten las muestras.

Área superficial de los catalizadores evaluados
D: diámetro-L: longitudAnálisis químico
Los resultados del contenido de Cu y Ni se muestran en la Tabla 4. Allí se observa que la carga de metales obtenida por espectroscopía de absorción atómica es menor a la carga nominal (20% p/p CuO + NiO), probablemente debido a pérdidas por lixiviación de los precursores, Cu(NO3)2·3H2O y Ni(NO3)2·6H2O durante la preparación del catalizador por impregnación húmeda. Sin embargo, no es posible descartar posibles interferencias del soporte (carbón activado) en la espectroscopía de absorción atómica (24). Aunque la relación molar Cu-Ni real de los catalizadores preparados no es igual a la relación nominal (Cu-Ni = 2-1), varía en un rango muy estrecho (1,8-1 a 2,3-1). Esto permite concluir que la diferencia en la actividad catalítica se debe principalmente al tamaño de los pellets y no al contenido de Cu y Ni en los mismos.

D-L: diámetro- longitud
Resultados de la actividad catalítica
La Figura 4 muestra la conversión de monóxido de carbono en la reacción de desplazamiento de gas de agua sobre los catalizadores peletizados de Cu-Ni en función de la temperatura entre 180 y 350 °C.
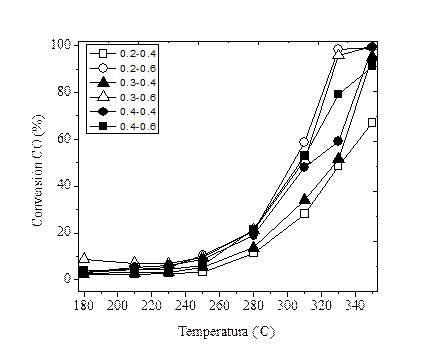
Figura 4
Conversión del CO en función de la temperatura para diferentes valores de diámetro-longitud de los pellets
Los resultados de la Figura 4 muestran que la actividad catalítica es afectada significativamente por la temperatura en el rango 180-250 °C. Lo anterior concuerda con los datos previamente reportados por Lin y colaboradores (25), Chen y colaboradores (9) y Arbeláez y colaboradores (10), quienes encontraron conversiones del monóxido de carbono menores al 20% para temperaturas inferiores a 240 °C. A temperaturas superiores, la conversión de CO aumentó con la temperatura, se obtienen conversiones de CO alrededor del 95% a 350 °C; el catalizador con el menor diámetro y longitud mostró una conversión de CO alrededor del 65%. Sin embargo, como los pellets usados tienen diferente tamaño (diámetro y longitud) pero conservan un volumen empacado de 1,5 cm3 y el mismo peso total del lecho catalítico empacado a través del cual fluyen los reactivos, la evaluación del modelo estadístico permitió correlacionar la interacción favorable entre estas dos variables del sistema y proponer el modelo más adecuado para estimar la conversión del monóxido de carbono en términos del diámetro y la longitud.
Conclusiones
Se desarrolló un modelo estadístico para obtener las condiciones más adecuadas en la reacción de desplazamiento de agua a partir de catalizadores bimetálicos de Cu-Ni:2-1 en forma de pellets cilíndricos de carbón activado. Un diseño factorial con dos factores indicó que tanto la longitud como el diámetro de los pellets influyen en la conversión del monóxido de carbono. Además se observó que el efecto principal de cada variable fue muy significativo al igual que el respectivo efecto combinado. Se encontró una mayor conversión del monóxido a mayor diámetro y longitud de los pellets: con los pellets de 0,4 cm de diámetro y 0,6 cm de longitud se obtuvo una mayor conversión de monóxido de carbono en la reacción de desplazamiento de agua.
Agradecimientos
Los autores agradecen a la Universidad Cooperativa de Colombia por el apoyo financiero a través de proyecto 1539, a la Universidad de Antioquia y a la Universidad Nacional de Colombia.
Referencias
1. Dou, B.; Song, Y.; Wang, C.; Chen, H.; Xu, Y. Hydrogen production from catalytic steam reforming of biodiesel byproduct glycerol: Issues and challenges. Renew Sustain Energy Rev 2014, 30 950–60. DOI: https://doi.org/10.1016/j.rser.2013.11.029.
2. Lucia, U.; Overview on fuel cells. Renew Sustain Energy Rev 2014, 30, 164–9. DOI: https://doi.org/10.1016/j.rser.2013.09.025.
3. Jain, R.; Maric, R. Synthesis of nano-Pt onto ceria support as catalyst for water–gas shift reaction by Reactive Spray Deposition Technology. Appl Catal A Gen 2014 475, 461–8. DOI: https://doi.org/10.1016/j.apcata.2014.01.053.
4. Panagiotopoulou, P.; Kondarides, D. Effects of promotion of TiO2 with alkaline earth metals on the chemisorptive properties and water–gas shift activity of supported platinum catalysts. Appl Catal B Environ 2011 101, 738–46. DOI: https://doi.org/10.1016/j.apcatb.2010.11.016.
5. Andreeva, D.; Idakiev, V.; Tabakova, T.; Ilieva, L.; Falaras, P.; Bourlinos A, et al. Low-temperature water-gas shift reaction over Au/CeO2 catalysts. Catal. Today 2002, 72, 51–7. DOI: https://doi.org/10.1016/S0920-5861(01)00477-1.
6. Rodriguez, J.; Gold-based catalysts for the water–gas shift reaction: Active sites and reaction mechanism. Catal Today 2011, 160, 3–10. DOI: https://doi.org/10.1016/j.cattod.2010.06.030.
7. Karakaya, C.; Otterstätter, R.; Maier, L.; Deutschmann, O. Kinetics of the water-gas shift reaction over Rh/Al2O3 catalysts. Appl Catal A Gen 2014, 470, 31–44. DOI: https://doi.org/10.1016/j.apcata.2013.10.030.
8. Goscianska, J.; Ziolek, M.; Gibson, E.; Daturi, M. Novel mesoporous zirconia-based catalysts for WGS reaction. Appl Catal B Environ 2010, 97, 49–56. DOI: https://doi.org/10.1016/j.apcatb.2010.03.023.
9. Chen, C.; Ruan, C.; Zhan, Y.; Lin, X.; Zheng, Q.; Wei, K. The significant role of oxygen vacancy in Cu/ZrO2 catalyst for enhancing water–gas-shift performance. Int J Hydrogen Energy 2014, 39, 317–24. DOI: https://doi.org/10.1016/j.ijhydene.2013.10.074.
10. Arbeláez, O.; Reina, T.; Ivanova, S.; Bustamante, F.; Villa, A.; Centeno, M.; Odriozola, J.; Mono and bimetallic Cu-Ni structured catalysts for the water gas shift reaction. Appl Catal A Gen 2015, 497, 1–9. DOI: https://doi.org/10.1016/j.apcata.2015.02.041.
11. Lin, J.; Guliants, V.; Alumina-supported Cu-Ni and Ni-Cu core–shell nanoparticles: Synthesis, characterization, and catalytic activity in water–gas-shift reaction. Appl Catal A Gen 2012, 445-446, 187–94. DOI: https://doi.org/10.1016/j.apcata.2012.08.013.
12. Saw, E.; Oemar, U.; Tan, X.; Du, Y.; Borgna, A.; Hidajat, K.; et al. Bimetallic Ni–Cu catalyst supported on CeO2 for high-temperature water–gas shift reaction: Methane suppression via enhanced CO adsorption. J Catal 2014, 314, 32–46. DOI: https://doi.org/10.1016/j.jcat.2014.03.015.
13. Rad, A.; Khoshgouei, M.; Rezvani, S.; Rezvani, A. Study of Cu–Ni/SiO2 catalyst prepared from a novel precursor, [Cu(H2O)6][Ni(dipic)2].2H2O/SiO2, for water gas shift reaction. Fuel Process Technol 2012, 96, 9–15. DOI: https://doi.org/10.1016/j.fuproc.2011.12.015.
14. Zhang, X.; Zhang, Y.; Liu, Q.; Zhou, W. Surface properties of activated carbon from different raw materials. Int J Min Sci Technol 2012, 22, 483–6. DOI: https://doi.org/10.1016/j.ijmst.2012.01.007.
15. Akhtar, F.; Andersson, L.; Ogunwumi, S.; Hedin, N.; Bergström, L. Structuring adsorbents and catalysts by processing of porous powders. J Eur Ceram Soc 2014, 34, 1643–66. DOI: https://doi.org/10.1016/j.jeurceramsoc.2014.01.008.
16. Bruner, E.; Puri, M. L. Nonparametric methods in factorial designs. Statistical papers. 2001, 42, 1-52.
17. R Core Team. (2015). R: A language and environment for statistical computing (Programa informático). Vienna: R Foundation for Statistical Computing. http://www.r-project.org/ 2015:2015 (consultado el 13 de abril de 2017)
18. Zarei, A.; Seyed, S.; Rezaei, M.; Meshkani, F. A comparative study of experimental investigation and response surface optimization of steam reforming of glycerol over nickel nano-catalysts. Int J Hydrogen Energy 2016, 41, 10178–92. DOI: https://doi.org/10.1016/j.ijhydene.2016.05.047.
19. Moradi, M.; Daryan, J.; Mohamadalizadeh, A. Response surface modeling of H2S conversion by catalytic oxidation reaction over catalysts based on SiC nanoparticles using Box−Behnken experimental design. Fuel Process Technol 2013, 109, 163–71. DOI: https://doi.org/10.1016/j.fuproc.2012.10.013.
20. Tounsadi, H.; Khalidi, A.; Farnane, M.; Abdennouri, M.; Barka, N. Experimental design for the optimization of preparation conditions of highly efficient activated carbon from Glebionis coronaria L. and heavy metals removal ability. Process Saf Environ Prot 2016, 102, 710–23. DOI: https://doi.org/10.1016/j.psep.2016.05.017.
21. Orrego, A.; Arbeláez, O.; Bustamante, F.; Villa, A H. Pelletization of catalysts supported on activated carbon. A Case Study : clean synthesis of dimethyl carbonate from methanol and CO2 Revista Facultad de Ingeniería 2016, 78, 38–47. DOI: https://doi.org/10.17533/udea.redin.n78a05.
22. Arbeláez, O.; Orrego, A.; Bustamante, F.; Villa, A. Direct Synthesis of Diethyl Carbonate from CO2 and CH3CH2OH over Cu–Ni/AC Catalyst. Top Catal 2012, 55, 668–72. DOI: https://doi.org/10.1007/s11244-012-9849-4.
23. Montgomery, D. Diseño y análisis de experimentos, 2nd ed. Limusa Wesley: Mexico, 2002.
24. Welz, B.; Vale, M.; Pereira, E.; Castilho, I.; Dessuy, M. Continuum source atomic absorption spectrometry: past, present and future aspects – a critical review. J. Braz. Chem. Soc. 2012, 25, 799-821 DOI: https://doi.org/10.5935/0103-5053.20140053.
25. Lin, J.; Biswas, P.; Guliants, V.; V. Misture, S. Hydrogen production by water–gas shift reaction over bimetallic Cu–Ni catalysts supported on La-doped mesoporous ceria. Appl Catal A Gen 2010, 387, 87–94. DOI: https://doi.org/10.1016/j.apcata.2010.08.003.
Enlace alternativo
https://revistas.unal.edu.co/index.php/rcolquim/article/view/64699 (html)
Recibido: 8 de mayo de 2017; Aceptado: 29 de septiembre de 2017
Resumen
Se sintetizaron catalizadores bimetálicos de Cu-Ni en relación molar 2-1 en pellets cilíndricos de carbón activado de diferentes diámetros (0,2 cm; 0,3 cm y 0,4 cm) y longitud (0,4 cm y 0,6 cm); se evaluaron en la reacción de desplazamiento de agua a 330 °C y presión atmosférica. Se realizó un diseño experimental para evaluar el efecto de las variables, diámetro y longitud, sobre la conversión del monóxido de carbono. La importancia de los principales factores y sus interacciones se examinaron por medio del análisis de varianza (ANOVA). Los resultados mostraron que tanto el diámetro como la longitud afectan significativamente la conversión del monóxido de carbono. Los resultados de la actividad catalítica, bajo las mejores condiciones de diámetro (0,4 cm) y longitud (0,6 cm) de los pellets, mostraron una conversión del monóxido de carbono del 96%.
Palabras clave
Reacción de desplazamiento de agua, pellets de Cu-Ni, diseño experimental, conversión de monóxido de carbono.Abstract
Bimetallic Cu-Ni catalysts were synthesized with molar ratio 2-1 in cylindrical activated carbon pellets of different diameters (0.2 cm, 0.3 cm, and 0.4 cm) and length (0.4 cm and 0.6 cm). They were evaluated in the water gas shift reaction at 330 °C and atmospheric pressure. An experimental design was developed to evaluate the effect of variables, diameter and length of the pellets, over carbon monoxide conversion. The importance of the main factors and their interactions were examined by analysis of variance (ANOVA). The results showed that both diameter and length of the pellets had significant effect on carbon monoxide conversion. The results of the catalytic activity indicated that up to 96% CO conversion was obtained at the optimum characteristic of the pellets, diameter of 0.2 cm and length of 0.6 cm.
Keywords
water gas shift reaction, pellets of Cu-Ni, experimental design, carbon monoxide conversion.Resumo
Foram sintetizados catalisadores bimetálicos de Cu-Ni em proporção molar 2-1 sobre pastilhas de carvão ativado de diferentes diâmetros (0,2 cm; 0,3 cm e 0,4 cm) e comprimentos (0,4 cm e 0,6 cm) e foram avaliados na reação de mudança de vapor de agua a 330 °C e pressão atmosférica. Um desenho experimental foi desenvolvido para avaliar o efeito de variáveis, diâmetro e comprimento, na conversão de monóxido de carbono. A importância dos principais fatores e as suas interações foram examinados por análise de variância (ANOVA), os resultados mostraram que tanto o diâmetro e o comprimento afetam significativamente a conversão de monóxido de carbono. Os resultados da atividade catalítica com as melhores condições de diâmetro (0,4 cm) e o comprimento (0,6 cm) das pastilhas mostrou uma conversão de monóxido de carbono de 96%.
Palavras-chave
reação de deslocamento do gás de água, grânulos de Cu-Ni, desenho experimental, conversão de monóxido de carbono.Introducción
La reacción de desplazamiento de agua, Ecuación [1], conocida como water gas shift (WGS) es un proceso industrial que permite la conversión del monóxido de carbono y agua en dióxido de carbono e hidrógeno, además de su uso en la mezcla de gases de gran importancia en la síntesis de amoníaco y metanol (1), en procesos de refinería de hidrocarburos y en aplicaciones en las celdas de combustible, siendo estas últimas ampliamente estudiadas, dado su elevado potencial energético y la baja emisión de gases de efecto invernadero (2).
Sin embargo, uno de los principales inconvenientes de esta reacción es la limitación termodinámica del equilibrio. Dado su carácter exotérmico, ΔH0 = -41,2 kJ·mol-1, la conversión del monóxido de carbono y la producción de hidrógeno se favorecen a bajas temperaturas. La reacción se ha realizado en presencia de catalizadores heterogéneos que han permitido mejorar la conversión del monóxido de carbono y la selectividad hacia el hidrógeno.
Entre los catalizadores comúnmente utilizados se destacan Pt/CeO2 (3), Pt/TiO2 (4), Au/CeO2 (5), Au/TiO2 (6) y Rh/Al2O3 (7) aunque, debido al área superficial relativamente baja (50-400 m2·g-1), los elevados costos, la desactivación a temperaturas altas por sinterización del metal y la rápida desactivación de los mismos, bajo los ciclos de encendido y apagado frecuente (8), se motiva la sustitución de estos catalizadores por otros que contengan metales de menor costo, manteniendo el rendimiento catalítico del sistema.
En comparación con los metales nobles soportados, los catalizadores de Cu y Cu-Ni tales como Cu/ZrO2 (9), Cu-Ni/carbón activado (10), Cu-Ni/Al2O3 (11), Cu-Ni/CeO2 (12), Cu-Ni/SiO2 (13), han sido reportados como una alternativa prometedora en la reacción de desplazamiento de agua. Entre los diferentes soportes, el carbón activado es un material de elevada área superficial, económico, altamente disponible, y con adecuada resistencia mecánica y térmica (14). En un trabajo anterior (10) se reportó la preparación de catalizadores mono y bimetálicos Cu-Ni a partir de pellets cilíndricos (3 mm de diámetro y 4 mm de longitud) de carbón activado y su actividad en la reacción de desplazamiento de agua. Bajo las condiciones experimentales reportadas (mezcla modelo de reacción: 4,5% de CO; 30,5% de H2O, 65% de N2), se encontró que, a temperaturas mayores de 220 °C, la conversión del CO aumenta con la temperatura, siendo el catalizador bimetálico Cu-Ni en relación molar 2-1 el de mayor actividad catalítica. Específicamente, a 330 °C se obtuvo una conversión de CO del 99% y una selectividad del 98% hacia el hidrógeno.
Los análisis de difracción de rayos X del catalizador mostraron líneas de difracción con posiciones intermedias a las señales de Cu y Ni puros, lo cual evidenció la formación de la solución sólida Cu-Ni. Adicionalmente, el aumento del parámetro de red con el aumento de la concentración de Cu se ajustó a la Ley de Vegard, confirmando la formación de una solución sólida entre los dos metales, relacionada con la mayor actividad catalítica de este catalizador, en comparación con los materiales preparados con otras relaciones de Cu-Ni.
Estudios adicionales de espectroscopía fotoelectrónica de Rayos X (XPS) mostraron que el catalizador bimetálico Cu-Ni, en relación molar 2-1, exhibió señales de Cu2p3/2, Cu2p1/2, Ni 2p3/2 y Ni2p1/2 de 933,2; 953,1; 858,5 y 876,6; respectivamente. Estas se encuentran a una mayor energía de enlace que las respectivas señales de Cu2p3/2 (932,8 eV), Cu2p1/2 (952,8 eV), Ni 2p3/2 856,7 eV) y Ni2p1/2 (874,6 eV) de los catalizadores monometálicos de Cu y Ni, y se ha reportado como una evidencia de la formación de la solución sólida Cu-Ni. Adicionalmente, este mismo catalizador, mantuvo una conversión de CO estable (alrededor del 50%) durante 20 h de análisis continuo bajo condiciones reales de mezcla de reacción (9% CO; 30,5% H2O; 11% CO2; 50% H2; 1,5% N2) a 330 ºC.
Para catalizadores peletizados se ha determinado que la actividad catalítica está controlada por el diámetro y longitud de los mismos, los cuales afectan el volumen de los espacios vacíos que se forman durante el empaquetamiento. Del mismo modo, está directamente relacionado con la caída de presión y las limitaciones difusionales externas e internas del lecho catalítico (15).
En este artículo se reportan los resultados de un diseño experimental para estudiar el efecto de la longitud y el diámetro de catalizadores pelletizados Cu-Ni sobre la conversión de CO en la reacción de desplazamiento de agua. Para investigar las condiciones más adecuadas de diámetro y longitud de los catalizadores y establecer la conversión máxima de CO, se ajustó un modelo polinominal de segundo orden, se verificaron todos sus supuestos y se obtuvo la superficie de respuesta con la que se identificó la influencia de dichas variables en la actividad catalítica del sistema en términos de la conversión del CO.
Ahora bien, los resultados de diseño experimental se pueden analizar a partir de un enfoque paramétrico, conocido como análisis ANOVA, donde se debe cumplir el supuesto de normalidad, varianza constante e independencia en el término de error. También se pueden analizar mediante enfoques no paramétricos, por ejemplo el método de Kruscal-Wallis, cuando no es posible validar el supuesto de normalidad del término de error (16). Debido a lo anterior, se optó por el primer enfoque ya que con el modelo bajo consideración se validaron todos los supuestos del modelo.
Materiales y métodos
Diseño experimental y análisis estadístico
La determinación de las condiciones que maximizarían la conversión de CO, en el rango de trabajo de las variables estudiadas, se llevó a cabo en dos etapas: 1) la formulación del diseño de experimentos, y 2) el ajuste de un modelo polinomial a los datos experimentales para determinar el valor de los factores (diámetro y longitud) que satisficieran las condiciones más adecuadas.
Diseño de experimentos
El diseño de experimentos tiene por objeto reducir el número de pruebas experimentales, incluyendo diversas combinaciones que permitan estimar los efectos sinérgicos y antagonistas de la interacción entre el diámetro y la longitud del catalizador sobre la conversión del monóxido de carbono. Las variables del proceso fueron codificadas como +1, -1 y 0, las cuales corresponden a un valor alto, bajo y un punto central, respectivamente. El programa R (Project for statistical computing) versión 3.2.5 de análisis estadístico (17), se utilizó para generar el diseño experimental y analizar los datos obtenidos. Los experimentos se realizaron de acuerdo con un diseño de dos factores AxB con seis réplicas (experimentos). El número total de experimentos, los niveles examinados y los rangos experimentales de cada variable independiente se presentan en la Tabla 1 .
x1:
longitud, x2: diámetroTabla 1
Ajuste del modelo
Un modelo polinomial permite determinar el valor más adecuado de las variables del sistema con las cuales se maximiza la respuesta del mismo (18). En este trabajo se utilizó un modelo cuadrático (Ecuación [2]) de regresión polinomial para modelar la conversión del monóxido de carbono como una función matemática y analizar la interacción entre las variables independientes (diámetro y longitud de los pellets) del diseño experimental.
Donde y es la respuesta que corresponde a la conversión del monóxido de carbono, β0 es el intercepto, βi es el coeficiente lineal, βii es el coeficiente cuadrático de cada variable independiente, βij es el coeficiente de interacción entre las variables, i y j son los índices para las variables x1 (longitud) y x2 (diámetro) y ε es el error aleatorio que muestra las diferentes fuentes de variabilidad (19), con los supuestos usuales de normalidad, independencia y homogeneidad de varianza. La adecuación del modelo de regresión de segundo orden se evaluó teniendo en cuenta: el coeficiente de determinación (R2), la significancia del modelo de regresión mediante el valor de la prueba F, el análisis de varianza ANOVA, el coeficiente de variación CV, la desviación media absoluta (AAD) (20) y la verificación de todos los supuestos del modelo. Adicionalmente, se presentan los gráficos de la superficie de respuesta y de contorno, para percibir la naturaleza de la superficie de respuesta.
Preparación de los pellets de Cu-Ni:2-1/AC
Los pellets se prepararon de acuerdo a la metodología previamente reportada (21) que consistió en el siguiente procedimiento: se mezcló carbón activado comercial (AC) Merck (90% de pureza, tamaño de partícula <100 μm) con una solución etanólica de carboximetilcelulosa (10% p/p) como aglutinante. La solución se agitó y se calentó suavemente durante 24 h hasta la evaporación del etanol. El sólido restante se humedeció con suficiente agua para formar una pasta, que se presionó en un sistema hidráulico uniaxial a una tonelada métrica durante 15 min. El carbón compactado se extruyó luego en forma cilíndrica y se cortó en pellets de varios diámetros (0,2 cm; 0,3 cm y 0,4 cm) y longitudes (0,4 cm y 0,6 cm). Después de secar a 80 °C durante 12 h, los pellets se pirolizaron durante 1 h a 700 °C con 25 mL·min-1 de N2 a una velocidad de calentamiento de 20 °C·min-1. Los catalizadores bimetálicos Cu-Ni en relación molar 2-1 se prepararon mediante impregnación húmeda de los pellets obtenidos.
Por otro lado, la metodología utilizada para la impregnación de los pellets se basa en el trabajo de Arbeláez y colaboradores (22). Se utilizó Cu(NO3)2.3H2O (Carlo Erba, España, 99,5%) y Ni(NO3)2.6H2O (Merck, Estados Unidos, 99%) como precursores metálicos con una carga nominal de óxidos metálicos (CuO + NiO) del 20% p/p. Los precursores disueltos en hidróxido de amonio fueron mezclados durante 15 min y adicionados a los pellets de carbón activado. Se agitó durante 12 h, luego se rotoevaporó a 25 rpm y 180 mm Hg de vacío, los pellets impregnados se secaron a 90 °C durante 12 h y se pirolizaron en N2 (25 mL·min-1) a 0,5 °C·min-1 hasta 500 °C, temperatura que se mantuvo durante 3 h. Todas las muestras se almacenaron en un disecador bajo atmósfera inerte antes de usarlos.
Caracterización de los catalizadores
Los pellets se caracterizaron por análisis químico elemental y área superficial, para comprobar que las diferencias en actividad se debieran a la diferencia de tamaño y no al contenido de los metales.
Análisis químico
Esta técnica se empleó para determinar el contenido de Cu y Ni en los catalizadores preparados. Se usó un espectrómetro Thermo Electron Corporation, modelo S4, utilizando rodio como fuente de radiación operando a 40 kV. Antes del análisis las muestras (0,05 g) se sometieron a digestión en 7,5 mL de HCl y 2,5 mL de HNO3, aforándose con agua destilada en un balón de 100 mL.
Área superficial
El área superficial específica se determinó mediante adsorción de N2 usando un Autochem II 2920 (Micromeritics). Antes del análisis, las muestras se sometieron a desgasificación a 250 °C durante 0,5 h en una corriente de N2/He con una velocidad de calentamiento de 0,5 °C·min-1.
Evaluación de la actividad catalítica
Los ensayos de la actividad se llevaron a cabo utilizando el sistema de reacción mostrado en la Figura 1. Para esto se ubicaron alrededor de 1,5 cm3 (aproximadamente 250 mg) de catalizador pelletizado en un reactor tubular de flujo continuo de acero inoxidable (0,75 cm diámetro interno), operado a presión atmosférica con variación de la temperatura entre 180 y 350 °C, usando una mezcla modelo (4,5% CO; 30,5% H2O; 65% N2). El agua se introdujo al sistema en forma gaseosa utilizando una bomba de HPLC; el agua se vaporizó antes de entrar en el reactor. Antes de las mediciones catalíticas, las muestras se redujeron in situ, con un flujo de 120 mL·min-1 de H2/N2 (10% H2) a 10 ºC·min-1 hasta 400 ºC; esta temperatura se mantuvo durante 2 h.
Figura 1
Los contenidos de CO, CO2 y agua se cuantificaron en línea con un analizador de gases ABB y la actividad se expresó en términos de la conversión del monóxido de carbono (Ecuación [3])
Donde COin y COout son las concentraciones de dióxido de carbono a la entrada y a la salida del reactor, respectivamente
Resultados y discusión
Análisis de Varianza y prueba estadística
En el proceso se detectaron dos observaciones atípicas (observaciones 13 y 14) las cuales influyen de manera importante en la violación del supuesto de homogeneidad de varianza y normalidad. En la literatura se sugiere el uso de métodos de estimación ponderada, una alternativa para no eliminar estas observaciones y no optar por métodos menos robustos (no paramétricos) (23). En este análisis se usó una ponderación de 10-2 para las dos observaciones atípicas y de una unidad para el resto. Los resultados del análisis de varianza, incluyendo sólo los términos significativos, se muestran en la Tabla 2.
g.l.: grados de libertadTabla 2.
En la Tabla 2 se presentan los valores del estadístico F (cociente entre las medias cuadráticas del respectivo factor y el residual), el cual se usa para probar la significancia de las variables individuales y sus interacciones sobre la variable de respuesta. Se verificaron todos los supuestos del modelo, independencia, normalidad y homogeneidad de varianza. Se observa que, para la longitud, el diámetro y la combinación entre longitud y diámetro, los valores del estadístico F fueron 57,41; 44,12 y 70,62; respectivamente. Teniendo en cuenta los valores p de la Tabla 2, se concluye que tanto la longitud como el diámetro de los pellets son altamente significativos sobre la conversión del monóxido de carbono. El efecto combinado entre el diámetro y la longitud arrojó un valor F de 70,62 (el valor p más bajo), lo cual indica que la interacción entre estas dos variables es altamente significativa.
Un modelo es considerado significativo cuando el valor p es menor que 0,05 y los valores p que exceden 0,100 no juegan un papel importante en el modelo. Por tanto, en el caso de la conversión de monóxido de carbono, la longitud y el diámetro de los pellets son estadísticamente significativos. A partir del modelo anterior se consideró un modelo de regresión cuadrático en el diámetro y lineal en los factores, este modelo se presenta en la siguiente sección.
Modelo estadístico
El análisis de varianza realizado previo al modelo, permite predecir que la ecuación de regresión de los datos experimentales debe incluir tanto el efecto lineal del diámetro y la longitud, y un efecto cuadrático en el diámetro, así como el efecto combinado de cada una de estas variables. El modelo de regresión resultante, que predice la conversión (en escala logarítmica), a partir de una dependencia lineal tanto de la longitud x1, como del diámetro x2, una relación cuadrática del diámetro y la interacción entre las dos variables se presenta en la Ecuación [4].
Todos los parámetros asociados al modelo ajustado de [4] fueron significativos. El diámetro y la longitud de los pellets con coeficientes 0,14033 y 0,11762, respectivamente, tienen un efecto positivo sobre el logaritmo de la conversión del monóxido de carbono. Del modelo se puede inferir que los pellets con mayor diámetro y longitud son los más activos en la reacción. Igualmente, se observa que la interacción entre estas dos variables tiene un efecto negativo (-0,202) sobre la conversión del monóxido de carbono, la cual decrece cuando disminuye tanto el diámetro como la longitud, parámetros relacionados con la porosidad y el tiempo de retención de los reactivos en el lecho catalítico.
Para un lecho catalítico empacado, la adsorción se debe evaluar en función del volumen, porque los huecos donde no se produce la adsorción se forman entre las partículas como resultado del empaquetamiento, siendo mayor a menor diámetro y longitud, afectando la densidad aparente del sistema catalítico. La adecuación del modelo ajustado se corroboró con el valor del coeficiente de correlación R2 = 0,941, una medida de la variación alrededor de la media explicada por el modelo considerado, asegurando un buen ajuste a los datos experimentales.
El modelo matemático para la conversión del monóxido de carbono se utilizó para construir la superficie de respuesta (Figura 2) y las gráficas de contorno (Figura 3) las cuales son útiles para visualizar la dependencia y la interacción de cada una de las variables.
Figura 2.
La Figura 2 muestra que la región de mínima conversión de monóxido de carbono se localizó en la región de mínimo diámetro y longitud de los pellets, también se observa que el efecto de un aumento en el diámetro es mayor que un aumento en la longitud. Este efecto está relacionado con la disminución de la densidad aparente y el aumento de la porosidad del lecho. Los resultados muestran que los pellets de 0,4 cm de diámetro y 0,6 cm de longitud son los más activos en la reacción de desplazamiento de agua dando una conversión de 96%. Adicionalmente, el gráfico de contornos (Figura 3) reveló que existen interacciones significativas entre el diámetro y la longitud. Sin embargo, no se observa ni un punto de máxima, ni de mínima en la conversión del monóxido de carbono, aunque se comprueba que un aumento de la longitud y del diámetro mejora la conversión del monóxido de carbono.
Figura 3.
Caracterización de los catalizadores
La Tabla 3 muestra los resultados del área superficial de los pellets evaluados. Estos resultados muestran que el área superficial de los pellets fue aproximadamente igual, variando entre 402 y 442 m2·min-1, por lo cual la diferencia en actividad catalítica de los pellets no estaría asociada al área superficial de los mismos. Comparada con el área superficial del carbón activado (891 m2·min-1), la de los pellets disminuyó entre 55 y 50%. Esta diferencia en área superficial sería consecuencia de la incorporación de los metales y las altas temperaturas de calcinación y reducción (600°C) a las que se someten las muestras.
D: diámetro-L: longitud Tabla 3.
Análisis químico
Los resultados del contenido de Cu y Ni se muestran en la Tabla 4. Allí se observa que la carga de metales obtenida por espectroscopía de absorción atómica es menor a la carga nominal (20% p/p CuO + NiO), probablemente debido a pérdidas por lixiviación de los precursores, Cu(NO3)2·3H2O y Ni(NO3)2·6H2O durante la preparación del catalizador por impregnación húmeda. Sin embargo, no es posible descartar posibles interferencias del soporte (carbón activado) en la espectroscopía de absorción atómica (24). Aunque la relación molar Cu-Ni real de los catalizadores preparados no es igual a la relación nominal (Cu-Ni = 2-1), varía en un rango muy estrecho (1,8-1 a 2,3-1). Esto permite concluir que la diferencia en la actividad catalítica se debe principalmente al tamaño de los pellets y no al contenido de Cu y Ni en los mismos.
D-L: diámetro- longitud
Resultados de la actividad catalítica
La Figura 4 muestra la conversión de monóxido de carbono en la reacción de desplazamiento de gas de agua sobre los catalizadores peletizados de Cu-Ni en función de la temperatura entre 180 y 350 °C.
Figura 4
Los resultados de la Figura 4 muestran que la actividad catalítica es afectada significativamente por la temperatura en el rango 180-250 °C. Lo anterior concuerda con los datos previamente reportados por Lin y colaboradores (25), Chen y colaboradores (9) y Arbeláez y colaboradores (10), quienes encontraron conversiones del monóxido de carbono menores al 20% para temperaturas inferiores a 240 °C. A temperaturas superiores, la conversión de CO aumentó con la temperatura, se obtienen conversiones de CO alrededor del 95% a 350 °C; el catalizador con el menor diámetro y longitud mostró una conversión de CO alrededor del 65%. Sin embargo, como los pellets usados tienen diferente tamaño (diámetro y longitud) pero conservan un volumen empacado de 1,5 cm3 y el mismo peso total del lecho catalítico empacado a través del cual fluyen los reactivos, la evaluación del modelo estadístico permitió correlacionar la interacción favorable entre estas dos variables del sistema y proponer el modelo más adecuado para estimar la conversión del monóxido de carbono en términos del diámetro y la longitud.
Conclusiones
Se desarrolló un modelo estadístico para obtener las condiciones más adecuadas en la reacción de desplazamiento de agua a partir de catalizadores bimetálicos de Cu-Ni:2-1 en forma de pellets cilíndricos de carbón activado. Un diseño factorial con dos factores indicó que tanto la longitud como el diámetro de los pellets influyen en la conversión del monóxido de carbono. Además se observó que el efecto principal de cada variable fue muy significativo al igual que el respectivo efecto combinado. Se encontró una mayor conversión del monóxido a mayor diámetro y longitud de los pellets: con los pellets de 0,4 cm de diámetro y 0,6 cm de longitud se obtuvo una mayor conversión de monóxido de carbono en la reacción de desplazamiento de agua.
Agradecimientos
Los autores agradecen a la Universidad Cooperativa de Colombia por el apoyo financiero a través de proyecto 1539, a la Universidad de Antioquia y a la Universidad Nacional de Colombia.
Referencias
Referencias
Dou, B.; Song, Y.; Wang, C.; Chen, H.; Xu, Y. Hydrogen production from catalytic steam reforming of biodiesel byproduct glycerol: Issues and challenges. Renew Sustain Energy Rev 2014, 30 950–60. DOI: http://dx.doi.org/10.1016/j.rser.2013.11.029.
Lucia, U.; Overview on fuel cells. Renew Sustain Energy Rev 2014, 30, 164–9. DOI: http://dx.doi.org/10.1016/j.rser.2013.09.025.
Jain, R.; Maric, R. Synthesis of nano-Pt onto ceria support as catalyst for water–gas shift reaction by Reactive Spray Deposition Technology. Appl Catal A Gen 2014 475, 461–8. DOI: http://dx.doi.org/10.1016/j.apcata.2014.01.053.
Panagiotopoulou, P.; Kondarides, D. Effects of promotion of TiO2 with alkaline earth metals on the chemisorptive properties and water–gas shift activity of supported platinum catalysts. Appl Catal B Environ 2011 101, 738–46. DOI: http://dx.doi.org/10.1016/j.apcatb.2010.11.016.
Andreeva, D.; Idakiev, V.; Tabakova, T.; Ilieva, L.; Falaras, P.; Bourlinos A, et al. Low-temperature water-gas shift reaction over Au/CeO2 catalysts. Catal. Today 2002, 72, 51–7. DOI: http://dx.doi.org/10.1016/S0920-5861(01)00477-1.
Rodriguez, J.; Gold-based catalysts for the water–gas shift reaction: Active sites and reaction mechanism. Catal Today 2011, 160, 3–10. DOI: http://dx.doi.org/10.1016/j.cattod.2010.06.030.
Karakaya, C.; Otterstätter, R.; Maier, L.; Deutschmann, O. Kinetics of the water-gas shift reaction over Rh/Al2O3 catalysts. Appl Catal A Gen 2014, 470, 31–44. DOI: http://dx.doi.org/10.1016/j.apcata.2013.10.030.
Goscianska, J.; Ziolek, M.; Gibson, E.; Daturi, M. Novel mesoporous zirconia-based catalysts for WGS reaction. Appl Catal B Environ 2010, 97, 49–56. DOI: http://dx.doi.org/10.1016/j.apcatb.2010.03.023.
Chen, C.; Ruan, C.; Zhan, Y.; Lin, X.; Zheng, Q.; Wei, K. The significant role of oxygen vacancy in Cu/ZrO2 catalyst for enhancing water–gas-shift performance. Int J Hydrogen Energy 2014, 39, 317–24. DOI: http://dx.doi.org/10.1016/j.ijhydene.2013.10.074.
Arbeláez, O.; Reina, T.; Ivanova, S.; Bustamante, F.; Villa, A.; Centeno, M.; Odriozola, J.; Mono and bimetallic Cu-Ni structured catalysts for the water gas shift reaction.
Appl Catal A Gen 2015, 497, 1–9. DOI: http://dx.doi.org/10.1016/j.apcata.2015.02.041.
Lin, J.; Guliants, V.; Alumina-supported Cu-Ni and Ni-Cu core–shell nanoparticles: Synthesis, characterization, and catalytic activity in water–gas-shift reaction. Appl Catal A Gen 2012, 445-446, 187–94. DOI: http://dx.doi.org/10.1016/j.apcata.2012.08.013.
Saw, E.; Oemar, U.; Tan, X.; Du, Y.; Borgna, A.; Hidajat, K.; et al. Bimetallic Ni–Cu catalyst supported on CeO2 for high-temperature water–gas shift reaction: Methane suppression via enhanced CO adsorption. J Catal 2014, 314, 32–46. DOI: http://dx.doi.org/10.1016/j.jcat.2014.03.015.
Rad, A.; Khoshgouei, M.; Rezvani, S.; Rezvani, A. Study of Cu–Ni/SiO2 catalyst prepared from a novel precursor, [Cu(H2O)6][Ni(dipic)2].2H2O/SiO2, for water gas shift reaction. Fuel Process Technol 2012, 96, 9–15. DOI: http://dx.doi.org/10.1016/j.fuproc.2011.12.015.
Zhang, X.; Zhang, Y.; Liu, Q.; Zhou, W. Surface properties of activated carbon from different raw materials. Int J Min Sci Technol 2012, 22, 483–6. DOI: http://dx.doi.org/10.1016/j.ijmst.2012.01.007.
Akhtar, F.; Andersson, L.; Ogunwumi, S.; Hedin, N.; Bergström, L. Structuring adsorbents and catalysts by processing of porous powders. J Eur Ceram Soc 2014, 34, 1643–66. DOI: http://dx.doi.org/10.1016/j.jeurceramsoc.2014.01.008.
Bruner, E.; Puri, M. L. Nonparametric methods in factorial designs. Statistical papers. 2001, 42, 1-52.
R Core Team. (2015). R: A language and environment for statistical computing (Programa informático). Vienna: R Foundation for Statistical Computing. http://www.r-project.org/ 2015:2015 (consultado el 13 de abril de 2017)
Zarei, A.; Seyed, S.; Rezaei, M.; Meshkani, F. A comparative study of experimental investigation and response surface optimization of steam reforming of glycerol over nickel nano-catalysts. Int J Hydrogen Energy 2016, 41, 10178–92. DOI: http://dx.doi.org/10.1016/j.ijhydene.2016.05.047.
Moradi, M.; Daryan, J.; Mohamadalizadeh, A. Response surface modeling of H2S conversion by catalytic oxidation reaction over catalysts based on SiC nanoparticles using Box−Behnken experimental design. Fuel Process Technol 2013, 109, 163–71. DOI: http://dx.doi.org/10.1016/j.fuproc.2012.10.013.
Tounsadi, H.; Khalidi, A.; Farnane, M.; Abdennouri, M.; Barka, N. Experimental design for the optimization of preparation conditions of highly efficient activated carbon from Glebionis coronaria L. and heavy metals removal ability. Process Saf Environ Prot 2016, 102, 710–23. DOI: http://dx.doi.org/10.1016/j.psep.2016.05.017.
Orrego, A.; Arbeláez, O.; Bustamante, F.; Villa, A H. Pelletization of catalysts supported on activated carbon. A Case Study : clean synthesis of dimethyl carbonate from methanol and CO2 Revista Facultad de Ingeniería 2016, 78, 38–47. DOI: http://dx.doi.org/10.17533/udea.redin.n78a05.
Arbeláez, O.; Orrego, A.; Bustamante, F.; Villa, A. Direct Synthesis of Diethyl Carbonate from CO2 and CH3CH2OH over Cu–Ni/AC Catalyst. Top Catal 2012, 55, 668–72. DOI: http://dx.doi.org/10.1007/s11244-012-9849-4.
Montgomery, D. Diseño y análisis de experimentos, 2nd ed. Limusa Wesley: Mexico, 2002.
Welz, B.; Vale, M.; Pereira, E.; Castilho, I.; Dessuy, M. Continuum source atomic absorption spectrometry: past, present and future aspects – a critical review. J. Braz. Chem. Soc. 2012, 25, 799-821 DOI: http://dx.doi.org/10.5935/0103-5053.20140053.
Lin, J.; Biswas, P.; Guliants, V.; V. Misture, S. Hydrogen production by water–gas shift reaction over bimetallic Cu–Ni catalysts supported on La-doped mesoporous ceria. Appl Catal A Gen 2010, 387, 87–94. DOI: http://dx.doi.org/10.1016/j.apcata.2010.08.003.
Cómo citar
IEEE
ACM
ACS
APA
ABNT
Chicago
Harvard
MLA
Turabian
Vancouver
Descargar cita
CrossRef Cited-by
1. C.F. Imbachi-Gamba, A.L. Villa. (2021). Statistical analysis of the influence of synthesis conditions on the properties of hierarchical zeolite Y. Materials Today Chemistry, 20, p.100442. https://doi.org/10.1016/j.mtchem.2021.100442.
2. You Han, Yulian Wang, Tengzhou Ma, Wei Li, Jinli Zhang, Minhua Zhang. (2020). Mechanistic understanding of Cu-based bimetallic catalysts. Frontiers of Chemical Science and Engineering, 14(5), p.689. https://doi.org/10.1007/s11705-019-1902-4.
Dimensions
PlumX
Visitas a la página del resumen del artículo
Descargas
Licencia
Derechos de autor 2018 Revista Colombiana de Química

Esta obra está bajo una licencia internacional Creative Commons Atribución 4.0.
Los autores/as conservarán sus derechos de autor y garantizarán a la revista el derecho de primera publicación de su obra, el cuál estará simultáneamente sujeto a la Licencia de reconocimiento de Creative Commons (CC. Atribución 4.0) que permite a terceros compartir la obra siempre que se indique su autor y su primera publicación en esta revista.
Los autores/as podrán adoptar otros acuerdos de licencia no exclusiva de distribución de la versión de la obra publicada (p. ej.: depositarla en un archivo telemático institucional o publicarla en un volumen monográfico) siempre que se indique la publicación inicial en esta revista.
Se permite y recomienda a los autores/as difundir su obra a través de Internet (p. ej.: en archivos telemáticos institucionales o en su página web) antes y durante el proceso de envío, lo cual puede producir intercambios interesantes y aumentar las citas de la obra publicada. (Véase El efecto del acceso abierto).